Who are the Clinical Trials Management System (CTMS) Market Top Vendors and What are Their Offerings?

Now it’s effortless to manage all your Clinical Trials Management System (CTMS) Market data in one powerful place; Access the Dashboard
| Companies | Headquarters | Clinical Trials Management Systems |
| Veeva Systems | Pleasanton, U.S. | Veeva Vault CTMS |
| Medidata (Dassault Systèmes) | New York, U.S. | Medidata Rave CTMS |
| Oracle Life Sciences | Austin, U.S. | Oracle Siebel CTMS or ClearTrial |
| IQVIA | Durham, U.S. | IQVIA Technologies CTMS |
| Advarra | Columbia, U.S. | Clinical Conductor CTMS |
| SimpleTrials | San Luis, U.S. | SimpleTrials CTMS |
| MasterControl | Salt Lake City, U.S. | MasterControl Clinical Excellence |
| Clinion | Hyderabad, India | Clinion CTMS |
| Ennov | Paris, France | Ennov Clinical (CTMS) |
| RealTime Software Solutions | San Antonio, U.S. | RealTime-CTMS |
Market Growth
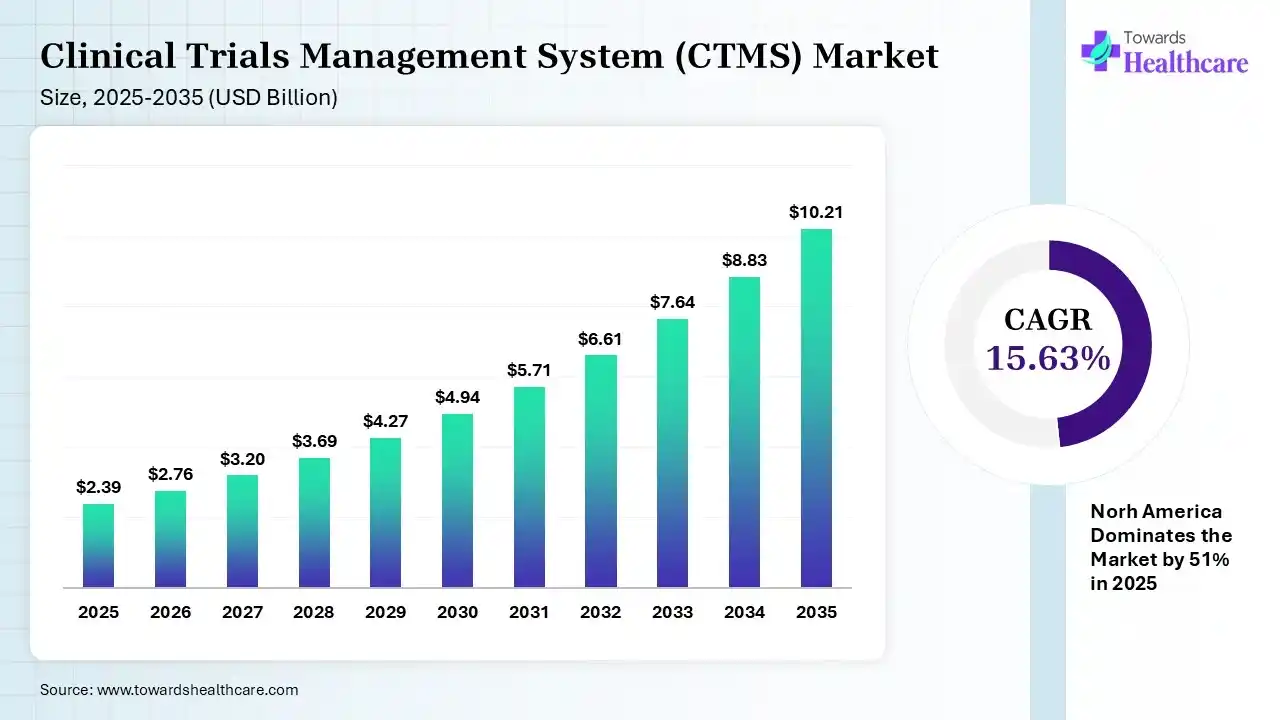
The global clinical trials management system (CTMS) market size was estimated at USD 2.39 billion in 2025 and is predicted to increase from USD 2.76 billion in 2026 to approximately USD 10.21 billion by 2035, expanding at a CAGR of 15.63% from 2026 to 2035.
What are the Recent Developments in the Market?
- In September 2025, a collaboration between Arkivum and PHARMASEAL, consisting of Clinical Trial Management System (CTMS) and Electronic Trial Master File (eTMF), where they will integrate the unified trial management platform of PHARMASEAL with GxP-validated digital archiving and preservation technology of Arkivum to offer an end-to-end trial technology solution.
- In April 2025, Veeva SiteVault CTMS, integrating Site Vault eISF and SiteVault eConsent, was launched by Veeva Systems as a single system for comprehensive management of clinical trials.
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
