Who are the Blood-Based Biomarker for Alzheimer’s Disease Diagnostics Market Top Vendors and What are Their Offerings?

Take a closer look at the market - Download Your Free Sample
| Companies | Headquarters | Blood-Based Biomarkers for Alzheimer’s Disease Diagnostics |
| Roche Diagnostics | Basel, Switzerland | Elecsys p Tau181 |
| Fujirebio | Tokyo, Japan | Lumipulse G p Tau217/β-Amyloid 1-42 Ratio |
| C2N Diagnostics | St. Louis, U.S. | PrecivityAD2 |
| Quanterix | Billerica, U.S. | LucentAD |
| Quest Diagnostics | Secaucus, U.S. | AD-Detect |
| Labcorp | Burlington, U.S. | Amyloid-Beta 42/40 Ratio Test |
| Eli Lilly & Co. | Indianapolis, U.S. | P-tau217 immunoassay |
| Siemens Healthineers | Erlangen, Germany | Atellica Solution |
| Sysmex Corporation | Kobe, Japan | Amyloid-beta plasma protein marker |
| Abbott Laboratories | Abbott Park, U.S. | ARCHITECT p Tau assays |
SWOT Analysis
Strengths
- The blood-based biomarker for Alzheimer’s disease diagnostics offers a minimally invasive approach, which increases its adoption rates.
- Their affordability also enhances their accessibility, increasing their acceptance rates.
- They also help in early and acute detection of Alzheimer’s disease, driving their adoption rates.
- It is also being used for the monitoring of the disease progression.
Weaknesses
- The lower specificity acts as the major weakness in the blood-based biomarker for Alzheimer’s disease diagnostics market, which limits its use.
- False positive results and standardization issues also reduce their use.
Opportunities
- The growing health awareness is increasing the use of blood-based biomarkers for Alzheimer’s disease diagnostics for the early detection of the disease.
- Growing advancements in personalized medicine are also increasing their use.
- Expanding routine check-ups are also increasing their adoption rates.
- Increasing technological advancements are also increasing the adoption of advanced tools, driving their innovations.
Threats
- Inconsistency due to assay variability leading to the batch failure limits their innovations.
- Stringent quality control and regulatory standards also slow down their innovations.
Market Value Chain Analysis
R&D
- The R&D of the blood-based biomarker for Alzheimer’s disease diagnostics focuses on ultrasensitive p-tau217 and neurofilament light (MfL) assay development.
- Key players: Roche Diagnostics, Quanterix, Eli Lilly & Co.
Clinical Trials and Regulatory Approvals
- The diagnostic accuracy is evaluated in the clinical trials and regulatory approvals of the blood-based biomarker for Alzheimer’s disease diagnostics to promote its use for treatment monitoring, patient enrichment, and target engagement.
- Key players: Roche Diagnostics, Eli Lilly & Co.
Patient Support and Services
- The blood collection networks, financial assistance programs, and online portal are provided in the patient support and services of the blood-based biomarker for Alzheimer’s disease diagnostics.
- Key players: Quest Diagnostics, Labcorp.
Market Trends
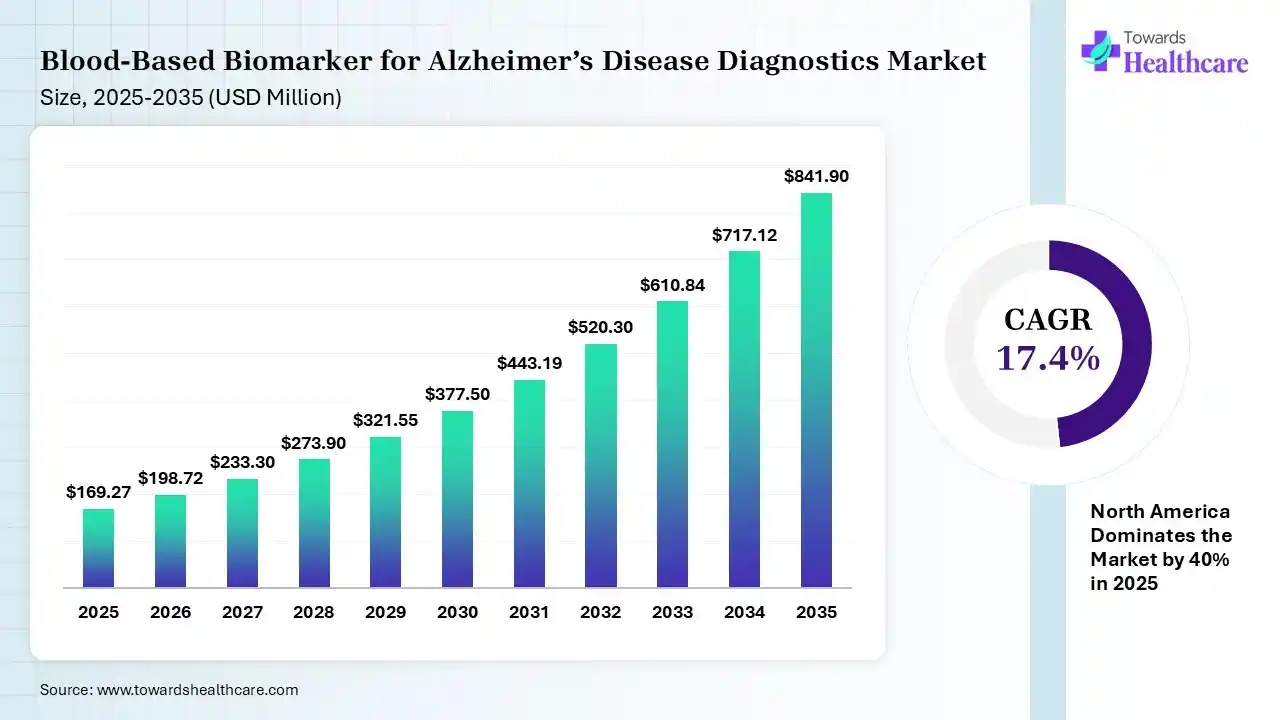
The global blood-based biomarker for Alzheimer’s disease diagnostics market size was estimated at USD 169.27 million in 2025 and is predicted to increase from USD 198.72 million in 2026 to approximately USD 841.9 million by 2035, expanding at a CAGR of 17.4% from 2026 to 2035. The growing incidence of Alzheimer’s disease is increasing the use of blood-based biomarkers. The growing R&D activities, government support, and launch of new products are also enhancing the market growth.
Recent Developments in the Market
- In March 2026, an affordable and non-invasive simple blood test leveraging p-tau and amyloid protein for the early detection of Alzheimer’s disease, titled Mispa i60, was launched by Neuberg Diagnostics.
- In March 2026, the third phase of the Diagnostics Accelerator (DxA) developed by Alzheimer's Drug Discovery Foundation (ADDF) was announced, where it also received an additional $50 million in funding, which will support the development of next-generation biomarkers, accelerating drug development, combination therapies, and precision medicine for Alzheimer's patients.
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
