Which are the Top Companies in the Antithrombin Market? Let's See!
- CSL Limited
- Grifols, S.A.
- Takeda Pharmaceutical Company Limited (which acquired Shire Plc)
- Octapharma AG
- LFB USA
- Kedrion S.p.A
- Lee Biosolutions
- Scripps Laboratories
- rEVO Biologics, Inc.
- Thermo Fisher Scientific
- Siemens Healthcare GmbH
- Diapharma Group, Inc.
- Merck KGaA
- Pfizer Inc.
- Sanofi
- Novartis AG
- Bio-Techne Corporation
- Biocon Ltd.
- Jiangsu Hengrui Medicine Co., Ltd.
- China Biologic Products, Inc.
What is Antithrombin?
The antithrombin market is driven by the increasing prevalence of thrombosis, rising use in surgical and obstetric care, adoption in neonatal/pediatric care, growing biologics manufacturing, wider application in rare disease management, and advances in recombinant technology that improve production scalability and safety profiles.
The global antithrombin market covers the production, purification, formulation, distribution, and clinical use of antithrombin (AT) products, including antithrombin III concentrates derived from plasma, recombinant antithrombin produced through bioengineering, and antithrombin used in research and diagnostics. Antithrombin is an essential serine protease inhibitor used to prevent and treat thrombotic complications in patients with hereditary antithrombin deficiency, disseminated intravascular coagulation (DIC), sepsis-related coagulopathy, cardiopulmonary bypass surgery, and in heparin-resistant conditions.
Market Growth
The global antithrombin market size is calculated at US$ 420.33 in 2025, grew to US$ 438.4 million in 2026, and is projected to reach around US$ 640.37 million by 2035. The market is expanding at a CAGR of 4.3% between 2026 and 2035.
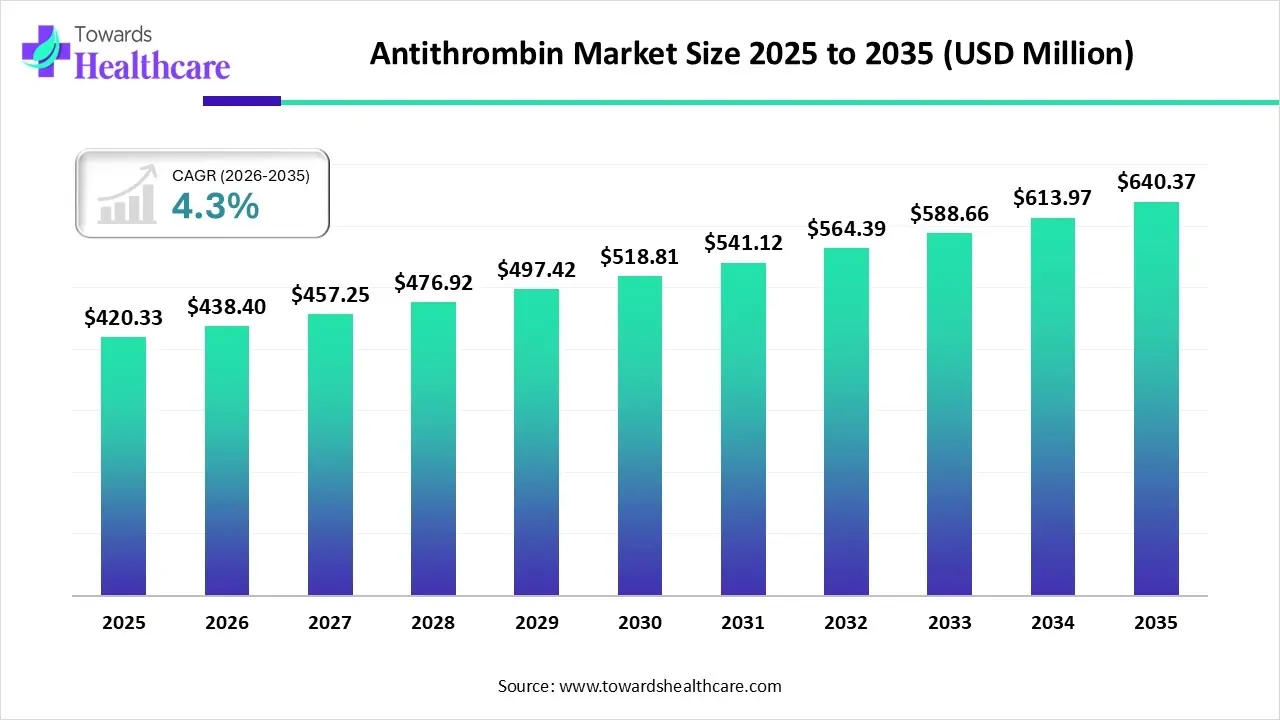
Antithrombin Market Outlook
- Industry Growth Overview: The rising number of surgical procedures requiring anticoagulation therapy and the rising prevalence of blood clotting disorders are driving the market. Recombinant antithrombin is one example of a biotechnology advancement that improves safety and efficacy and has sustained market growth over the past ten years.
- Global Expansion: Because of its sophisticated healthcare system and high level of awareness, North America currently has the largest antithrombin market share. Due to the increasing incidence of thrombotic diseases in China and India as well as better healthcare facilities, the Asia-Pacific region is expected to grow at the fastest rate.
- Major Investors: Grifols, CSL, Takeda Pharmaceutical Company, and Octapharma AG are important investors and businesses. These players participate in R&D funding and strategic initiatives for innovative treatments, such as the Grifols/Endpoint Health partnership for the treatment of sepsis.
Top Vendors in the Antithrombin Market & Their Offerings
Takeda Pharmaceutical Company Limited
- Offerings: Plasma-derived antithrombin; coagulation therapies
- Contributions to Antithrombin Market: Expands global therapeutic access and supports critical-care use
- Market Role: Major global supplier
- Key Highlights: Strong regulatory focus and large manufacturing base
Octapharma AG
- Offerings: Human plasma-derived antithrombin concentrates
- Contributions to Antithrombin Market: Ensures reliable supply for surgical and ICU thrombosis management
- Market Role: Leading European producer
- Key Highlights: Robust plasma-fractionation capabilities
LFB USA
- Offerings: Antithrombin concentrates
- Contributions to Antithrombin Market: Serves hereditary AT deficiency and specialized clinical needs
- Market Role: Niche U.S. biotherapeutics player
- Key Highlights: Focus on rare-disease biologics
Kedrion S.p.A
- Offerings: Plasma-derived antithrombin
- Contributions to Antithrombin Market: Expands donor-based production for hospitals worldwide
- Market Role: Key plasma-therapy provider
- Key Highlights: Broad global plasma-collection network
Lee Biosolutions
- Offerings: Antithrombin reagents and biomarkers
- Contributions to Antithrombin Market: Supports diagnostics, assays, and research applications
- Market Role: Specialized biochemical supplier
- Key Highlights: Enables lab testing and innovation in AT analysis
Company Landscape
1. Grifols, S.A.
Company Overview
- Corporate Information (Headquarters, Year Founded, Ownership Type):
- Headquarters: Sant Cugat del Vallès, Barcelona, Spain
- Year Founded: 1909 (precursor laboratory)
- Ownership Type: Publicly traded (MCE: GRF, MCE: GRF.P, NASDAQ: GRFS)
- History and Background:
- Pioneered in the plasma industry, tracing roots to a clinical analysis lab established in 1909.
- A global leader in essential plasma-derived medicines and transfusion medicine.
- One of the world's largest plasma collection networks (approximately 400 centers).
- Key Milestones/Timeline:
- 1988: Opened first subsidiary in Portugal, beginning international expansion.
- 2006: Listed on the Madrid stock exchange.
- 2011: Began trading on NASDAQ following the acquisition of Talecris Biotherapeutics.
- 2025 (Nov): Received expanded FDA approval for THROMBATE III in pediatric patients.
- Business Overview:
- Focuses on the research, development, manufacturing, and commercialization of plasma-derived medicines and other biopharmaceuticals.
- The largest plasma collector in Europe and third largest worldwide.
- Total revenue was €7.2 billion in 2024.
- Business Segments/Divisions:
- Biopharma: Plasma-derived therapies (including antithrombin, immunoglobulins, albumin, alpha-1 antitrypsin, etc.).
- Diagnostic: Transfusion medicine, clinical and immunological diagnostics.
- Bio Supplies: Biological supplies for research and manufacturing.
- Others: Includes Grifols Engineering, which designs and builds plasma and manufacturing facilities.
- Geographic Presence:
- Provides products and solutions in more than 110 countries.
- Plasma collection network spans North America, Europe, Africa, and the Middle East, with a presence in China through a partnership.
- Key Offerings:
- Antithrombin: THROMBATE III® (Antithrombin III [Human]) - indicated for hereditary antithrombin deficiency (hATd).
- Other Biologics: IG, Albumin, A1PI, Coagulation factors.
- End-Use Industries Served:
- Hospitals and Specialty Clinics (especially Intensive Care, Hematology, Pulmonology, Neurology).
- Transfusion and Clinical Diagnostic Laboratories.
- Life-Science Research.
- Key Developments and Strategic Initiatives:
- Mergers & Acquisitions: Successfully completed the delisting of Biotest in June 2025 following its acquisition.
- Partnerships & Collaborations: Completed the sale of a 20% equity stake in Shanghai RAAS (SRAAS) and forged a strategic alliance with Haier Group in June 2024.
- Product Launches/Innovations: Grifols' Biotest received its first regulatory approval for its new fibrinogen concentrate (Prufibry®) in Germany (November 2025).
- Capacity Expansions/Investments: Targeting a plasma fractionation capacity of 26 million liters per year by 2026.
- Regulatory Approvals: Received U.S. FDA expanded indication for THROMBATE III® to include pediatric patients with hATd (November 2025).
- Distribution channel strategy: Highly integrated global plasma collection, fractionation, and distribution network ensures a stable supply chain for its plasma-derived products.
- Technological Capabilities/R&D Focus:
- Core Technologies/Patents: Proprietary plasma fractionation processes and viral inactivation technologies for high-quality plasma products.
- Research & Development Infrastructure: Over 1,200 researchers across 12 R&D centers globally.
- Innovation Focus Areas: Plasma-derived treatments, recombinant antibody drugs, and new therapies for neurological, immunological, and hematological disorders.
- Competitive Positioning:
- Strengths & Differentiators: Vertically integrated business model (plasma collection to final product), large and growing global plasma collection network, and a strong pipeline for rare and chronic diseases.
- Market presence & ecosystem role: A dominant global player in the plasma protein therapeutics market, serving a critical role as a primary supplier of life-saving, plasma-derived medicines.
- SWOT Analysis:
- Strengths: Vertical integration, leading global plasma collector, strong revenue growth in Biopharma (12.1% in Q3 2024).
- Weaknesses: Significant debt load, reliance on plasma supply.
- Opportunities: Expansion into China via SRAAS/Haier partnership, new indications for existing therapies (e.g., pediatric THROMBATE III approval).
- Threats: Competition from recombinant and synthetic alternatives, regulatory scrutiny, plasma collection variability.
- Recent News and Updates:
- Press Releases: November 18, 2025: Grifols receives expanded U.S. FDA indication for THROMBATE III to include pediatric patients with hereditary antithrombin deficiency.
- Industry Recognitions/Awards: Named one of TIME magazine’s "World’s Best Companies 2025" and one of Forbes’ World's Best Employers for the second consecutive year.
2. CSL Behring (CSL Limited)
Company Overview
- Corporate Information (Headquarters, Year Founded, Ownership Type):
- Headquarters: Melbourne, Victoria, Australia (CSL Limited) / King of Prussia, Pennsylvania, USA (CSL Behring)
- Year Founded: 1916 (Commonwealth Serum Laboratories)
- Ownership Type: Publicly traded (ASX: CSL, OTC: CSLLY)
- History and Background:
- Originated as a government-owned body in Australia. Privatized in 1994.
- CSL Behring was established through the integration of Centeon, Aventis Behring, and ZLB Bioplasma, focusing on plasma protein biotherapeutics.
- Key Milestones/Timeline:
- 1916: Founded as Commonwealth Serum Laboratories in Australia.
- 2004: Acquisition of Aventis Behring forms CSL Behring.
- 2022: Completed the acquisition of Vifor Pharma, expanding into iron deficiency and nephrology.
- 2025 (Nov): Announced investment of approximately $1.5 billion in the U.S. to manufacture plasma-derived therapies.
- Business Overview:
- A leading global biotechnology company driven by a promise to save lives and protect the health of people.
- Discovers, develops, manufactures, and markets biopharmaceuticals.
- CSL Group Revenue for FY2025 was US$15.6 billion.
- Business Segments/Divisions:
- CSL Behring: Global leader in plasma protein biotherapies, including antithrombin, immunoglobulins, albumin, and coagulation factors.
- CSL Seqirus: Global leader in influenza vaccines.
- CSL Vifor: Global leader in iron deficiency and nephrology (acquired in 2022).
- CSL Plasma: Operates one of the world's largest plasma collection networks (over 349 centers).
- Geographic Presence:
- Delivers products to patients in more than 100 countries.
- Major manufacturing presence in the US, Germany, Switzerland, and Australia.
- Key Offerings:
- Antithrombin: ATNF (Antithrombin Human) and Thrombate III (in certain markets/legacy products). CSL Behring's primary product for AT replacement is a plasma-derived concentrate.
- Other Biologics: HAE treatments (e.g., ANDEMBRY®), Immunoglobulins (Hizentra®, Privigen®), Recombinant and Plasma-derived Coagulation Factors.
- End-Use Industries Served:
- Hospitals and Specialty Clinics (Hematology, Immunology, Critical Care).
- Dialysis and Nephrology Centers (through CSL Vifor).
- Public Health Programs (via CSL Seqirus vaccines).
- Key Developments and Strategic Initiatives:
- Mergers & Acquisitions: Focus on integrating CSL Vifor to strengthen its position in specialty pharmaceuticals.
- Partnerships & Collaborations: Partnered with Dutch biotech VarmX BV in September 2025 to develop a first-in-class investigational coagulation treatment. Signed an LOI with the pan-Canadian Pharmaceutical Alliance (pCPA) in October 2025 for public reimbursement of its Hemophilia B gene therapy, HEMGENIX®.
- Product Launches/Innovations: Launched ANDEMBRY® (garadacimab) in Japan and received EU/Swiss approval in early 2025 for Hereditary Angioedema (HAE).
- Capacity Expansions/Investments: Major investment of approximately $1.5 billion announced in November 2025 for U.S. plasma-derived therapies manufacturing.
- Regulatory Approvals: Received Swissmedic and European Commission approval for ANDEMBRY® (garadacimab) in HAE prevention (Feb 2025).
- Distribution channel strategy: Utilizes a combination of direct sales and distribution partners to serve its global market, leveraging its large CSL Behring business for plasma product distribution.
- Technological Capabilities/R&D Focus:
- Core Technologies/Patents: Expertise in plasma fractionation, recombinant protein technology, and emerging cell and gene therapy platforms.
- Research & Development Infrastructure: Over 2,000 R&D employees globally; R&D investment of $5.8 billion over the last 5 years.
- Innovation Focus Areas: Advancing treatments in hematology, immunology, respiratory/cardiovascular/metabolic disorders, and vaccines. Strong focus on gene therapy (e.g., HEMGENIX®).
- Competitive Positioning:
- Strengths & Differentiators: Broad and diversified portfolio across CSL Behring, Seqirus, and Vifor; global R&D footprint; strong financial performance (Net Profit After Tax of US$3.0 billion in FY25); integrated plasma collection.
- Market presence & ecosystem role: A dominant force in global plasma therapies, flu vaccines, and specialty pharmaceuticals, holding a key position in rare disease treatment markets.
- SWOT Analysis:
- Strengths: Diversified revenue streams (plasma, vaccines, nephrology), strong R&D pipeline, global scale, industry-leading quality/safety standards.
- Weaknesses: High cost of plasma sourcing, market competition for innovative therapies.
- Opportunities: Growth of CSL Vifor in nephrology, expansion of gene therapy (HEMGENIX®), and geographic market penetration (e.g., MEA).
- Threats: Development of synthetic/recombinant alternatives to plasma products, public health crises impacting plasma collection.
- Recent News and Updates:
- Press Releases: November 18, 2025: CSL announces approximately $1.5 billion investment in U.S. manufacturing for plasma-derived therapies. October 26, 2025: CSL Honored as Overall Winner of the 2025 ISPE Facility of the Year Awards for its Broadmeadows facility.
- Industry Recognitions/Awards: Overall Winner of the 2025 ISPE Facility of the Year Awards (Pharma 4.0 category).
Antithrombin Market Value Chain Analysis
R&D
It includes preclinical testing to find novel antithrombin products and comprehend mechanisms, as well as basic research and target identification (such as SERPINC1 gene variants).
Companies involved in the research and development phase include Grifols, CSL Behring, Takeda, Octapharma, LFB, Kedrion, and Siemens Healthineers.
Clinical Trials and Regulatory Approvals
It involves using human subjects in a series of trial phases to demonstrate safety and efficacy before submitting and receiving regulatory approval (e.g., FDA, EMA).
Companies managing clinical trials and seeking regulatory approvals include Grifols, CSL Behring, Takeda, Octapharma, LFB, Kedrion, and Siemens Healthineers.
Formulation and Final Dosage Preparation
It involves converting the active pharmaceutical ingredient (API) into a final product (e.g., injectable solution, specific dosage units) that is stable and ready for the market.
Companies responsible for formulating and preparing the final antithrombin products for market include Grifols, CSL Behring, Takeda, Octapharma, LFB, Kedrion, and Siemens Healthineers.
Recent Developments in the Antithrombin Market
- In November 2025, the U.S. Food and Drug Administration (FDA) approved an expanded indication for THROMBATE III, the company's antithrombin III [human concentrate], to include pediatric patients with hereditary antithrombin deficiency (hATd), according to a statement released by Spanish plasma-based drug manufacturer Grifols (MCE: GRF).
- In March 2025, the first antithrombin-lowering (AT) treatment, Qfitlia (fitusiran), was approved by the U.S. Food and Drug Administration (FDA) for routine prophylaxis to prevent or lessen the frequency of bleeding episodes in adult and pediatric patients (aged 12 or older) with hemophilia A or B, with or without factor VIII or IX inhibitors.
Collaborate with our experts to explore the Antithrombin Market at sales@towardshealthcare.com

