Top Companies in the Non-Antibody Fusion Proteins Market & Their Offerings

| Companies | Headquarters | Offerings |
| Amgen (Immunex legacy) | California, USA | Fc-fusion proteins, such as etanercept (Enbrel), focus on autoimmune and inflammatory diseases. |
| Regeneron Pharmaceuticals + Bayer AG | New York, USA | Fusion protein-based biologics like aflibercept (VEGF-trap) for ophthalmology and oncology applications. |
| Bristol Myers Squibb | New York City, USA | Fusion protein therapeutics such as abatacept (CTLA-4 Ig) for autoimmune diseases. |
| Pfizer Inc. | New York City, USA | Development of protein-based biologics and fusion protein candidates targeting inflammation and rare diseases. |
| Eli Lilly and Company | Indiana, USA | Biologic therapies, including engineered protein constructs and fusion-based therapeutics for metabolic and autoimmune disorders. |
What are the Recent Developments in the Non-Antibody Fusion Proteins Market?
- In January 2025, Merck completed a $1.3 billion acquisition of the bispecific antibody CN201, strengthening its pipeline with a late-stage therapy targeting B-cell disorders.
- In January 2024, Abzena launched two advanced cell line development platforms, AbZelect and AbZelectPRO, designed to speed up the creation of production cell lines for recombinant proteins and antibodies. These systems help streamline the development of complex biologics, enabling faster transition from early research stages to clinical trials and regulatory submission.
Supply Chain Analysis
R&D
- Research and development in non-antibody fusion proteins centers on creating engineered molecules by linking functional protein components such as scaffolds, ligands, or enzymes to improve targeting, stability, and therapeutic effectiveness. These designs address key limitations of monoclonal antibodies, including large molecular size, high manufacturing costs, and limited tissue penetration.
- Key players: Amgen, Regeneron Pharmaceuticals, Bristol Myers Squibb, Sanofi, Takeda Pharmaceutical Company
Clinical Trials
- Clinical trials for non-antibody fusion proteins are increasingly exploring novel scaffold-based designs such as adnectins, Kunitz domains, and SIRPα to enhance targeting precision, stability, and half-life compared to traditional antibodies. Several candidates are progressing through mid-stage trials for autoimmune and hematologic conditions, reflecting strong clinical interest and therapeutic potential in this space.
- Key players: AbbVie, Pfizer, Novartis, Eli Lilly, AstraZeneca
Patient Support and Services
- Patient support and service strategies for non-antibody fusion proteins focus on maintaining consistent product quality, ensuring stability and safety, and optimizing pharmacokinetic management for better treatment outcomes. Compared to traditional antibodies, these therapies require specialized handling and monitoring to support effective and safe use in patients.
- Key players: Roche, Merck & Co., Gilead Sciences, Biogen, CSL Limited
Market Growth
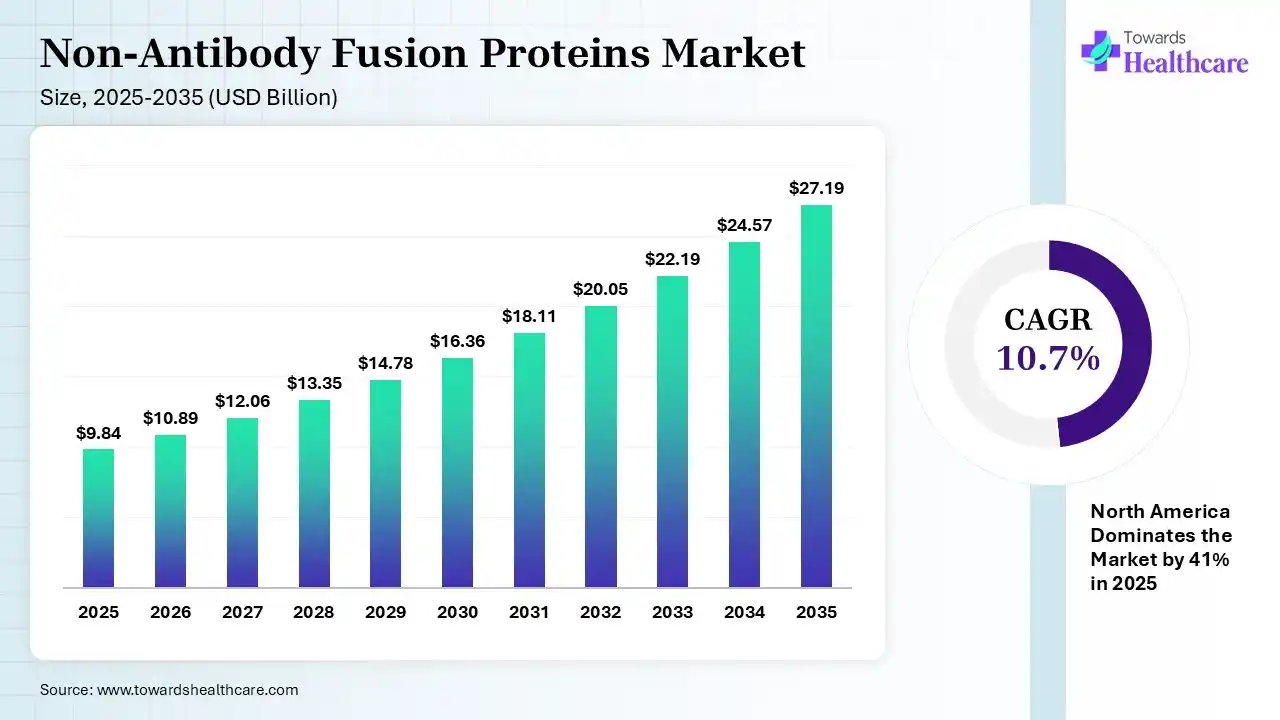
The global non-antibody fusion proteins market size was estimated at USD 9.84 billion in 2025 and is predicted to increase from USD 10.89 billion in 2026 to approximately USD 27.19 billion by 2035, expanding at a CAGR of 10.7% from 2026 to 2035.
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
