Let's See Top Companies in the Pemphigus Vulgaris Market
- Pfizer Inc.
- F. Hoffmann-La Roche Ltd.
- Sanofi
- Amgen Inc.
- GlaxoSmithKline plc (GSK)
- Novartis AG
- AstraZeneca
- AbbVie Inc.
- Teva Pharmaceutical Industries Ltd.
- Argenx SE
- Cabaletta Bio Inc.
- Daewoong Pharmaceutical Co. Ltd.
- HanAll Biopharma Co. Ltd.
- Merck & Co., Inc.
- GRIFOLS SA
- Boehringer Ingelheim International GmbH
- Regeneron Pharmaceuticals Inc.
- Johnson & Johnson Services, Inc.
- Zydus Group
- Alexion Pharmaceuticals
What is Pemphigus Vulgaris?
The global pemphigus vulgaris market includes pharmaceutical therapies, biologics, immunosuppressants, diagnostics, and supportive care solutions used for the treatment and management of pemphigus vulgaris, a rare autoimmune blistering disorder of the skin and mucous membranes.
The market covers corticosteroids, monoclonal antibodies, immunoglobulins, plasmapheresis, hospitalization services, and long-term disease management. Growth is driven by rising diagnosis rates, increasing awareness of rare autoimmune diseases, advancements in biologic therapies, improved access to specialty dermatology care, and expanding clinical research pipelines.
Market Growth
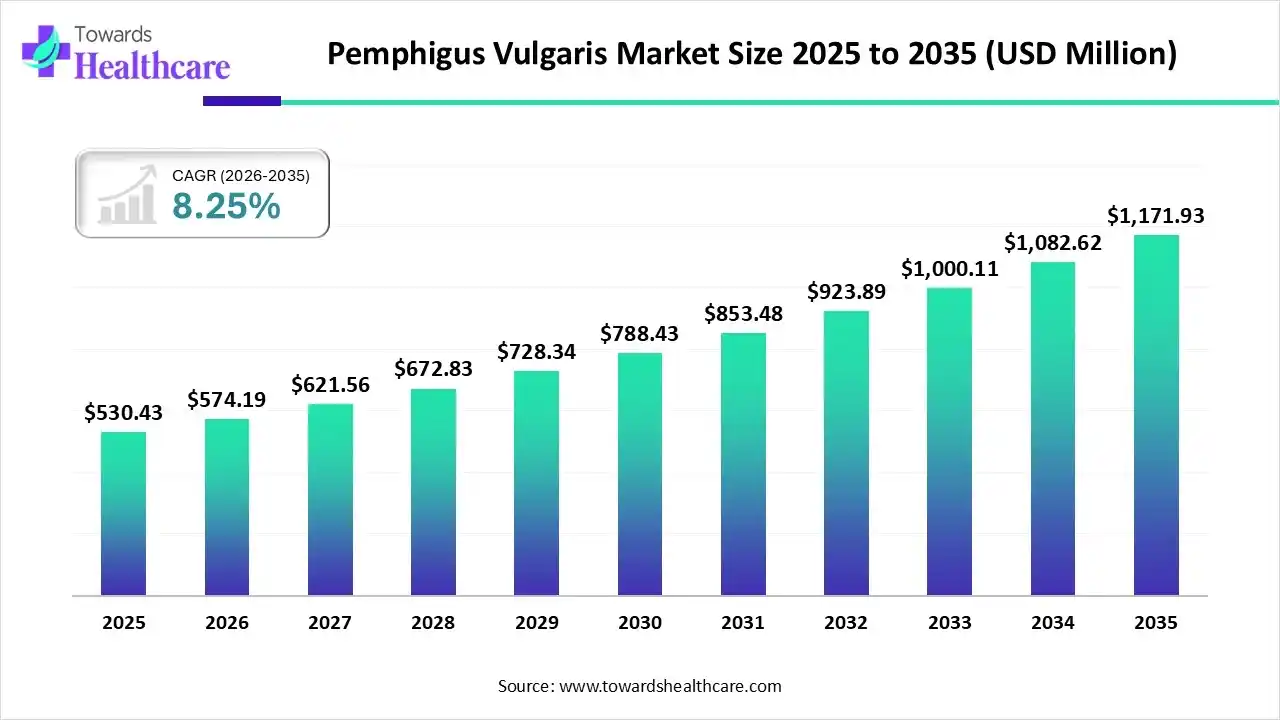
The global pemphigus vulgaris market size is calculated at US$ 530.43 million in 2025, grew to US$ 574.19 million in 2026, and is projected to reach around US$ 1171.93 million by 2035. The market is expanding at a CAGR of 8.25% between 2026 and 2035.
Top Vendors in the Pemphigus Vulgaris (PV) Market – Pointwise Summary
1. Pfizer Inc.
-
Key Product / Pipeline: Ruxience (rituximab-pvvr), a biosimilar to an anti-CD20 biologic.
-
Status / Contribution: Approved; competes with the reference biologic.
-
Advantages / Focus Area: Offers a cost-effective biosimilar alternative.
-
Challenges / Trial Info: Needs to maintain supply reliability and manage competition.
2. Sanofi
-
Key Product / Pipeline: Rilzabrutinib (PRN1008), an oral BTK inhibitor in Phase 3 for PV.
-
Status / Contribution: PEGASUS Phase 3 trial did not meet the primary endpoint.
-
Advantages / Focus Area: Small-molecule approach vs biologics; potential convenience benefits.
-
Challenges / Trial Info: Development setback due to failed Phase 3; reassessment ongoing.
3. Amgen Inc.
-
Key Product / Pipeline: Riabni (rituximab-arrx), a biosimilar to CD20 biologic used in PV.
-
Status / Contribution: Launched; competes in the biosimilar biologic market.
-
Advantages / Focus Area: Strong biologics-development and manufacturing expertise.
-
Challenges / Trial Info: Faces typical biosimilar competition and market pressures.
4. GlaxoSmithKline plc (GSK)
-
Key Product / Pipeline: Ofatumumab (subcutaneous) program for PV (via Stiefel, a GSK company).
-
Status / Contribution: Phase III study initiated for PV.
-
Advantages / Focus Area: Targets CD20 using an alternative antibody, expanding treatment options.
-
Challenges / Trial Info: Still investigational; not yet broadly approved for PV.
5. Novartis AG
-
Key Product / Pipeline: Ianalumab (VAY736), a BAFF-R inhibitor in Phase 3 trials.
-
Status / Contribution: Pipeline stage; positive topline data reported.
-
Advantages / Focus Area: Novel targeted mechanism offering a new potential treatment paradigm.
-
Challenges / Trial Info: Not approved yet; final efficacy and regulatory outcomes are pending.
Pemphigus Vulgaris Market Value Chain Analysis
Research and Development (R&D): Finding possible targets, creating novel medications, and carrying out preliminary research in labs are all part of this first phase. Driven by an understanding of autoimmune mechanisms, it focuses on novel treatments such as cell therapies, small molecules, and biologics.
Companies: Cabaletta Bio Inc, HanAll Biopharma Co Ltd, Argenx SE, BioXpress Therapeutics SA, Octagon Therapeutics Inc, ImmunoWork LLC, and Daewoong Pharmaceutical Co Ltd
Clinical Trials and Regulatory Approvals: After a medication is created, it goes through extensive clinical testing to evaluate its effectiveness and safety. After that, a request for regulatory approval from organizations like the FDA is made, necessitating a thorough evaluation before going on sale.
Companies: Cabaletta Bio, Argenx SE, Roche (F. Hoffmann-La Roche Ltd), Dr. Reddy's Laboratories, Principia Biopharma, Genentech, Inc., and Kemia, Inc
Distribution to Hospitals, Pharmacies: Distribution channels transfer the medication from producers to final consumers following approval. To guarantee patient access, this covers both hospital and retail pharmacies as well as internet pharmacies.
Companies: F. Hoffmann-La Roche Ltd, Pfizer Inc, AbbVie Inc, CSL Behring GmbH, Novartis AG, Teva Pharmaceutical Industries Ltd, and Johnson & Johnson Services, Inc.
Company Landscape
F. Hoffmann-La Roche Ltd. (Roche)
Corporate Information:
- Headquarters: Basel, Switzerland
- Year Founded: 1896
- Ownership Type: Publicly Traded (SIX Swiss Exchange: ROG)
History and Background:
- Founded by Fritz Hoffmann-La Roche, initially focusing on vitamin preparations and later expanding into pharmaceutical and diagnostic products.
- Became a pioneer in oncology and, more recently, a leader in personalized healthcare.
Key Milestones/Timeline:
- 1896: Company founding.
- Mid-20th Century: Major contributions to psychotropic drugs (e.g., Valium).
- 1990s-2000s: Emergence as a leader in oncology with products like Herceptin and Avastin.
- 2006: FDA approval of Rituximab (Rituxan/MabThera) for non-Hodgkin's lymphoma, later expanding to autoimmune conditions.
- 2018: FDA approval of Rituxan (rituximab) for the treatment of moderate to severe pemphigus vulgaris (PV) in adults, marking a major advancement.
Business Overview:
A global healthcare company with two main divisions: Pharmaceuticals and Diagnostics.
Business Segments/Divisions:
- Pharmaceuticals: Focuses on developing and commercializing therapeutic medicines, primarily in oncology, immunology, infectious diseases, and neuroscience.
- Diagnostics: Provides a broad portfolio of diagnostic solutions, including in vitro diagnostics, tissue diagnostics, and point-of-care testing.
Geographic Presence:
Global presence, with major operations in North America, Europe, Asia-Pacific, and emerging markets. North America is a significant revenue driver.
Key Offerings:
- Rituxan/MabThera (rituximab): Anti-CD20 monoclonal antibody (a cornerstone of PV treatment).
- Extensive portfolio in oncology, immunology (e.g., Ocrevus, Actemra), ophthalmology, and hematology.
End-Use Industries Served:
- Hospitals and Specialty Clinics
- Research Institutes and Laboratories
- Physician Offices/Ambulatory Surgical Centers
Key Developments and Strategic Initiatives:
- Mergers & Acquisitions: Continues to strategically acquire biotech companies to strengthen its pipeline in neuroscience, oncology, and immunology.
- Partnerships & Collaborations: Engages in numerous R&D collaborations with academic institutions and biotech firms to explore novel targets and digital health solutions.
- Product Launches/Innovations: Focus on next-generation biologics and subcutaneous formulations of existing intravenous therapies.
- Capacity Expansions/Investments: Ongoing investments in manufacturing capabilities for biologics and advanced diagnostic systems.
- Regulatory Approvals: Rituxan is the only FDA-approved biologic for PV. Continues to pursue approvals for new indications and novel molecules.
Distribution Channel Strategy:
Primarily focused on Hospital Pharmacies and Specialty Pharmacies due to the nature of complex biologic infusions and the requirement for specialized handling and administration (e.g., intravenous Rituximab).
Technological Capabilities/R&D Focus:
- Core Technologies/Patents: Expertise in monoclonal antibodies, small molecules, and personalized medicine platforms driven by advanced diagnostics.
- Research & Development Infrastructure: Robust global R&D network, with a high R&D spend focused on addressing high-unmet need areas like rare autoimmune diseases.
- Innovation Focus Areas: Personalized healthcare, leveraging real-world data and AI for drug discovery, and developing novel targeted therapies including FcRn inhibitors for autoimmune conditions.
Competitive Positioning:
- Strengths & Differentiators: The pioneer and market leader in anti-CD20 therapy for PV (Rituxan). Strong legacy and expertise in immunology and complex biologics. Integrated Pharmaceutical and Diagnostics divisions.
- Market presence & ecosystem role: Dominant player in the autoimmune and oncology markets globally. Sets the standard of care for anti-CD20 therapy.
SWOT Analysis:
- Strengths: Global leadership in biologics; established effectiveness and FDA approval of Rituxan for PV; large financial resources for R&D.
- Weaknesses: High cost of biologics; patent expiration and competition from biosimilars (e.g., Truxima, Ruxience).
- Opportunities: Further pipeline development in targeted PV therapies (e.g., FcRn inhibitors); expansion into emerging markets; utilizing diagnostics to improve PV patient identification.
- Threats: Aggressive competition from biosimilar manufacturers and novel therapeutic classes (e.g., BTK inhibitors, CAR-T).
Recent News and Updates (2024-2025):
- Press Releases (Example Focus): Continued communication regarding the use and efficacy of Rituximab in autoimmune disorders, including data presentations at major dermatology and rheumatology congresses in 2024.
- Industry Recognitions/Awards: Routinely recognized as a top global pharmaceutical company for innovation and R&D investment.
AbbVie Inc.
Corporate Information:
- Headquarters: North Chicago, Illinois, USA
- Year Founded: 2013 (Spun off from Abbott Laboratories)
- Ownership Type: Publicly Traded (NYSE: ABBV)
History and Background:
- Formed as a research-based pharmaceutical company specializing in complex biologic medicines, separating from Abbott Laboratories to focus on biopharma.
- Inherited the blockbuster drug Humira (adalimumab), establishing a dominant position in the immunology market.
Key Milestones/Timeline:
- 2013: Spin-off from Abbott Laboratories.
- Early 2010s: Focus on growing the immunology franchise with Humira.
- 2019: Acquired Allergan, significantly expanding its portfolio into neuroscience and aesthetics.
- 2023-2024: Faced the loss of exclusivity for Humira in the US, leading to a strong focus on next-generation immunology drugs and biosimilar strategies.
- 2024-2025: Increased focus on expanding its biosimilar presence for key biologics, including Rituximab biosimilars, in various global markets.
Business Overview:
A global biopharmaceutical company dedicated to developing and commercializing advanced therapies across key therapeutic areas.
Business Segments/Divisions:
- Immunology: Core focus area, including treatments for rheumatoid arthritis, psoriasis, Crohn's disease, and other autoimmune conditions.
- Oncology: Products for various cancers.
- Neuroscience: Treatments for diseases like migraine and movement disorders (expanded via Allergan acquisition).
- Aesthetics: (Allergan Aesthetics)
Geographic Presence:
Significant global footprint, with major revenues from the United States and strong presence across Europe, Asia-Pacific, and Latin America.
Key Offerings:
- Rituximab Biosimilars: Key to their presence in the PV market, competing with Roche's branded Rituxan.
- Humira (adalimumab): TNF inhibitor (though not approved for PV, it dominates the autoimmune market).
- Skyrizi (risankizumab) & Rinvoq (upadacitinib): Next-generation immunology products.
End-Use Industries Served:
- Hospitals and Specialty Clinics
- Retail and Specialty Pharmacies
- Managed Care Organizations
Key Developments and Strategic Initiatives:
- Mergers & Acquisitions: The large-scale acquisition of Allergan (2019) diversified its portfolio. Focused on smaller, strategic acquisitions to bolster its pipeline.
- Partnerships & Collaborations: Collaborations for drug discovery and pipeline development, particularly in immunology and oncology.
- Product Launches/Innovations: Heavy investment in launching and scaling next-generation immunology drugs (Skyrizi, Rinvoq) and launching biosimilars to maintain market share.
- Capacity Expansions/Investments: Investment in manufacturing and R&D to support the growth of its new immunology and oncology pipelines.
- Regulatory Approvals: Securing approvals for new immunology products in various indications and geographies. Actively promoting biosimilars for Rituximab globally.
Distribution Channel Strategy:
- Leverages a robust Specialty Pharmacy and Retail Pharmacy network for self-administered products like Humira, Skyrizi, and Rinvoq.
- Uses Hospital/Infusion Center channels for intravenous biosimilars of Rituximab.
Technological Capabilities/R&D Focus:
- Core Technologies/Patents: Expertise in monoclonal antibodies (TNF, IL-23, JAK inhibition), small molecule development, and biosimilar manufacturing.
- Research & Development Infrastructure: Major R&D centers globally, with significant investment in developing immunology and oncology assets.
- Innovation Focus Areas: Developing therapies with improved dosing and efficacy for complex autoimmune disorders, and maintaining a leadership position in immunology post-Humira loss of exclusivity.
Competitive Positioning:
- Strengths & Differentiators: Global leader in immunology (historically Humira). Strong commercial infrastructure and deep expertise in autoimmune markets. Aggressive strategy in the biosimilar space to capture market share from branded biologics like Rituxan.
- Market presence & ecosystem role: High market access and established relationships with payers and healthcare providers due to its legacy in immunology. A key driver in the biosimilar segment of the PV market.
SWOT Analysis:
- Strengths: Leading immunology portfolio; diversified therapeutic areas; strong global commercial presence.
- Weaknesses: Major revenue risk due to Humira biosimilar competition; reliance on a few blockbuster drugs.
- Opportunities: Growth of next-generation immunology assets (Skyrizi, Rinvoq); increasing market penetration of its Rituximab biosimilar to compete directly in the PV market.
- Threats: Competition from novel PV therapies (e.g., CAR-T, FcRn inhibitors); aggressive pricing from other biosimilar manufacturers.
Recent News and Updates (2024-2025):
- Press Releases (Example Focus): Announcements in early 2024 regarding the successful commercial launch and increased market share of its biosimilars in ex-US markets. Continued positive Phase III data announcements for next-generation immunology drugs.
- Industry Recognitions/Awards: Recognized for best-in-class product portfolios and patient support programs in the immunology space.
Recent Developments in the Pemphigus Vulgaris Market
- In October 2025, initial dose data from the RESET-PVTM trial, which evaluated rese-cel (resecabtagene autoleucel, formerly known as CABA-201) at 1 x 106 cells/kg without preconditioning in three evaluable patients with pemphigus vulgaris (PV), were presented by Cabaletta Bio, Inc., a clinical-stage biotechnology company dedicated to creating and introducing the first curative targeted cell therapies specifically for patients with autoimmune diseases.
- In March 2025, the Japanese Ministry of Health, Labour, and Welfare has approved Chugai (a subsidiary of Roche) and Zenyaku Kogyo's co-marketed Rituxan® (rituximab) for children with "frequently relapsing or steroid-dependent nephrotic syndrome" (IV injection, 100 mg and 500 mg) that is not yet incurable.
Partner with our experts to explore the Cell Culture Microcarriers Market at sales@towardshealthcare.com
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
