March 2026

The global blood-based biomarker for Alzheimer’s disease diagnostics market size was estimated at USD 169.27 million in 2025 and is predicted to increase from USD 198.72 million in 2026 to approximately USD 841.9 million by 2035, expanding at a CAGR of 17.4% from 2026 to 2035. The growing incidence of Alzheimer’s disease is increasing the use of blood-based biomarkers. The growing R&D activities, government support, and launch of new products are also enhancing the market growth.
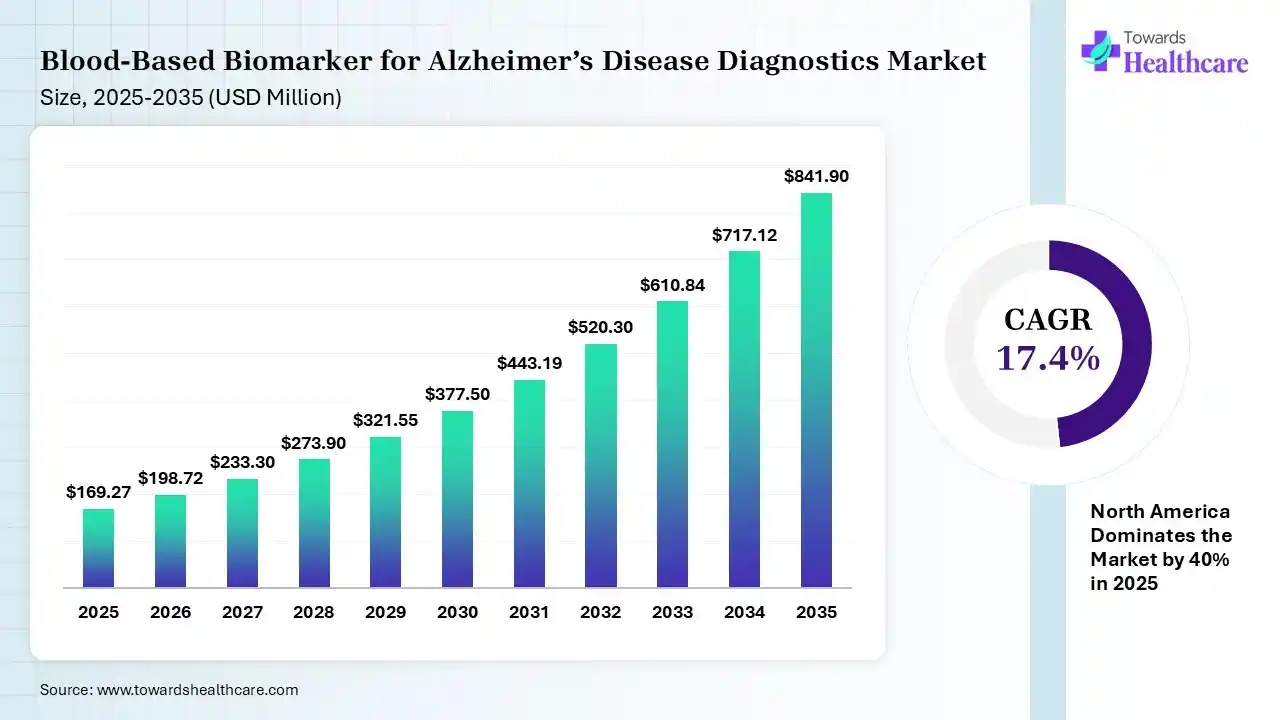
The blood-based biomarker for Alzheimer’s disease diagnostics market is driven by growing demand for non-invasive screening, expanding new therapies, and regulatory breakthroughs. The blood-based biomarker for Alzheimer’s disease diagnostics refers to the molecules measured in blood indicating the presence, risk, and progression of Alzheimer’s disease. They help in early disease detection, disease monitoring, diagnosis confirmation, and support drug development.
The use of AI in the blood-based biomarker for Alzheimer’s disease diagnostics market is increasing as it helps in the detection of biomarker changes, leading to an early detection of Alzheimer's disease. It also helps in predicting disease onset and high-risk individuals, where it also helps in integrating multiple biomarkers, enhancing the diagnostic accuracy. It also offers automated interpretation, disease progression monitoring, drug development, and clinical trials as well.
Expanding Applications
The growing blood-based biomarker for Alzheimer’s disease diagnostics applications are driving their adoption in detection and diagnosis. They are being used for therapeutic monitoring, the development of precision medication, and clinical trial applications.
Growing Approvals
A rise in the blood-based biomarker for Alzheimer’s disease diagnostics approval rates is increasing their adoption across healthcare settings, as well as creating new opportunities for the development of new high-sensitivity, specific, and portable diagnostics.
Technological Innovations
The growing technological advancements are promoting the development of ultra-sensitive assay platforms, multiplex panels, digital tools, and next-generation technologies to enhance the blood-based biomarker for Alzheimer’s disease diagnostics applications.
| Table | Scope |
| Market Size in 2026 | USD 198.72 Million |
| Projected Market Size in 2035 | USD 841.9 Million |
| CAGR (2026 - 2035) | 17.4% |
| Leading Region | North America by 40% |
| Historical Data | 2020 - 2023 |
| Base Year | 2025 |
| Forecast Period | 2026 - 2035 |
| Measurable Values | USD Millions/Units/Volume |
| Market Segmentation | By Biomarker Type, By Technology Platform, By Application, By End User, By Test Type, By Sample Type, By Distribution Channel, By Region |
| Top Key Players | Roche Diagnostics, Fujirebio, C2N Diagnostics, Quanterix , Quest Diagnostics, LabCorp, Eli Lilly & Co., Siemens Healthineers, Sysmex Corporation, Abbott Laboratories |
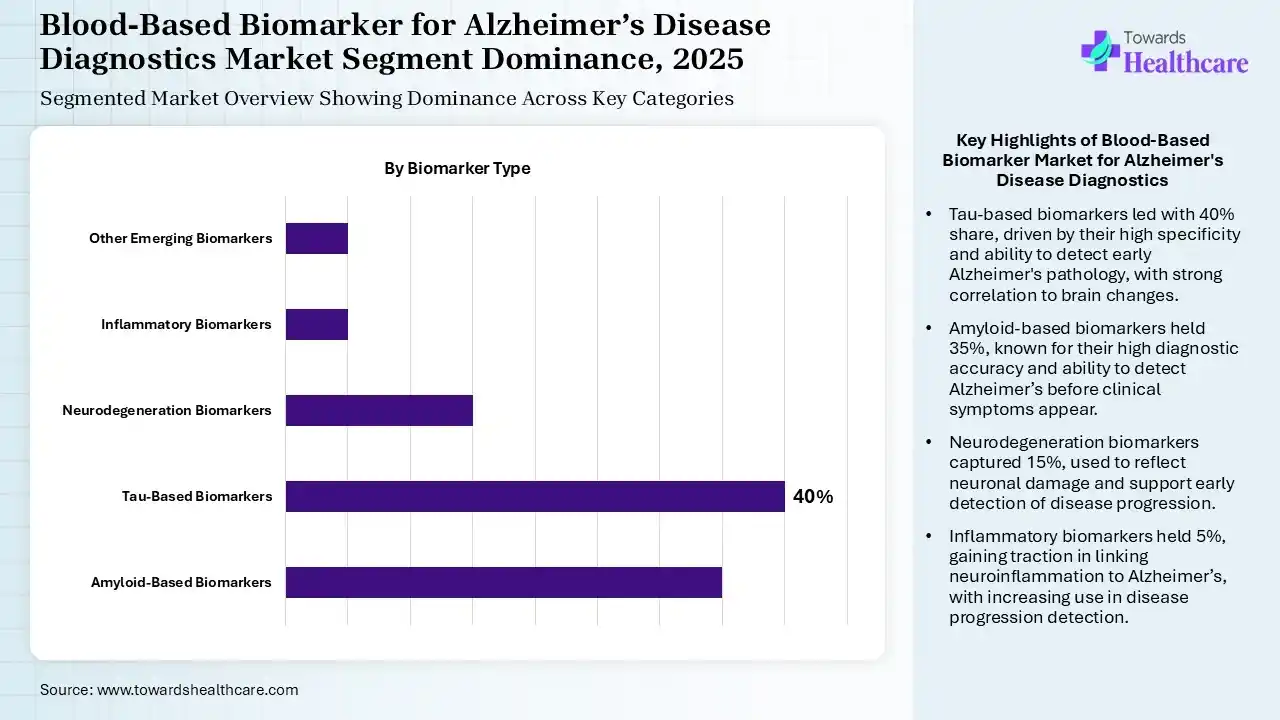
The Tau-Based Biomarkers Segment Dominated the Market in 2025
| Segment | Share 2025 (%) |
| Amyloid-Based Biomarkers | 35% |
| Tau-Based Biomarkers | 40% |
| Neurodegeneration Biomarkers | 15% |
| Inflammatory Biomarkers | 5% |
| Other Emerging Biomarkers | 5% |
The tau-based biomarkers segment led the blood-based biomarker for Alzheimer’s disease diagnostics market with 40% share in 2025 and is expected to grow with the fastest CAGR during the forecast period, due to a growth in the early Alzheimer’s disease pathology detection. Their high specificity also increased their use. The strong correlation between brain changes and enhanced diagnostic accuracy also increased their use.
The amyloid-based biomarkers segment held the second-largest share of 35% in 2025, driven by their high diagnostic accuracy. They also help in the early detection of Alzheimer’s disease before the onset of clinical symptoms. The growing technological advancements are also increasing their innovation and use as a screening tool.
The neurodegeneration biomarkers segment held 15% share of the blood-based biomarker for Alzheimer’s disease diagnostics market in 2025, due to their ability to reflect neuronal damage progression. This helps in the early detection of neurodegeneration and disease progression. Growing brain disorders are also increasing their demand.
The inflammatory biomarkers segment held 5% share of the market in 2025, driven by their growing application in linking the neuroinflammation to Alzheimer’s disease. The growing advancements in the anti-inflammation and immunomodulatory treatment are also increasing their use. It is also being used in the detection of disease progression, where its growing integration with other biomarkers is increasing its innovations.
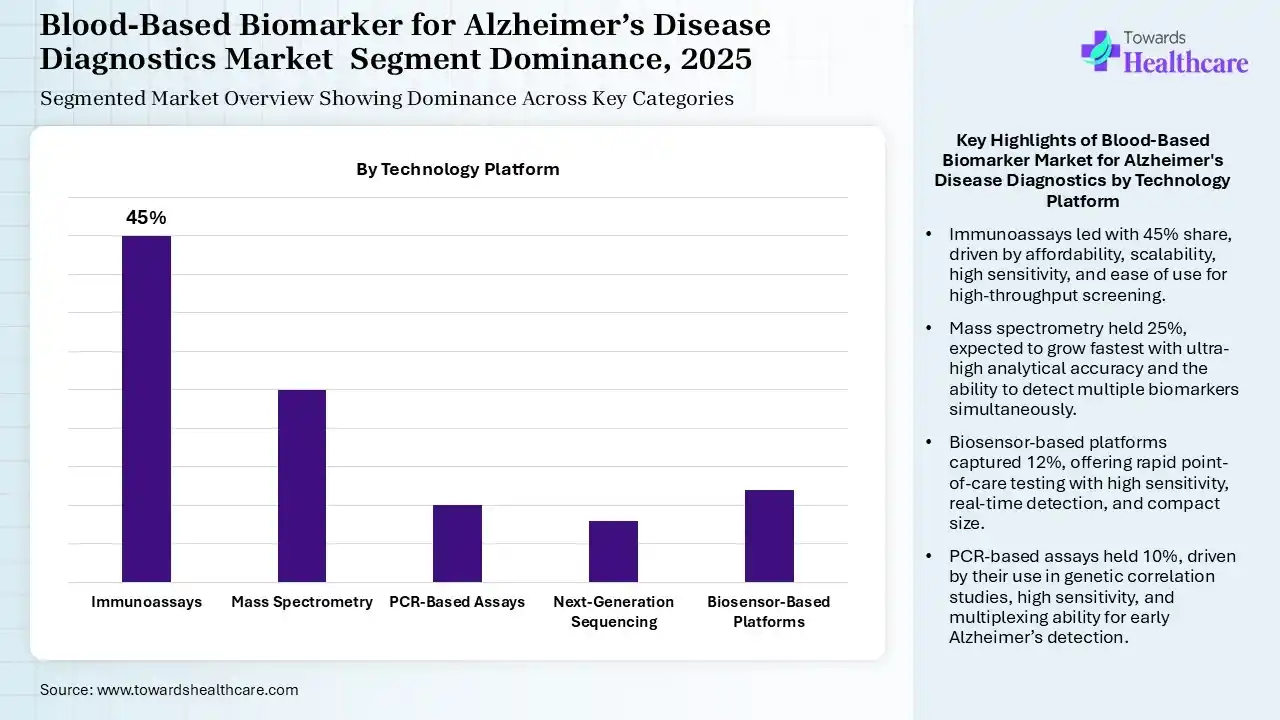
The Immunoassays Segment Dominated the Market in 2025
| Segment | Share 2025 (%) |
| Immunoassays | 45% |
| Mass Spectrometry | 25% |
| PCR-Based Assays | 10% |
| Next-Generation Sequencing | 8% |
| Biosensor-Based Platforms | 12% |
The immunoassays segment held the dominating share of 45% of the blood-based biomarker for Alzheimer’s disease diagnostics market in 2025, driven by their affordable scalability. Their high sensitivity and specificity also increased their use. Additionally, their easy use, high-throughput screening, and widespread availability also increased their use.
The mass spectrometry segment held the second-largest share of 25% of the market in 2025 and is expected to grow at the fastest CAGR during the forecast period, due to its high sensitivity detection. Their ultra-high analytical accuracy is also increasing their adoption rates. Additionally, the simultaneous detection of multiple biomarkers is also increasing their demand.
The biosensor-based platforms segment held 12% share of the blood-based biomarker for Alzheimer’s disease diagnostics market in 2025, driven by their rapid point-of-care testing. Their high sensitivity and real-time detection also increased their use. Furthermore, their compact sizes and multiplexing potential are also increasing their adoption rates.
The PCR-based assays segment held 10% share of the market in 2025, due to their growing use in genetic correlation studies. Their high sensitivity and security are also increasing their use in the early detection of Alzheimer’s disease. The multiplexing ability and requirement of minimal sample volume are also increasing their demand.
The Early Detection/Screening Segment Dominated the Market in 2025
| Segment | Share 2025 (%) |
| Early Detection / Screening | 30% |
| Diagnosis Confirmation | 25% |
| Disease Monitoring | 15% |
| Drug Development & Clinical Trials | 20% |
| Risk Assessment / Predictive Testing | 10% |
The early detection/screening segment registered its dominance over the global blood-based biomarker for Alzheimer’s disease diagnostics market with a share of 30% in 2025 and is expected to grow with the fastest CAGR during the forecast period, due to growth in the demand for preventive healthcare. The growth in Alzheimer’s cases also increased the use of blood-based biomarker diagnostics. Additionally, their non-invasive approach and affordability also increased their use.
The diagnosis confirmation segment held the second-largest share of 25% of the market in 2025, driven by its increasing use in clinical decision-making. Their high accuracy and rapid results are also increasing their adoption rates. Moreover, their growing integration with multi-modal diagnostics is also increasing their demand.
The drug development & clinical trials segment held 20% share of the blood-based biomarker for Alzheimer’s disease diagnostics market in 2025, as the blood-based biomarker for Alzheimer’s disease diagnostics helps in accelerating the biomarker-based clinical trials. It also helps in the early patient selection and recruitment. They also help in the development of precision medicines.
The disease monitoring segment held 15% share of the market in 2025, driven by the ability to track the disease progression. The blood-based biomarker for Alzheimer’s disease diagnostics also helps in the detection of treatment failure and support disease modifying therapies. It also helps in adjusting the drug dose, depending on the patient's response.
The Hospitals Segment Dominated the Market in 2025
| Segment | Share 2025 (%) |
| Hospitals | 35% |
| Diagnostic Laboratories | 30% |
| Research Institutes | 15% |
| Pharmaceutical & Biotechnology Companies | 12% |
| Point-of-Care Settings | 8% |
The hospitals segment held the largest share of 35% of the blood-based biomarker for Alzheimer’s disease diagnostics market in 2025, due to growth in the patient volume. This increased the clinical application of the blood-based biomarker for Alzheimer’s disease diagnostics. The presence of well-equipped infrastructure and skilled professionals also increased patient trust, which contributed to their increased use.
The diagnostic laboratories segment held the second-largest share of 30% of the market in 2025 and is expected to grow at the fastest CAGR during the forecast period, driven by the expanding testing infrastructure. Moreover, the adoption of high technologies is also increasing the use of new blood-based biomarkers for Alzheimer’s disease diagnostics. Their affordable operation are also attracting the patients.
The research institutes segment held 15% share of the blood-based biomarker for Alzheimer’s disease diagnostics market in 2025, due to growing biomarker innovations. The advancements in multiplex studies and focus on early disease detection technologies are also increasing the use of blood-based biomarkers for Alzheimer’s disease diagnostics. Additionally, increasing funding is also accelerating their innovations.
The pharmaceutical & biotechnology companies segment held 12% share of the market in 2025, due to growing clinical trials and drug development. The growing advancements in precision medicine and companion diagnostics are also increasing the use of blood-based biomarkers for Alzheimer’s disease diagnostics. The growing partnerships are also increasing their innovations and use.
The point-of-care setting held 8% of the market share in 2025 and is estimated to be the fastest-growing during the forecast period because it enables rapid decentralized testing, leading to quicker and better results.
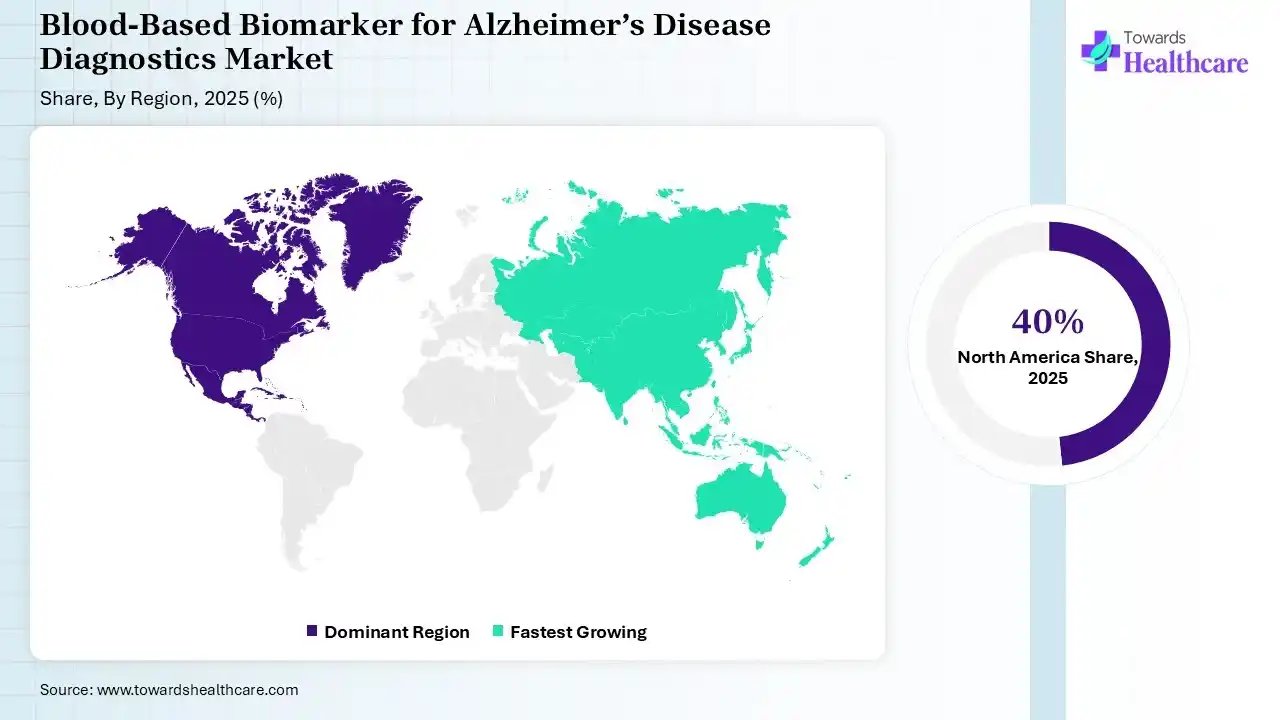
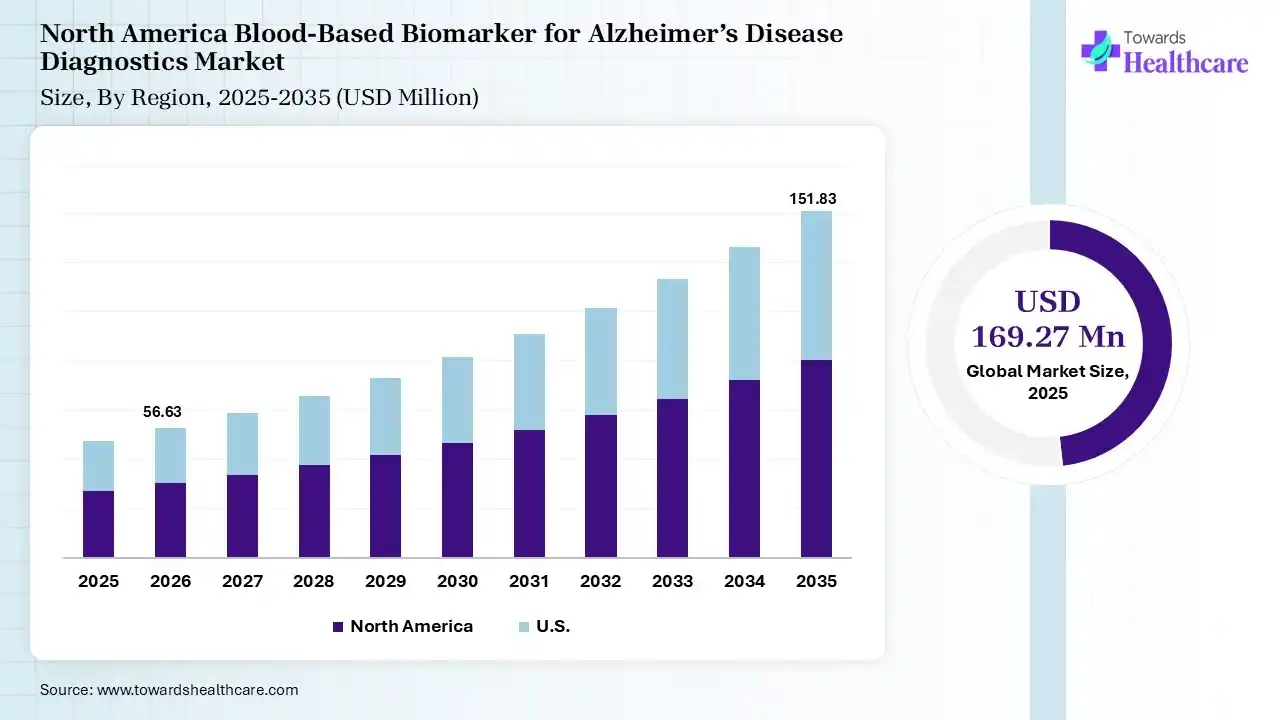
North America dominated the blood-based biomarker for Alzheimer’s disease diagnostics market with 40% in 2025, due to growth in the adoption of advanced diagnostics, where the rise in Alzheimer's disease also increased the use of blood-based biomarkers for Alzheimer’s disease diagnostics. The growth in healthcare spending and R&D activities also increased their innovation, which contributed to the market growth.
U.S. Market Trends
The growing incidence of Alzheimer’s disease in the U.S. is increasing the demand for blood-based biomarkers for Alzheimer’s disease diagnostics. The presence of advanced healthcare infrastructure is also increasing their adoption rates. Moreover, the growing clinical trials, healthcare investments, and technological advancements are also increasing their innovations.
Asia Pacific held a share of 20% of the blood-based biomarker for Alzheimer’s disease diagnostics market in 2025 and is expected to grow at the fastest CAGR during the forecast period, due to growth in the geriatric population. The rise in disease awareness is also increasing the adoption of blood-based diagnostics. Expanding healthcare is also increasing its use, enhancing the market growth.
India Market Trends
The rapid expansion of healthcare in India is increasing the adoption of the blood-based biomarker for Alzheimer’s disease diagnostics in India. A growing geriatric population and increasing government initiatives are also increasing their use. Additionally, growing R&D activities and health awareness are also increasing their innovations and adoption rates.
R&D
Patient Support and Services

| Companies | Headquarters | Blood-Based Biomarkers for Alzheimer’s Disease Diagnostics |
| Roche Diagnostics | Basel, Switzerland | Elecsys p Tau181 |
| Fujirebio | Tokyo, Japan | Lumipulse G p Tau217/β-Amyloid 1-42 Ratio |
| C2N Diagnostics | St. Louis, U.S. | PrecivityAD2 |
| Quanterix | Billerica, U.S. | LucentAD |
| Quest Diagnostics | Secaucus, U.S. | AD-Detect |
| Labcorp | Burlington, U.S. | Amyloid-Beta 42/40 Ratio Test |
| Eli Lilly & Co. | Indianapolis, U.S. | P-tau217 immunoassay |
| Siemens Healthineers | Erlangen, Germany | Atellica Solution |
| Sysmex Corporation | Kobe, Japan | Amyloid-beta plasma protein marker |
| Abbott Laboratories | Abbott Park, U.S. | ARCHITECT p Tau assays |
Strengths
Weaknesses
Opportunities
Threats
By Biomarker Type
By Technology Platform
By Application
By End User
By Test Type
By Sample Type
By Distribution Channel
By Region
March 2026
March 2026
March 2026
March 2026