Learn About the Major Players in the Bioanalytical Testing Services Market
- BioAgilytix
- Eurofins Scientific
- Precision for Medicine
- Illumina
- Bio-Rad Laboratories
- Altasciences
- QPS
- SpinoS Life Science
- IntoxLab
- Thermo Fisher Scientific
- Syneos Health
Company Profile
BioAgilytix
Overview
-
BioAgilytix is a leading provider of contract research services, specializing in large molecule bioanalytical testing to support the biopharmaceutical industry.
Top Products
-
Bioanalytical testing services for large molecule drugs.
Annual Revenue
-
Approx. $0.3 billion (2024 estimate).
Their Offerings
-
Specialized bioanalytical services, including immunoassays, biomarker analysis, and other laboratory services supporting the development of biopharmaceuticals.
Recent News or Update
-
BioAgilytix continues to expand its testing capabilities, particularly in the areas of cell and gene therapies, as well as its international presence.
Eurofins Scientific
Overview
-
Eurofins Scientific is a global group offering laboratory services in the fields of food, environment, pharmaceuticals, and biotechnology.
Top Products
-
Laboratory services for life sciences, including testing and analysis in food safety, environmental sciences, and pharmaceutical R&D.
Annual Revenue
-
$5.6 billion (2024 estimate).
Their Offerings
-
A wide range of laboratory services, including analytical testing, clinical diagnostics, and biotechnology research and development services.
Recent News or Update
-
Eurofins has continued to grow through acquisitions, expanding its capabilities in clinical diagnostics and pharmaceutical testing.
Precision for Medicine
Overview
-
Precision for Medicine provides a range of services aimed at improving the precision of clinical trials and drug development, with a focus on oncology, immunology, and rare diseases.
Top Products
-
Precision clinical trials and personalized medicine services for the biopharmaceutical industry.
Annual Revenue
-
$0.5 billion (2024 estimate).
Their Offerings
-
Services in clinical trials, laboratory testing, and biomarker discovery, supporting personalized medicine development in therapeutic areas like oncology and immunology.
Recent News or Update
-
Precision for Medicine has been enhancing its services in genomic testing and biomarkers, aiming to advance personalized medicine for clinical trials.
Illumina
Overview
-
Illumina is a global leader in DNA sequencing and array-based technologies, supporting genetic analysis for research, diagnostics, and drug development.
Top Products
-
DNA sequencing systems, gene expression analysis tools, and genomic testing products.
Annual Revenue
-
$5.1 billion (2024 estimate).
Their Offerings
-
High-throughput DNA sequencing platforms, reagents, and bioinformatics tools to enable genomic research and personalized healthcare.
Recent News or Update
-
Illumina continues to lead in next-generation sequencing technologies, with ongoing innovations in genomic testing for personalized medicine and clinical diagnostics.
Bio-Rad Laboratories
Overview
-
Bio-Rad Laboratories is a multinational biotechnology company that develops and manufactures products for life science research and clinical diagnostics.
Top Products
-
Diagnostic and research products, including immunoassays and PCR-based systems.
Annual Revenue
-
$2.7 billion (2024 estimate).
Their Offerings
-
Products for laboratory diagnostics, research, and clinical testing, including life science reagents, PCR systems, and protein analysis tools.
Recent News or Update
-
Bio-Rad continues to innovate in clinical diagnostics, with a focus on molecular diagnostics and next-generation research tools.
Altasciences
Overview
-
Altasciences is a contract research organization (CRO) that provides services for the pharmaceutical and biotechnology industries, specializing in early-stage drug development.
Top Products
-
Early-stage drug development services, clinical trials, and laboratory services.
Annual Revenue
-
Approx. $0.15 billion (2024 estimate).
Their Offerings
-
Preclinical and clinical development services, including toxicology, clinical trials, and laboratory services aimed at advancing drug discovery.
Recent News or Update
-
Altasciences is expanding its global presence and capabilities in clinical trials and preclinical testing, especially in the areas of early-stage drug development.
QPS
Overview
-
QPS is a global contract research organization (CRO) providing preclinical and clinical research services for the pharmaceutical, biotechnology, and medical device industries.
Top Products
-
Preclinical and clinical research services for drug development.
Annual Revenue
-
$0.1 billion (2024 estimate).
Their Offerings
-
A broad range of preclinical and clinical research services, including bioanalytical testing, toxicology studies, clinical trial management, and more.
Recent News or Update
-
QPS has been expanding its capabilities in clinical trials and regulatory affairs, focusing on providing comprehensive services for drug development.
SpinoS Life Science
Overview
-
SpinoS Life Science is a biotechnology company focused on providing services in clinical trials, R&D, and laboratory testing, particularly in the field of precision medicine.
Top Products
-
Clinical trial services and precision medicine testing.
Annual Revenue
-
Approx. $0.02 billion (2024 estimate).
Their Offerings
-
Clinical trial management services and R&D in precision medicine, focusing on innovative therapies and treatments for various diseases.
Recent News or Update
-
SpinoS Life Science is increasing its focus on precision medicine and genetic testing in clinical trials, aiming to support the development of targeted therapies.
IntoxLab
Overview
-
IntoxLab provides laboratory testing services specializing in alcohol and drug testing for forensic, clinical, and workplace environments.
Top Products
-
Alcohol and drug testing services for clinical and forensic purposes.
Annual Revenue
-
Approx. $0.01 billion (2024 estimate).
Their Offerings
-
Laboratory services for toxicology testing, focusing on alcohol and drug testing in various industries and environments.
Recent News or Update
-
IntoxLab is enhancing its forensic testing services, focusing on expanding its offerings in clinical toxicology.
Thermo Fisher Scientific
Overview
-
Thermo Fisher Scientific is a leading global provider of laboratory instruments, chemicals, reagents, and services for the healthcare, research, and industrial markets.
Top Products
-
Laboratory instruments, reagents, diagnostics, and biotechnology products.
Annual Revenue
-
$40.0 billion (2024 estimate).
Their Offerings
-
A wide range of scientific services and products, including laboratory reagents, instruments, clinical diagnostics, and drug discovery technologies.
Recent News or Update
-
Thermo Fisher continues to invest in expanding its research and diagnostics capabilities, including acquisitions to strengthen its life sciences and clinical research segments.
Syneos Health
Overview
-
Syneos Health is a global biopharmaceutical solutions organization that provides integrated biopharmaceutical solutions for clinical trials, regulatory services, and commercialization.
Top Products
-
Clinical trials and commercialization services for biopharmaceutical companies.
Annual Revenue
-
$2.6 billion (2024 estimate).
Their Offerings
-
Integrated solutions for the entire drug development lifecycle, from clinical trials to regulatory services and commercialization.
Recent News or Update
-
Syneos Health is enhancing its global capabilities in drug development and commercialization, with a focus on accelerating the delivery of therapies to market.
Market Growth
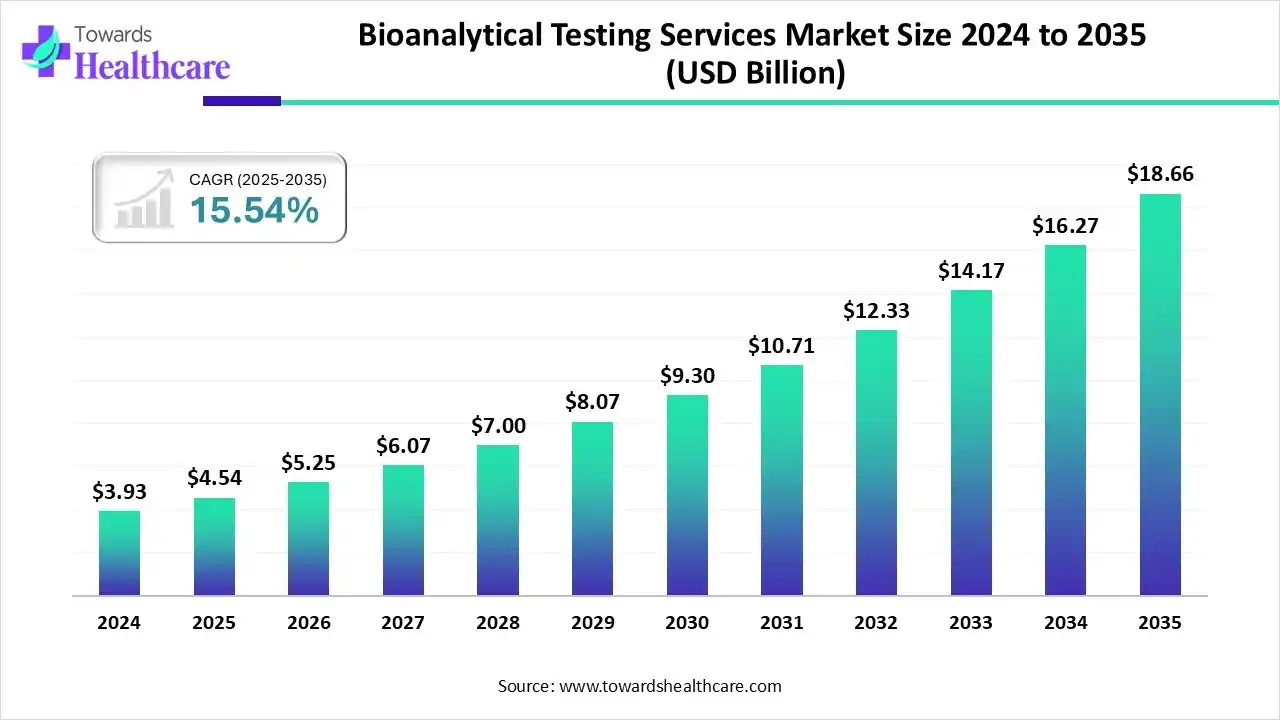
The global bioanalytical testing services market size is calculated at USD 4.54 billion in 2025, grew to USD 5.25 billion in 2026, and is projected to reach around USD 18.66 billion by 2035. The market is expanding at a CAGR of 15.54% between 2026 and 2035.
Other Major Company's Landscape:
Charles River Laboratories International, Inc. (CRL)
Company Overview : Charles River Laboratories is a leading global contract research organization (CRO) providing essential products and services to help pharmaceutical, biotechnology, medical device companies, and academic/government institutions accelerate their research and drug development efforts. It is a full-service, early-stage CRO.
Corporate Information (Headquarters, Year Founded, Ownership Type)
- Headquarters: Wilmington, Massachusetts, U.S.
- Year Founded: 1947
- Ownership Type: Public (NYSE: CRL)
- History and Background : Founded by Henry Foster, a veterinarian, who established a lab in Boston to supply local researchers with quality laboratory animals. It grew from a supplier of research models to a comprehensive, full-service, early-stage CRO through organic growth and strategic acquisitions.
Key Milestones/Timeline
- 1947: Founded.
- 1955: Headquarters relocated to Wilmington, MA.
- 2003: Merged with Inveresk (a U.K. research company specializing in clinical research and preclinical testing).
- 2014: Acquired Argenta and BioFocus, establishing full-service, early-stage CRO capabilities (target discovery through preclinical development).
- 2021: Acquired Cognate BioServices for $875 million, significantly expanding Cell and Gene Therapy Contract Development and Manufacturing Organization (CDMO) capabilities.
- Business Overview : Provides integrated drug discovery, non-clinical development, and safety assessment services, along with research models and manufacturing support services.
Business Segments/Divisions
- Research Models and Services (RMS): Research models, genetic models and services, and insourcing solutions.
- Discovery and Safety Assessment (DSA): Discovery, regulated safety assessment, and bioanalytical services.
- Manufacturing Support (Manufacturing): Microbial Solutions, Biologics Testing Solutions, and Advanced Therapies (Cell and Gene Therapy CDMO).
- Geographic Presence : Global operations spanning at least 20+ countries with over 60 research and testing facilities worldwide (as of January 2025). Significant presence in North America, Europe, and Asia.
Key Offerings
- Bioanalytical Services: LC/MS/MS, High-resolution MS (HRMS), Immunochemistry (PK/PD, Immunogenicity), Cell-Based Assays, and specialized support for large molecule, cell, and gene therapy programs.
- Safety Assessment
- Discovery Services
- Research Models & Services
- Biologics Testing and CDMO Services
End-Use Industries Served
- Pharmaceutical and Biotechnology Companies
- Academic Institutions
- Government Agencies
- Medical Device Companies
Key Developments and Strategic Initiatives
Mergers & Acquisitions
- Nov 2023: Acquired Noveprim, enhancing non-human primate (NHP) supply chain capabilities.
- Jan 2023: Acquired SAMDI Tech, Inc., expanding high-throughput screening capabilities.
- Jan 2025: Made a Later Stage VC investment in Deciphex (Other Healthcare Technology Systems).
Partnerships & Collaborations
Continued strategic focus on integrated service models, offering clients end-to-end support from discovery to commercial manufacturing.
Product Launches/Innovations
- Ongoing expansion of non-animal testing capabilities through the development and adoption of New Approach Methodologies (NAMs).
- Focus on Next-Generation Sequencing (NGS) for animal-free pathogen testing in biologics.
Capacity Expansions/Investments
- Investments of $300 million in alternative technologies, with an emphasis on NAMs.
- Implementation of a cost reduction program targeting $75 million in annual savings over several years (as of Q2 2025).
- Launched the Charles River Incubator Program in Wilmington, MA (2024).
Regulatory Approvals
Maintains key global accreditations and compliance standards, including FDA and EMA compliance, and AAALAC International Accreditation.
Distribution channel strategy
Primarily direct sales through dedicated global sales and scientific teams, emphasizing integrated, end-to-end solutions, particularly for early-stage development clients.
Technological Capabilities/R&D Focus
Core Technologies/Patents
- Proprietary research model breeding and maintenance technologies.
- Advanced Cell and Gene Therapy manufacturing platforms (via Cognate BioServices acquisition).
- Proprietary microbial detection solutions.
Research & Development Infrastructure
Extensive global network of state-of-the-art research and testing facilities.
Innovation Focus Areas
- New Approach Methodologies (NAMs): Focus on in vitro models (human cell-based assays, organoids), in silico techniques (AI, machine learning predictive modeling), and in chemico methods to reduce reliance on animal testing (as of Oct 2025).
- Cell and Gene Therapy Services: Continual investment and innovation in CDMO services for advanced therapeutics.
Competitive Positioning
Strengths & Differentiators
- Integrated Early-Stage Expertise: Unique, fully integrated offering from discovery (including research models) through safety assessment and early clinical support.
- Market Leadership in Research Models: The largest supplier of research models globally.
- Strong Financial Performance: Q2 2025 non-GAAP operating margin improvement across all three segments.
Market Presence & Ecosystem Role
- Pivotal player in the life sciences research ecosystem, critical for both large pharmaceutical and smaller biotechnology firms.
- Approximate market share of 35-40% in the broader preclinical research services market.
SWOT Analysis
- Strengths: Integrated service model, global infrastructure, market leadership in research models, strong cash generation.
- Weaknesses: High dependence on animal models (despite NAMs' investment), operational complexity due to diverse segments, and regulatory/ethical scrutiny regarding animal welfare.
- Opportunities: Growing demand for Cell and Gene Therapy services, expansion of NAMs, improvement in biotech funding (expected in H2 2025), targeted expansion in high-growth regions like China.
- Threats: Increased price competition from smaller CROs (particularly in Safety Assessment), cyclical demand from smaller biotech firms, and potential budget fluctuations in government R&D funding.
Recent News and Updates
Press Releases
- Oct 15, 2025: Announced the creation of a global, cross-functional Scientific Advisory Board to guide the strategic focus on New Approach Methodologies (NAMs).
- Aug 6, 2025: Announced Second-Quarter 2025 Results, with non-GAAP diluted EPS increasing 11.4% and non-GAAP operating margin improving across all three segments.
Industry Recognitions/Awards
Information not explicitly found in recent, concise news snippets, but generally recognized as a leading global CRO.
Labcorp Drug Development (A Segment of Labcorp)
Company Overview: Labcorp Drug Development, formerly Covance, is a global contract research organization providing a full suite of drug development services, from preclinical testing and clinical trials to commercialization. It is a key segment of the larger Labcorp life sciences company.
- Corporate Information (Headquarters, Year Founded, Ownership Type)
- Headquarters (Parent Company: Labcorp): Burlington, North Carolina, U.S.
- Year Founded (Parent Company: Labcorp): 1978 (as Roche BioMedical)
- Ownership Type: Public (NYSE: LH)
- History and Background: The Drug Development segment was significantly shaped by the 2015 acquisition of Covance by Labcorp, which established Labcorp as a powerhouse in both clinical diagnostics and drug development services. Labcorp itself has a history of strategic mergers and acquisitions to build its extensive testing network.
Key Milestones/Timeline
- 1978: Founded (as Roche BioMedical).
- 1995: Laboratory Corporation of America Holdings was established via a merger.
- 2015: Acquired Covance, transforming the company's drug development capabilities.
- 2021: Acquired Toxikon (nonclinical contract research) and Personal Genome Diagnostics (PGDx) (oncology testing).
- Sep 2024: Completed establishment of a new laboratory in Greenfield, expanding molecular bio analytics capability.
- Sep 2025: Completed the acquisition of select oncology and clinical testing assets from BioReference Health.
- Business Overview: The Drug Development segment offers end-to-end solutions to the pharmaceutical, biotechnology, and medical device industries, including bioanalytical, clinical development, central laboratory, and preclinical services.
Business Segments/Divisions
- Diagnostics Laboratories: Provides clinical laboratory testing services (Parent company's largest segment).
- Biopharma Laboratory Services (Drug Development): Offers comprehensive drug development, medical device, and diagnostic development services, which include bioanalytical testing.
- Geographic Presence: Global presence, serving clients in more than 100 countries. Operates one of the largest clinical laboratory networks in the world, with a strong presence in the U.S.
Key Offerings
- Bioanalytical Services: LC/MS/MS, Immunoassay, Cell-Based Assays, and specialized large molecule and regulated bioanalysis (e.g., PK/PD, immunogenicity, biomarker testing).
- Central Laboratory Services
- Clinical Development Services
- Preclinical (Nonclinical) Safety Assessment
- Companion Diagnostics
End-Use Industries Served
- Pharmaceutical Companies
- Biotechnology Companies
- Medical Device and Diagnostic Companies
- Hospitals and Healthcare Providers (via parent company)
Key Developments and Strategic Initiatives
Mergers & Acquisitions
- Sep 2025: Completed acquisition of select oncology and clinical testing assets from BioReference Health, further solidifying oncology leadership.
- Oct 2025 (Expected Close): Signed an agreement to acquire select outreach laboratory assets from Community Health Systems across 13 states.
- Oct 2025: Signed an agreement to acquire select assets of Empire City Laboratories (New York and New Jersey).
Partnerships & Collaborations
- Signed several strategic agreements with health systems and regional/local laboratories in 2025 to expand market reach and service offerings.
- Sep 2025: Announced a collaboration with Roche to advance digital pathology capabilities.
Product Launches/Innovations
- Oct 2025: To offer the first FDA-Cleared Blood Test to rule out Alzheimer 's-related Amyloid Pathology in primary care settings.
- Jul 2025: Introduced Labcorp Test Finder, a generative AI tool to simplify lab test search.
- Apr 2025: Launched Molecular Residual Disease and Liquid Biopsy Solutions.
- Expanded its oncology and genetic testing portfolio, including the expanded use of OmniSeq® INSIGHT.
Capacity Expansions/Investments
- Completed a new laboratory establishment in Greenfield (Sep 2024) to expand molecular bio analytics capability.
- Ongoing investment in digital and AI capabilities to enhance areas like pathology, cytology, and microbiology.
Regulatory Approvals
- Jul 2025: Achieved IVDR CE-Marking for PGDx elio™ tissue complete in the EU.
- Oct 2025: Announced offering of the first FDA-Cleared Blood Test for Alzheimer's Disease (fully launched Aug 2025).
Distribution channel strategy
Leverages its extensive network of patient-service centers and laboratories for diagnostics, while the Drug Development segment utilizes a direct sales model for CRO services to biopharma clients.
Technological Capabilities/R&D Focus
Core Technologies/Patents
- Advanced capabilities in genomic testing (PCR, next-generation sequencing).
- Expertise in molecular diagnostics and advanced biomarker testing.
Research & Development Infrastructure
Extensive global laboratory infrastructure, including its central lab and specialized testing facilities.
Innovation Focus Areas
- Personalized Medicine and Oncology: Leading in advanced testing for precision medicine, including liquid biopsy, genetic testing, and companion diagnostics.
- Digital Pathology and AI: Investment in AI and digital tools to enhance efficiency and customer experience.
- Neurology and Alzheimer's Disease: Focus on developing and launching new tests for neurological conditions.
Competitive Positioning
Strengths & Differentiators
- Scale and Scope: Dual leadership in both clinical diagnostics and drug development (including bioanalytical).
- Strong Financial Performance: Q3 2025 revenue for Biopharma Laboratory Services was $799.1 million, up 8.3% organically.
- Unmatched Network: Extensive network of patient service centers and laboratories enhances data collection and clinical trial support.
Market presence & ecosystem role
- One of the largest global life sciences companies; a dominant force in both the clinical lab and CRO markets.
- Drug Development segment annual revenue (2023 data) is approximately $4.2 billion.
SWOT Analysis
- Strengths: Vast scale, comprehensive testing portfolio (5,000+ tests), strong financial profile, integrated clinical and drug development services, active strategic acquisition strategy (2025).
- Weaknesses: High dependence on payer mix and reimbursement policies in the Diagnostics segment, complex regulatory environment, and involvement in various legal actions (common in the industry).
- Opportunities: Rapid growth in personalized and genomic medicine, expansion into international markets, leveraging AI and digital health technologies, and strategic acquisitions to expand market reach.
- Threats: Intense competition in the diagnostic market (e.g., Quest Diagnostics), changing reimbursement models, emerging technologies (e.g., point-of-care testing), and disrupting traditional models.
Recent News and Updates
Press Releases
- Oct 28, 2025: Announced 2025 Third Quarter Results, with the Biopharma Laboratory Services segment revenue at $799.1 million (8.3% organic growth). Updated full-year guidance with a higher Adjusted EPS range.
- Oct 23, 2025: To offer the first FDA-Cleared Blood Test to rule out Alzheimer 's-Related Amyloid Pathology in primary care.
Industry Recognitions/Awards
Information not explicitly found in recent, concise news snippets, but generally recognized as a global leader in laboratory and drug development services.
Top Companies & Their Offerings in the Bioanalytical Testing Services Market
- Charles River Laboratories: The company has experience of over 40 years in bioanalytical services to manage both small-scale studies and large clinical trials for numerous clients.
- ChemExpress: ChemExpress analysis service platform provides integrated services for quality control at all stages of drug development using more than 300 sophisticated instruments and high-level databases.
- IQVIA Laboratories: IQVIA Laboratories offers complete bioanalytical and ADME laboratory services for testing around the globe and across the product development spectrum.
- Intertek: The UK-based company provides GLP and GCP bioanalytical laboratory and bioanalysis testing support services for all phases of drug development, including method development and validation.
- SGS SA: SGS aims to control costs, increase efficiency, and accelerate clinical programs with its globally integrated bioanalytical solutions. The company has recently gained expertise in advanced ELISA equipment and the Gyrolab xPlore system.
Recent Developments in the Bioanalytical Testing Services Market
- In July 2025, Bio-Rad Laboratories, Inc. expanded its range of recombinant monoclonal anti-idiotypic antibodies and SpyCatcher reagents. The launch of novel products facilitates the development of robust and reliable bioanalytical assays against biologic drugs, as well as pharmacokinetic and anti-drug-antibody assays.
- In February 2025, Ardena announced the acquisition of a state-of-the-art drug product manufacturing facility from Catalent, located in Somerset. The company plans to open a new bioanalytical lab as part of its expanding services in North America to provide advanced analytical testing services for both small and large molecules
Partner with our experts to explore the Bioanalytical Testing Market at sales@towardshealthcare.com
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
