Explore Key Companies Shaping the Influenza Vaccine Market

- GlaxoSmithKline plc
- Sanofi
- AstraZeneca
- CSL Limited
- BIKEN Co., Ltd.
- Abbott
- SINOVAC
- Viatris Inc.
Market Growth
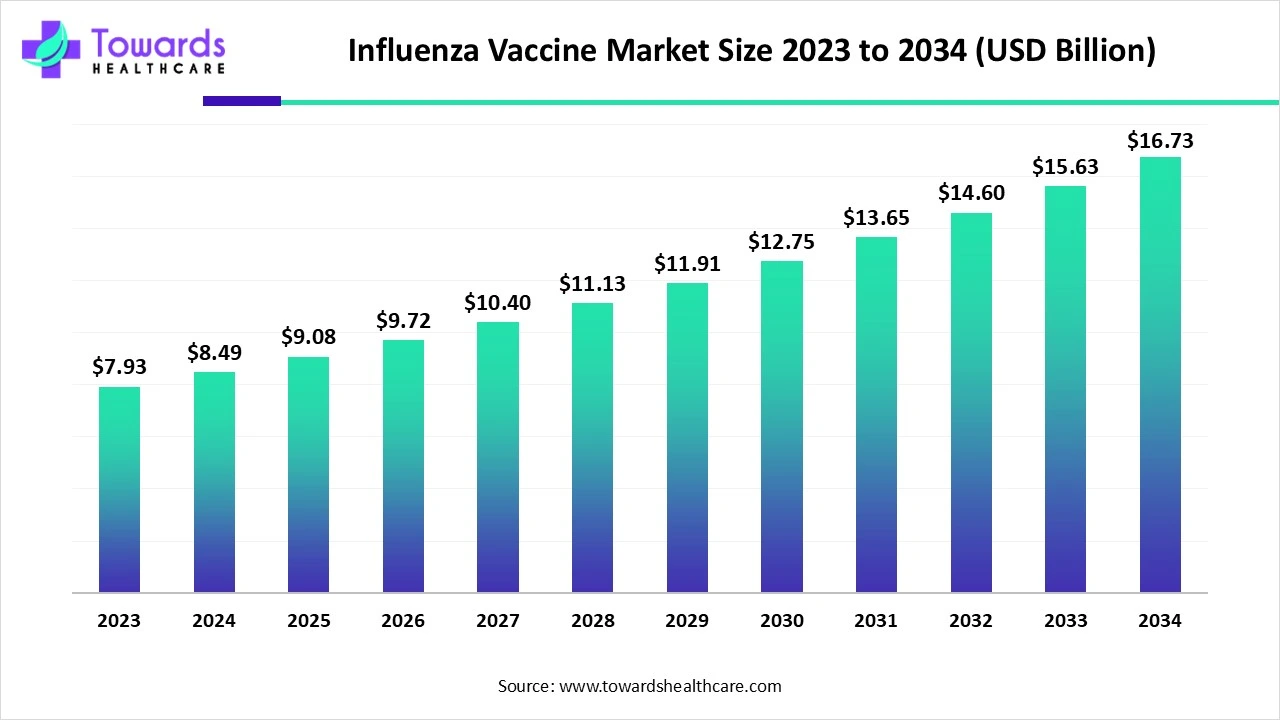
The global influenza vaccine market size is calculated at USD 8.49 billion in 2024, grew to USD 9.08 billion in 2025, and is projected to reach around USD 16.73 billion by 2034. The market is expanding at a CAGR of 7.02% between 2025 and 2034. The high occurrence of seasonal flu and growing recommendations for vaccination to prevent the illness, as well as the increasing geriatric population, drive the market growth, with complications arising from infection also contributing to market growth.
Influenza Vaccine Market Trends
- In June 2025, the Pan American Health Organization (PAHO) collaborated with the Ministry of Health Guyana to support the implementation of seasonal influenza vaccination in Guyana. The organizations will conduct seasonal influenza vaccination campaigns in the latter quarter of 2025.
- In March 2024, Osivax, a biopharmaceutical company, reported that all participants had completed their final visit in the Phase 2a clinical trial (NCT05734040). The trial was assessing OVX836, a broad-spectrum influenza A vaccine, as well as a candidate for quadrivalent influenza vaccines (QIVs).
Influenza Vaccine Market Outlook
- Industry Growth Overview: The market is expected to accelerate in the upcoming years, with increasing collaborations among intergovernmental organizations, growing awareness of disease prevention, and advances in vaccine manufacturing techniques.
- Major Investors: Government bodies hold the major share of investors, supporting the development and manufacturing of influenza vaccines. Private equity firms and venture capitalists also provide funding to enable companies to expand their manufacturing facilities.
- Global Expansion: Key players collaborate to expand their geographical locations and deliver their products to a wider patient population. Private firms collaborate with regulatory bodies to support global vaccination.
Strategic Initiatives
- The World Health Organization (WHO) launched the “Pandemic Influenza Preparedness Framework Partnership Contribution High-Level Implementation Plan III (2024-2030) Monitoring and Evaluation Framework” to facilitate influenza vaccine manufacturing.
- In May 2025, the U.S. Department of Health and Human Services (HHS) and the National Institute of Health (NIH) announced the development of a next-generation, universal vaccine platform, Generation Gold Standard. This initiative represents a decisive shift toward transparency, effectiveness, and comprehensive preparedness for influenza and coronavirus vaccines.
- The European Immunization Agenda 2030 (EIA2030) is an initiative and strategy developed by all 53 Member States to achieve the full benefits of vaccination in Europe. The strategy focuses on strengthening national immunization policies and addressing gaps in immunization coverage.
Top Companies’ Revenue Info.
- Moderna is an American pharmaceutical and biotech company that specializes in RNA therapeutics. The second-quarter 2025 revenue was $0.1 billion. The company projected annual revenue of $1.5 to $2.2 billion.
- CSL Seqirus is one of the world’s largest influenza vaccine providers and a major contributor to influenza prevention globally. The company generated a total revenue of $15.6 billion in FY2025, i.e., the year ended June 2025. It provides its products to more than 100 countries.
Top Companies’ Report
- GlaxoSmithKline reported total sales of £31.4 billion in 2024. It started shipping doses of its trivalent influenza vaccines, Flulaval and Fluarix, in July 2024, to the U.S. healthcare providers and pharmacies for the 2024-25 season. The company estimates to distribute 36 million doses of vaccines in the U.S. this season.
- In July 2024, CSL Seqirus started shipping its differentiated portfolio of influenza vaccines. CSL Seqirus is the only manufacturer in the U.S. to offer a differentiated influenza vaccine for all eligible people aged six months and above.
Value Chain Analysis – Influenza Vaccine Market
R&D
Advances in recombinant DNA technology and the increasing clinical evidence of mRNA vaccines enables researchers to develop novel vaccines based on these technologies.
Key Players: Bharat Biotech, Sanofi, and CSL Seqirus.
Clinical Trials & Regulatory Approvals
Clinical trials are essential to evaluate the efficacy, safety, tolerability, and immunogenicity of influenza vaccines. Regulatory agencies approve a vaccine based on clinical trial data.
Key Players: Osivax, BioNTech, Pfizer, and Moderna.
Distribution to Hospitals, Pharmacies
Distribution to hospitals and pharmacies involves a complex supply chain pathway that is managed by public and private companies. Hospitals and pharmacies increases vaccine distribution reach and capacity.
Latest Announcements by Industry Leaders
In July 2024, Sinergium Biotech in Argentina aims to accelerate the development of H5N1 mRNA vaccines for low- and middle-income countries (LMICs) through the WHO and Medicines Patent Pool’s mRNA Technology Transfer Programme. The initiative focuses on building vaccine production capacity and improving pandemic preparedness. Dr. Tedros Adhanom Ghebreyesus, Director-General of the WHO, stated, "This project ensures the world is better prepared for future pandemics." Charles Gore, Executive Director of MPP, added, "It enables LMICs to lead development and foster collaboration for better preparedness."
Recent Developments in the Influenza Vaccine Market
- In February 2024, the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency recommended approving two vaccines for active immunization against the H5N1 strain of the influenza A virus responsible for avian influenza, also known as bird flu.
- In March 2024, Cadila Pharmaceuticals in India introduced the Cadiflu Tetra Vaccine, an advanced quadrivalent influenza vaccine approved by the DCGI for both adults and children. Designed to target four influenza strains (A and B subtypes), the vaccine utilizes proprietary nanoparticle technology to replicate the virus’s structure without including its genetic material.
Contact our experts to explore the Influenza Vaccine Market at sales@towardshealthcare.com
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking

