Key Players' Offerings in the Drug Product Development Service Market

Thermo Fisher Scientific (Patheon)
Description: Its offerings comprise formulation development, analytical testing, sterile injectables, oral solid doses, softgels, & clinical trial logistics.
Catalent, Inc.
Description: This mainly covers small molecules, biologics (sterile injectables), & cell/gene therapies.
Lonza Group AG
Description: Its services include specialized, sterile parenteral dosage forms, formulation improvement, analytical development, stability studies, & container closure integrity testing.
Recipharm AB
Description: This offers diverse services from pre-formulation and API development to clinical trials, scale-up, and commercial manufacturing.
Piramal Pharma Solutions
Description: A firm specializes in the production of oral solids, liquids, creams, ointments, and sterile injectables.
Evotec SE
Description: It unveiled "Data-driven R&D Autobahn to Cures" to raise drug stability, bioavailability, and manufacturing effectiveness.
Siegfried Holding AG
Description: This facilitates complete CDMO services for oral solid dosage forms, steriles, & ophthalmics.
WuXi AppTec
Description: It extensively offers preclinical to commercial services for small molecules, biologics, & oligonucleotide therapeutics.
Labcorp (Fortrea)
Description: Their offering encompasses end-to-end consulting, regulatory affairs, and patient-focused solutions.
Eurofins Scientific
Description: They cover CDMO services for drug product development, including formulation, analytical development, & clinical trial manufacturing.
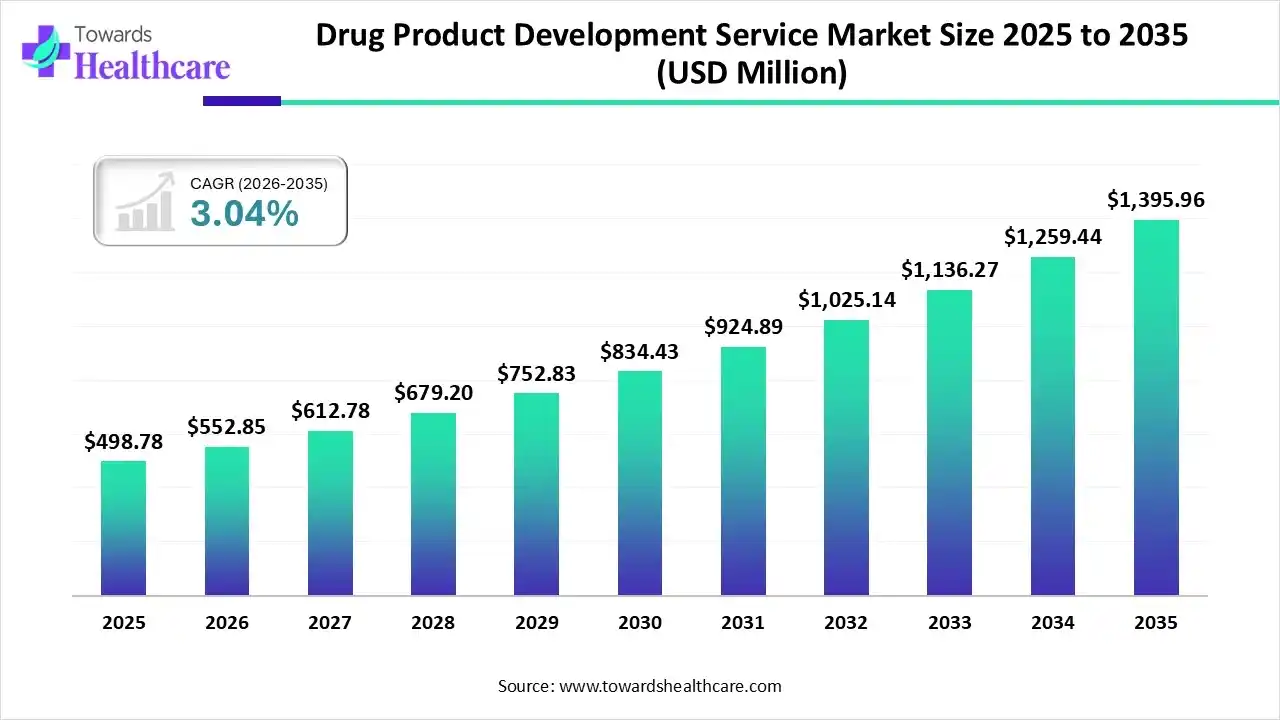
Market Growth
The global drug product development service market size was estimated at USD 340.04 million in 2025 and is predicted to increase from USD 350.38 million in 2026 to approximately USD 458.76 million by 2035, expanding at a CAGR of 3.04% from 2026 to 2035.
Recent Developments in the Drug Product Development Service Market
- In February 2026, Piramal Pharma Ltd. successfully developed, scaled up, and commercialised a tablet-in-capsule drug delivery system at its drug product facilities in Pithampur and Ahmedabad, India.
- In October 2025, Sai Life Sciences, Agility Life Sciences, & Centrix Pharma Solutions launched an Integrated CMC Partnership to facilitate innovator biopharma companies with end-to-end Chemistry, Manufacturing and Controls (CMC) services.
- In September 2025, Cytovance Biologics introduced its in-house formulation development services and promoted its service offerings to biotech and pharmaceutical clients.
Get easy access to all your healthcare reports in one simple dashboard, clear insights, organized data, and everything you need in one place: Access the Dashboard
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
