Latest Updates of Key Players in the Clinical Trial Drug Sourcing Market

Company Updates
| Company | Headquarters | Latest Update |
| Thermo Fisher Scientific Inc. | Denmark | Thermo Fisher and OpenAI aim to accelerate clinical trials and allow the delivery of novel medicines to patients sooner. |
| IQVIA Holdings Inc. | United States | In September 2025, IQVIA announced the launch of its Clinical Trial Financial Suite (CTFS), an AI-driven platform orchestrating all financial aspects of clinical trials. |
| Catalent, Inc | Florida | In September 2025, Science 37 and Catalent announced a strategic partnership redefining how investigational medicinal products (IMPs) are delivered directly to patients’ homes for clinical research. |
| Almac Group | Ireland | In November 2025, Almac Group announced a multi-million-pound investment to expand its facility in Singapore, as the company marks ten years of operations in the country. |
| Parexel International Corporation | North Carolina | Parexel remains the only CRO to be recognised with both an ACE Award and a Catalyst Award, highlighting its ongoing commitment to inclusivity, both in the organisation and in the way it serves patients. |
| ICON plc | Ireland | In January 2026, ICON plc announced a significant expansion of oncology research abilities in its Accellacare Site Network through the opening of the Brian Moran Cancer Institute at Duly Health and Care in Illinois. |
Value Chain Analysis
R&D:
- R&D processes in clinical trial drug sourcing involve strategic planning to procure, manage, and distribute Investigational Medicinal Products (IMPs) while confirming governing compliance.
- Key Players: PCI Pharma Services
Manufacturing Processes:
- Major manufacturing processes involved in clinical trial drug sourcing are specialized, massively regulated, and intended for flexibility, speed, and precision rather than more volume.
- Key Players: Catalent, Inc., and Clinigen Limited
Patient Services:
- Key services involve patient-centric logistics, targeted, patient-driven labeling, and subsidiary patient engagement via digital devices for compliance and communication.
- Key Players: Thermo Fisher Scientific and Almac Group
Market Growth Story
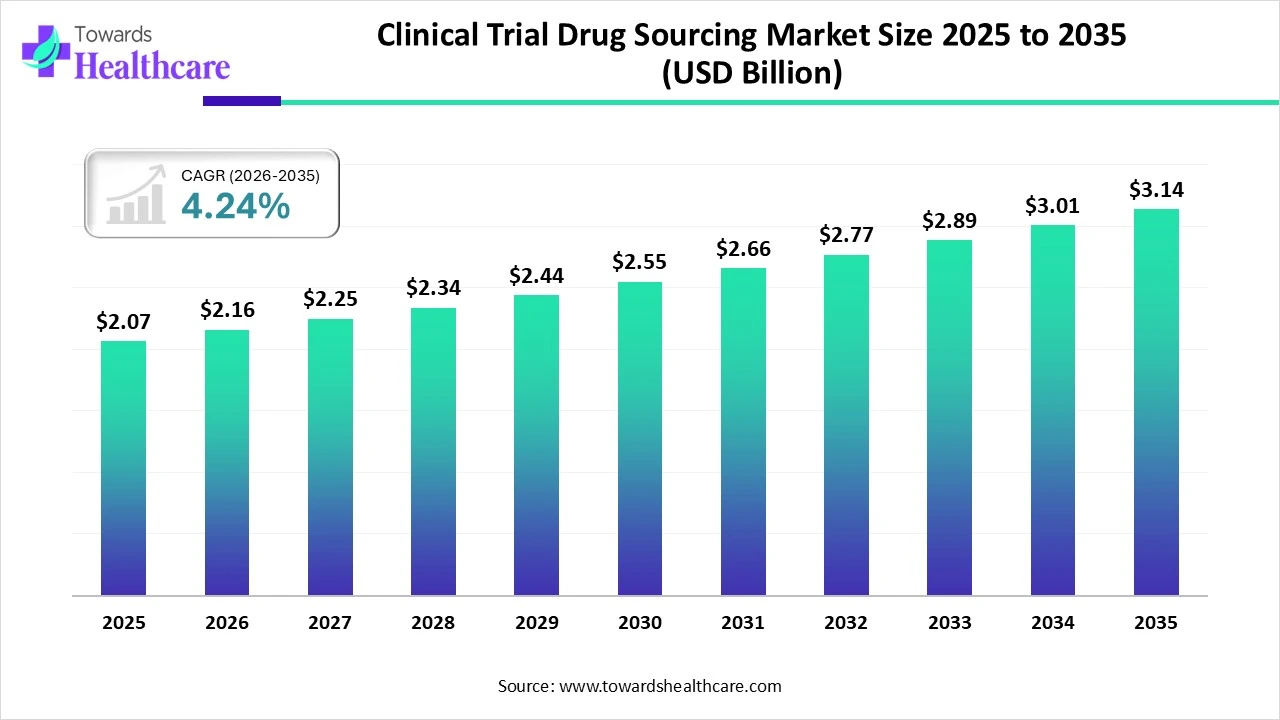
The global clinical trial drug sourcing market size was estimated at USD 2.07 billion in 2025 and is predicted to increase from USD 2.16 billion in 2026 to approximately USD 3.14 billion by 2035, expanding at a CAGR of 4.24% from 2026 to 2035.
Recent Developments in the Clinical Trial Drug Sourcing Market
- In October 2025, Takeda announced that it had entered into a license and collaboration agreement with Innovent Biologics for the development, manufacturing, and commercialization of two late-stage oncology medicines, IBI363 and IBI343, worldwide outside of Greater China.
- In September 2025, Pfizer Inc. and BioNTech SE announced positive topline results from a continuing Phase 3 clinical trial cohort evaluating the safety, tolerability, and immunogenicity of a 30-µg dose of the LP.8.1-acceptables monovalent COMIRNATY 2025-2026 Formula in adults aged 65 and older and in adults aged 18 to 64 with at least one underlying challenge condition for severe COVID-19.
One place. One view. Total control of your Clinical Trial Drug Sourcing data - Access the Dashboard
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
