May 2026

The global clinical trials management system (CTMS) market size was estimated at USD 2.39 billion in 2025 and is predicted to increase from USD 2.76 billion in 2026 to approximately USD 10.21 billion by 2035, expanding at a CAGR of 15.63% from 2026 to 2035.
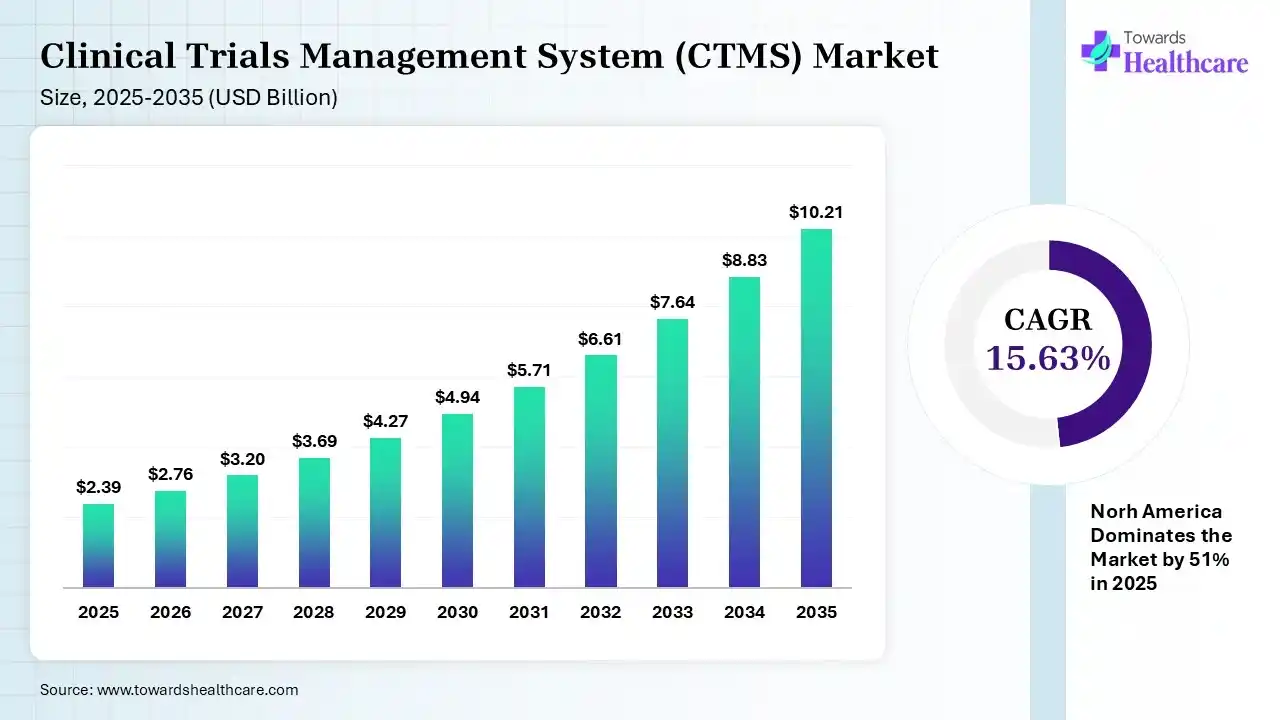
The growing R&D activities and outsourcing trends are increasing the adoption of CTMS for efficient trial management. Growing AI integration, hybrid trials, technological advancements, and new platform launches are also enhancing the market growth.
The clinical trials management system (CTMS) market is driven by the combination of increasing digital solutions, clinical trial complexities, and the shift towards decentralized models. The clinical trials management system (CTMS) refers to the software platform utilized for the management, planning, and tracking of clinical trials, ensuring the safety, speed, and efficiency of the trials. These systems help in patient recruitment, scheduling, site management, and offer regulatory compliance.
AI plays an important role in the clinical trials management system (CTMS) market, where it analyzes large data sets and helps in the recruitment of suitable patients. It also helps in the monitoring and planning of the clinical trials, reducing the workload. It also helps in maintaining data integrity by identifying inconsistent data, as well as offering automation and data-driven insights.
Expanding Decentralized Trials
The growing shift towards decentralized or hybrid trials is increasing the adoption of the CTMS for the management of remote trials, improving patient recruitment and retention, and real-time data management.
Increasing Focus on Data Security
The proprietary data and sensitive patient information are collected during the clinical trials, which increases the risk of cyberattacks driving from the adoption of the CTMS to offer enhanced data security and privacy.
Growing Innovations
The growing technological advancements are increasing the development of new cloud-based, SaaS, and mobile CTMS platforms to offer more flexible and user-friendly approaches for various organizations.
| Table | Scope |
| Market Size in 2026 | USD 2.76 Billion |
| Projected Market Size in 2035 | USD 10.21 Billion |
| CAGR (2026 - 2035) | 15.63% |
| Leading Region | North America by 39% |
| Historical Data | 2020 - 2023 |
| Base Year | 2025 |
| Forecast Period | 2026 - 2035 |
| Measurable Values | USD Millions/Units/Volume |
| Market Segmentation | By Solution Type, By Component, By Delivery Mode, By End-use, By Region |
| Top Key Players | Veeva Systems, Medidata (Dassault Systèmes), Oracle Life Sciences, IQVIA, Advarra, SimpleTrials, MasterControl, Clinion, Ennov, RealTime Software Solutions |
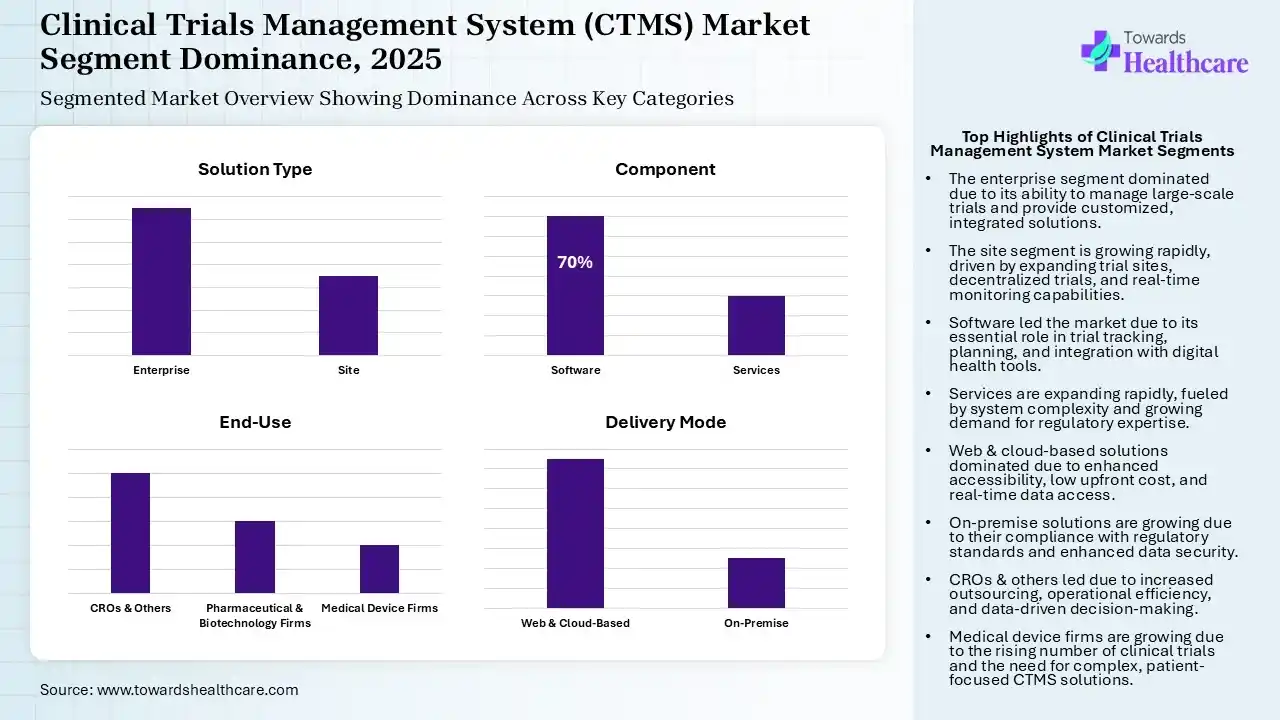
Why Did the Enterprise Segment Dominate in the Market in 2025?
| Segment | Share 2025 (%) |
| Enterprise | 65% |
| Site | 35% |
Explanation
The enterprise segment held the largest revenue share of the clinical trials management system (CTMS) market by 65% in 2025, due to its ability to manage large-scale trials. This increased their use across large pharmaceutical companies and CROs, where their customized solutions and end-to-end integration also increased their adoption rates. Their enhanced data security and regulatory compliance also increased their use.
Site
The site segment is expected to grow at a notable CAGR during the predicted time, due to expanding clinical trial sites and their complexities. The growing decentralized and hybrid trials are also increasing their demand. Additionally, their real-time monitoring, enhanced patient recruitment, and regulatory compliance are also increasing their use to reduce the administrative burden.
How Software Segment Dominated the Market in 2025?
| Segment | Share 2025 (%) |
| Software | 70% |
| Services | 30% |
Explanation
The software segment contributed the biggest revenue share of the clinical trials management system (CTMS) market by 70% in 2025, as it was essential for trial tracking, management, and planning. They also offered enhanced scalability and integration with other digital health tools and analytical platforms. Moreover, the shift towards digital solutions also increased their adoption rates as well as innovations.
Services
The services segment is expected to expand rapidly during the predicted time, due to growing system complexities. The growing outsourcing trend and increasing demand for regulatory expertise are also increasing the use of CTMS. Additionally, increasing cloud-based solutions and focusing on operational efficiency are also increasing their adoption rates.
Which Delivery Mode Type Segment Held the Dominating Share of the Market in 2025?
| Segment | Share 2025 (%) |
| Web & Cloud-Based | 75% |
| On-Premise | 25% |
Explanation
The web & cloud-based segment held the dominating share of the clinical trials management system (CTMS) market by 75% in 2025 and is expected to show the fastest growth rate during the foreseeable future, due to its enhanced accessibility and low upfront cost. Their real-time data access and faster deployment also increased their acceptance rates.
On Premise
The on-premises segment is expected to grow at a considerable CAGR during the upcoming year, due to compliance with regulatory standards and enhanced data security. They also offer seamless integration and consistent performance, which is increasing their demand. Additionally, their regular updates are also increasing their use across larger organizations.
What Made CROs & Others the Dominant Segment in the Market in 2025?
| Segment | Share 2025 (%) |
| CROs & Others | 50% |
| Pharmaceutical & Biotechnology Firms | 30% |
| Medical Device Firms | 20% |
Explanation
The CROs & others segment accounted for the highest revenue share of the clinical trials management system (CTMS) market by 50% in 2025 and is expected to grow with the highest CAGR during the foreseeable future, due to increased outsourcing trends, which enhanced the use of CTMS. These systems were also used to enhance operational efficiency and offer data-driven decisions.
Medical Device Firms
The medical device firms segment is expected to show lucrative growth during the upcoming year, due to the growing number of medical device clinical trials. Their stringent regulatory requirements, complex protocols, and focus on patient safety are also increasing the use of CTMS. Moreover, to handle multiple clients and provide seamless integration with other digital tools is also increasing their demand.
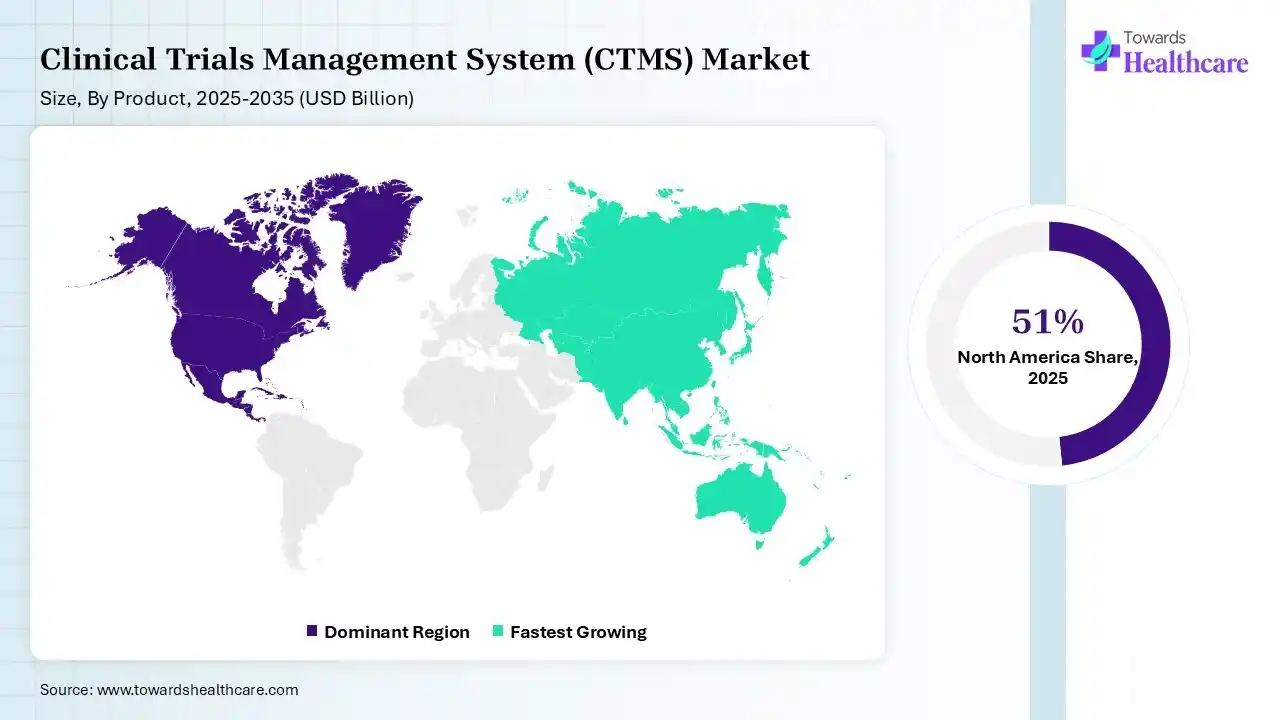
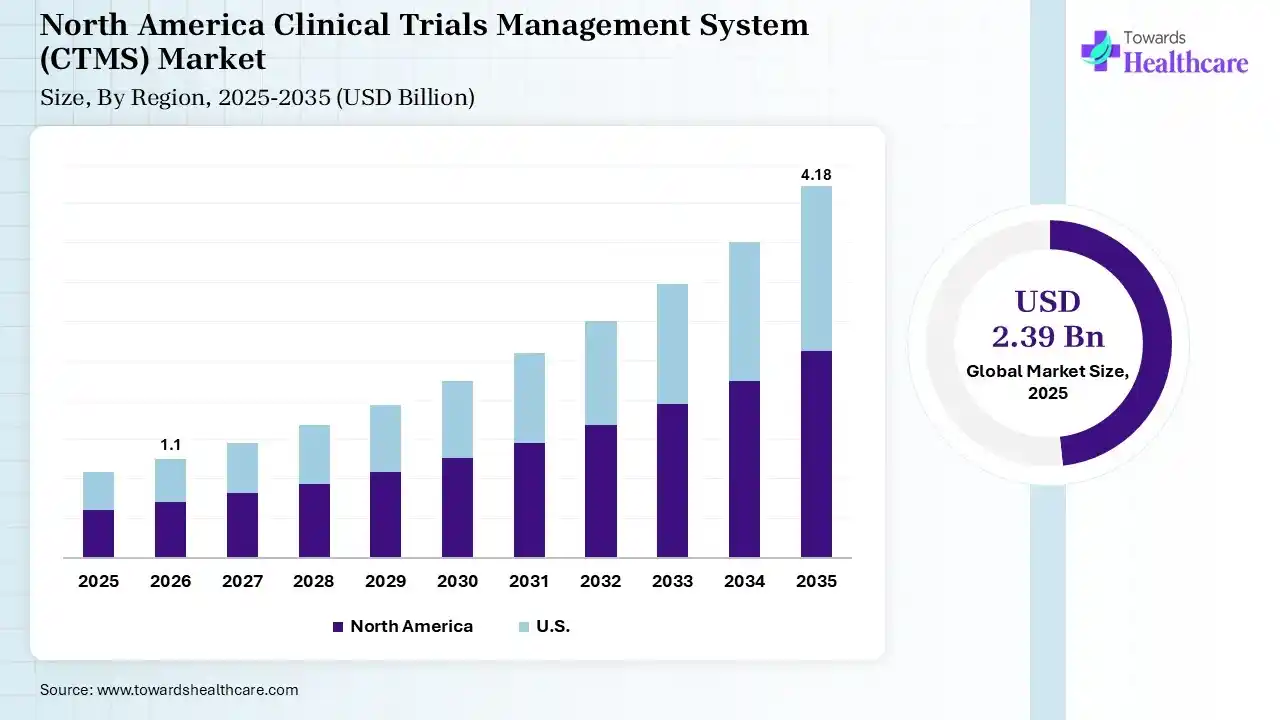
North America dominated the clinical trials management system (CTMS) market by 51% in 2025, due to the presence of robust industries, which increased the number of clinical trials, driving the adoption of CTMS. The well-established healthcare IT infrastructure and regulatory agencies also encouraged their use, where the growth in healthcare expenditure and technological advancements also contributed to the market growth.
U.S. Market Trends
The growing clinical trials across the U.S. are increasing the use of CTMS for their seamless organization and management. Their easy integration is also increasing their use across various organizations. At the same time, the advanced healthcare IT infrastructure is also enhancing its features, launching new CTMS platforms.
Asia Pacific is expected to host the fastest-growing clinical trials management system (CTMS) market during the forecast period, due to growing clinical trials driven by increasing R&D activities across the pharma and biotech industries. The expanding healthcare sector and CRO ecosystem are also increasing the demand for CTMS, where their growing advancements are also enhancing the market growth.
China Market Trends
The expanding industries in China are increasing the R&D activity and their clinical trials, increasing the demand for CTMS. The growing healthcare investment, government support, and expanding CRO networks are also increasing their adoption rates. Additionally, increasing technological advancements are also promoting their CTMS advancements.
Europe is expected to grow significantly in the clinical trials management system (CTMS) market during the forecast period, due to the presence of advanced healthcare infrastructures and stringent regulations, which are increasing the adoption of CTMS. Additionally, growing clinical trials, technological innovations, and government funding are also increasing their use, promoting the market growth.
UK Market Trends
The presence of well-developed pharmaceutical, medical devices, and biotechnology companies in the UK is increasing the adoption of CTMS for faster and seamless clinical trials. Furthermore, the growing outsourcing trends are also increasing their use, where advanced healthcare IT infrastructure is also increasing the development of new CTMS.

| Companies | Headquarters | Clinical Trials Management Systems |
| Veeva Systems | Pleasanton, U.S. | Veeva Vault CTMS |
| Medidata (Dassault Systèmes) | New York, U.S. | Medidata Rave CTMS |
| Oracle Life Sciences | Austin, U.S. | Oracle Siebel CTMS or ClearTrial |
| IQVIA | Durham, U.S. | IQVIA Technologies CTMS |
| Advarra | Columbia, U.S. | Clinical Conductor CTMS |
| SimpleTrials | San Luis, U.S. | SimpleTrials CTMS |
| MasterControl | Salt Lake City, U.S. | MasterControl Clinical Excellence |
| Clinion | Hyderabad, India | Clinion CTMS |
| Ennov | Paris, France | Ennov Clinical (CTMS) |
| RealTime Software Solutions | San Antonio, U.S. | RealTime-CTMS |
Strengths
Weaknesses
Opportunities
Threats
By Solution Type
By Component
By Delivery Mode
By End-use
By Region
May 2026
April 2026
April 2026
April 2026