Latest Updates of Key Players in the Clinical Endpoint Adjudication (CEA) Market

ICON plc
Headquarter: Ireland
Latest Update: In January 2026, ICON plc announced a significant expansion of oncology research capabilities within its Accellacare Site Network through the opening of the Brian Moran Cancer Institute at Duly Health and Care in Illinois.
Medidata
Headquarter: United States
Latest Update: Medidata Showcases AI-driven Innovations Reshaping Clinical Research and Development at NEXT Shanghai 2025.
Clario
Headquarter: United States
Latest Update: Clario generates the richest clinical evidence by fusing our deep scientific expertise and global scale into the broadest endpoint technology platform.
WCG
Headquarter: United States
Latest Update: It enhances the quality and efficiency of clinical trials, helping biopharmaceutical companies, CROs, and institutions facilitate the development of new.
Advarra
Headquarter: United States
Latest Update: Advarra's Site Technology Optimization & Scaling services provide strategic guidance and expert resources to best align technology with site workflows.
Ethical GmbH
Headquarter: Switzerland
Latest Update: Ethical's Cloud-based clinical endpoints adjudication management software, eAdjudication, helps drug manufacturers simplify assessment processes.
Value Chain Analysis
R&D:
- Clinical endpoint adjudication (CEA) R&D technology involves systematically assessing potential clinical events using a self-governing committee to ensure data precision and government compliance.
- Key Players: ICON plc. and IQVIA
Manufacturing Processes:
- It involves core operational phases confirm that multifaceted healthcare events are evaluated consistently and free of bias.
- Key Players: Clario and Medidata Solutions
Patient Services:
- Patient services in clinical endpoint adjudication are intended to ensure the safety of contributors, preserve the integrity of the research data, and offer an independent, expert assessment of healthcare events.
- Key Players: Fortrea and Syneos Health
Market Growth
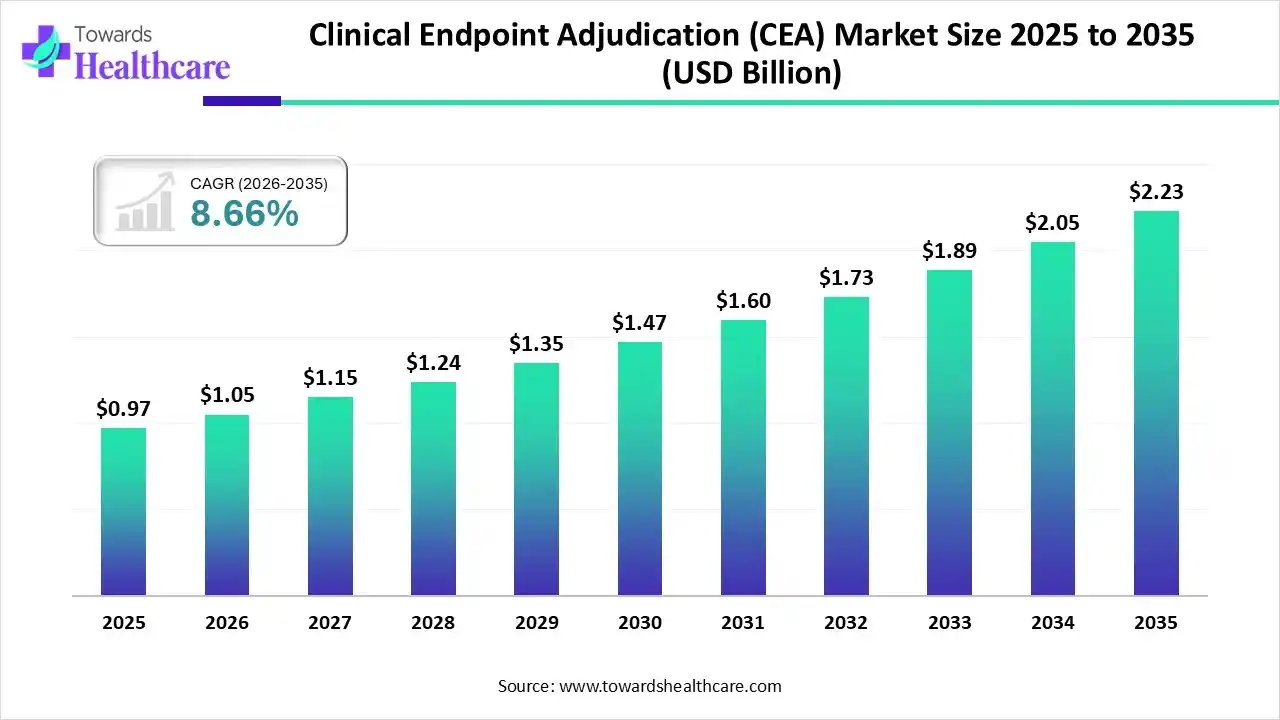
The global clinical endpoint adjudication (CEA) market size was estimated at USD 0.97 billion in 2025 and is predicted to increase from USD 1.05 billion in 2026 to approximately USD 2.23 billion by 2035, expanding at a CAGR of 8.66% from 2026 to 2035.
Recent Developments in the Clinical Endpoint Adjudication (CEA) Market
- In November 2025, BioDlink, a leading biologics CDMO, composed with Lepu Biopharma, announced the effective market launch of MEIYOUHENG, the world's first EGFR-targeting antibody-drug conjugate (ADC). The launch shows the first ADC fully manufactured by a CDMO to reach the market in China, underlining a new level of maturity for the region's biopharmaceutical ecosystem.
- In January 2026, BioCryst Pharmaceuticals, Inc. announced that it had completed its acquisition of Astria Therapeutics, Inc., as initially announced. The transaction strengthens its position as a frontrunner in genetic angioedema (HAE) and improves the organization's long-term growth trajectory.
- In July 2025, ESTEVE acquired Regis Technologies, a United States-based Contract Development and Manufacturing Organization (CDMO), headquartered in Chicago.
Become a valued research partner with us, please feel free to contact us at sales@towardshealthcare.com
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
