Top Companies in the Biosimilar Antibodies Market & Their Offerings

| Companies | Headquarters | Offerings |
| Roche | Basel, Switzerland | Focuses on oncology and immunology biologics, with biosimilar development through partnerships and strong expertise in monoclonal antibodies. |
| Mylan | Pennsylvania, USA | Broad biosimilar portfolio including monoclonal antibodies for oncology and autoimmune diseases, often developed in collaboration with global partners. |
| Teva Pharmaceutical Industries | Tel Aviv, Israel | Engages in biosimilar antibody development with a focus on oncology and immunology, leveraging large-scale manufacturing and global distribution. |
| Biogen | Massachusetts, USA | Develops and commercializes biosimilars primarily in immunology, with strong capabilities in biologics manufacturing and partnerships. |
| Fresenius Kabi | Bad Homburg, Germany | Offers a growing range of biosimilar monoclonal antibodies, focusing on oncology and autoimmune therapies with emphasis on hospital-based treatments. |
| Sandoz | Basel, Switzerland | A global leader in biosimilars, providing a wide portfolio of monoclonal antibodies across oncology and immunology, supported by strong R&D and commercialization capabilities |
What are the Recent Developments in the Biosimilar Antibodies Market?
- In February 2025, Celltrion received European Commission approval for several biosimilars, including Eydenzelt (referencing Eylea) for retinal disorders, and Stoboclo and Osenvelt (referencing Prolia and Xgeva), covering all approved uses of their original biologic counterparts.
- In December 2024, Biocon Biologics Ltd received approval from the U.S. Food and Drug Administration for YESINTEK, a biosimilar of Stelara. This monoclonal antibody is indicated for multiple conditions, including Crohn’s disease, ulcerative colitis, plaque psoriasis, and psoriatic arthritis.
Supply Chain Analysis
R&D
- Biosimilar antibody R&D focuses on detailed analytical testing, cell line development, and strict comparability studies to closely replicate the reference biologic’s structure, function, and performance. The process typically spans 6–9 years with costs ranging from $100M to $300M, emphasizing reverse engineering, optimized cell-based production, and clinical validation of similar pharmacokinetics.
- Key players: Amgen, Pfizer, Sandoz, Samsung Bioepis, Biocon, and Celltrion.
Clinical Trials
- Clinical trials for biosimilar antibodies are designed to confirm that they perform as safely and effectively as the original biologic, with no meaningful differences in immune response or outcomes, rather than proving efficacy from scratch.
- Key players: Roche, Novartis, Teva Pharmaceutical Industries, Dr. Reddy’s Laboratories, Lupin, and Mylan.
Regulatory Approvals
- Regulatory approval for biosimilar antibodies follows a streamlined but strict process that relies on the “totality of evidence” approach, ensuring the product closely matches the reference biologic in safety, quality, and effectiveness without meaningful differences.
- Key players: AbbVie, Eli Lilly and Company, Fresenius Kabi, Intas Pharmaceuticals, Zydus Lifesciences, and Alvotech.
Market Growth
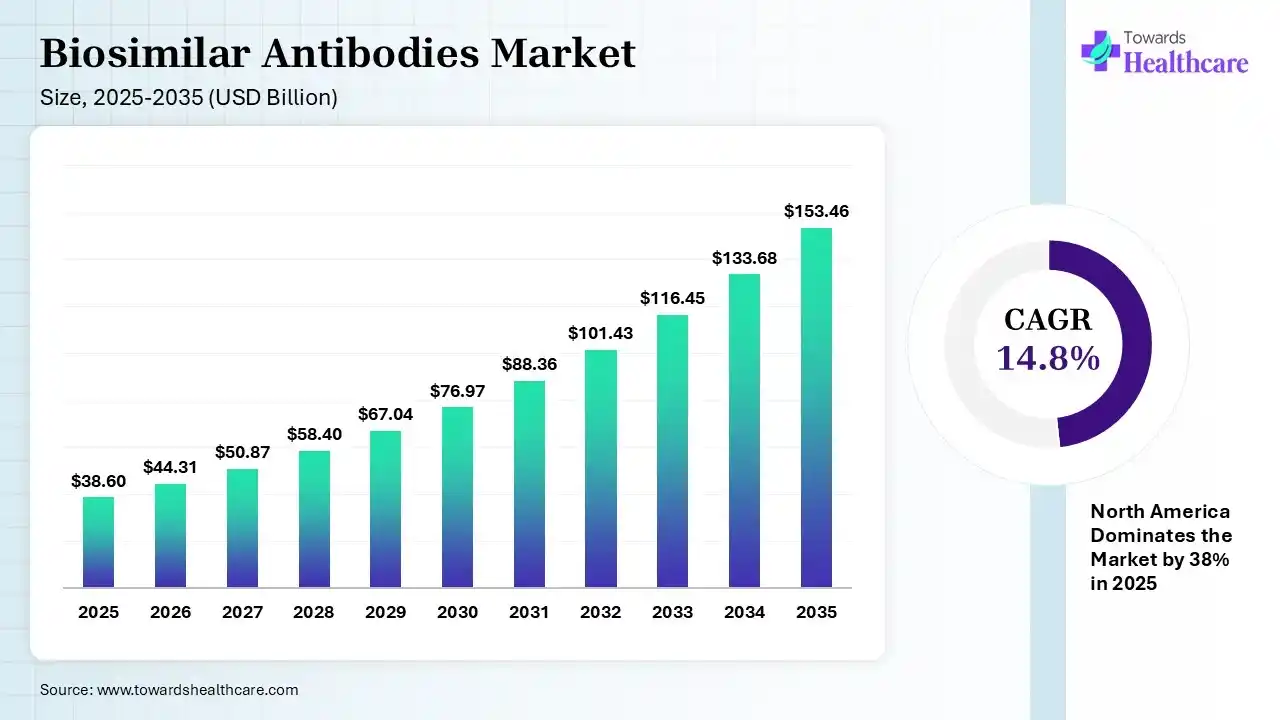
The global biosimilar antibodies market size was estimated at USD 38.6 billion in 2025 and is predicted to increase from USD 44.31 billion in 2026 to approximately USD 153.46 billion by 2035, expanding at a CAGR of 14.8% from 2026 to 2035.
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
