Top Giants Transforming the Drug Repurposing Market
- Excelra
- Fios Genomics
- Lantern Pharma, Inc.
- Novartis AG
- Paradigm Biopharmaceuticals Ltd
- Predictive Oncology
- Segue Therapeutics, LLC
- Sosei Group Corporation
- Teva Pharmaceutical Industries
Market Growth
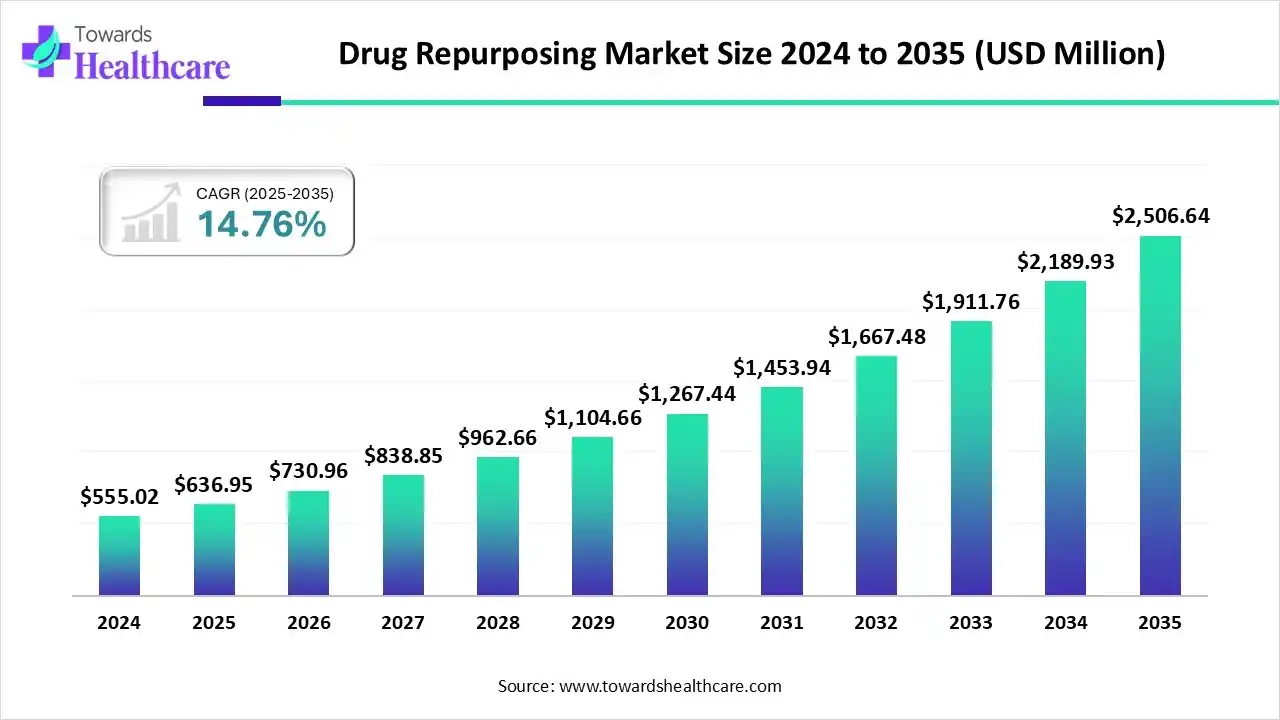
The global drug repurposing market size is calculated at US$ 636.95 million in 2025, grew to US$ 730.96 million in 2026, and is projected to reach around US$ 2506.64 million by 2035. The market is expanding at a CAGR of 14.76% between 2026 and 2035.
Company Landscape with Statistical Summary
Excelra
Overview
-
Headquarters: Hyderabad, India
-
Established: 2001
-
Nature of Business: Provides informatics and analytics services to life sciences companies, integrating clinical, molecular, and outcomes data to accelerate drug discovery and development
Top Products / Offerings
-
Multi‑omics data analytics for early-stage R&D
-
Clinical data management and outcomes informatics platforms
-
Custom analytics services for biotechnology and pharmaceutical clients
Regional Impact
-
Strong presence in India while serving global clients in the US and Europe
-
Supports smaller biotech firms with advanced analytics services
-
Enables multinational research collaborations, advancing life sciences innovation globally
SWOT Analysis
Strengths:
-
Niche expertise in bioinformatics and cheminformatics
-
Strong data-driven culture and leadership
-
Global client servicing with cost-efficient operations
Weaknesses:
-
Margin pressure as a service provider
-
Dependence on R&D budgets which can be cyclical
-
Scaling high-value analytical services requires significant talent and infrastructure
Opportunities:
-
Growing demand for AI/ML-driven drug discovery
-
Expanded partnerships with pharma and biotech
-
Emerging markets offer new client bases
Threats:
-
Large pharma developing in-house analytics capabilities
-
Rapid technological change requiring constant upgrades
-
Regulatory and data privacy challenges in cross-border analytics
Recent Development / Innovation / Announcement
-
Acquired a US-based informatics company to strengthen global bioinformatics offerings
-
Reached annual revenue of ~INR 223 crore, showing healthy growth
-
Expanded European client share while maintaining global service delivery
Fios Genomics
Overview
-
Headquarters: Edinburgh, UK
-
Established: 2008
-
Nature of Business: Specialist bioinformatics company offering data analysis services for genomics, transcriptomics, and proteomics to pharma, biotech, CROs, and academia
Top Products / Offerings
-
Bioinformatics reports translating omics data into actionable insights
-
Data analysis services including biomarker analysis, sequencing, proteomics, and metabolomics
-
Support for both human and non-human research species
Regional Impact
-
Serves global clients, bringing advanced bioinformatics outside traditional hubs
-
Located in an academic research cluster, facilitating industry-academic collaboration
-
Provides cost-efficient services enabling smaller firms to access cutting-edge research
SWOT Analysis
Strengths:
-
Deep specialist expertise in bioinformatics
-
Platform-agnostic service flexibility
-
Strong global client track record
Weaknesses:
-
Growth tied to project availability
-
Competition from large CROs
-
Limited scalability due to specialized talent requirements
Opportunities:
-
Surge in genomics and multi-omics creates increased demand
-
Collaborations with AI/ML firms for predictive analytics
-
Expansion into emerging markets
Threats:
-
Automation and AI may commoditize parts of bioinformatics
-
Data security and regulatory compliance concerns
-
Larger firms developing in-house capabilities
Recent Development / Innovation / Announcement
-
Enhanced service capabilities for multiple data platforms
-
Active in global service markets with top pharma clients
-
Integrated closely with academic research clusters
Lantern Pharma, Inc.
Overview
-
Headquarters: Dallas, Texas, USA
-
Nature of Business: Clinical-stage oncology company using AI, machine learning, and genomics to develop precision oncology therapeutics
-
Vision: Reduce cost, risk, and timeline of oncology drug development using proprietary RADR® platform
Top Products / Pipeline
-
LP‑300: Phase 2 trial for non-smoker NSCLC adenocarcinoma
-
LP‑184: Phase 1 trials in solid tumors including pancreatic, breast, bladder, CNS
-
LP‑284: Phase 1 in non-Hodgkin’s lymphoma
-
RADR® Platform: Uses AI/ML to select compounds, stratify patients, and optimize combinations
Regional Impact
-
US-based, serving global oncology markets
-
AI-driven development supports underserved regions
-
Facilitates collaboration with hospitals, clinical centers, and data banks globally
SWOT Analysis
Strengths:
-
Innovative AI/ML use in oncology
-
Multiple candidates in clinical development
-
Proprietary RADR® platform differentiates it in oncology space
Weaknesses:
-
Revenue dependent on clinical trial outcomes
-
AI/ML translation to market is unproven
-
High burn rate with limited product revenue
Opportunities:
-
Growing demand for personalized oncology therapies
-
Partnerships and licensing deals with larger pharma
-
Potential to repurpose abandoned drugs for new indications
Threats:
-
Clinical trial or regulatory failure risks
-
Competition from AI-driven drug discovery and traditional pharma
-
Biotech funding environment fluctuations
Recent Development / Innovation / Announcement
-
RADR® platform upgraded with AI-powered drug combination prediction
-
Funded with ~$15.9 million cash runway into June 2026
-
LP‑284 dosing initiated in Phase 1 trials for pancreatic and lymphoma indications
Predictive Oncology, Inc.
Overview
-
Headquarters: USA
-
Nature of Business: AI-driven oncology company using a tumor biobank and 3D tumor modeling to support drug discovery and personalized therapy
-
Value Proposition: Predicts tumor response to reduce drug development risk
Top Products / Offerings
-
AI-based tumor response prediction models
-
3D tumor modeling and response testing in vitro
-
Drug discovery support services for biopharma partners
Regional Impact
-
Serves global clients with predictive oncology tools
-
Large tumor biobank enables diverse population modeling
-
Collaborates with academic institutions and smaller biotech firms
SWOT Analysis
Strengths:
-
Proprietary tumor biobank (~150,000 samples)
-
Integration of AI/ML with biological modeling
-
Publicly listed, increasing visibility
Weaknesses:
-
Reliance on predictive models without market-ready drug assets
-
Revenue largely service-based until commercial drugs launch
-
Competition from other AI oncology firms
Opportunities:
-
Rising demand for personalized oncology tools
-
Licensing platform to larger pharma
-
Expansion into other disease areas
Threats:
-
Regulatory uncertainty around AI/ML in drug development
-
Data privacy and biobank management risks
-
Clients developing in-house capabilities
Recent Development / Innovation / Announcement
-
Developed predictive models for 21 new molecules with strong anti-tumor potential
-
Accuracy of 92% in predicting tumor sample responses
-
Expanded 3D tumor modeling services to existing client base
Paradigm Biopharmaceuticals Ltd
Overview
-
Headquarters: Melbourne, Australia
-
Established: 2014
-
Nature of Business: Drug repurposing company focusing on approved or under-used molecules for new indications
Top Products / Pipeline
-
Lead candidate: Injectable pentosan polysulphate sodium (iPPS/PPS) for osteoarthritis
-
Potential applications: OA, MPS, Ross River virus, Chikungunya virus, ARDS
-
Strategy: Leverage known safety profiles to reduce risk and time
Regional Impact
-
Targets global markets including US, EU, Canada, and Australia
-
Could impact millions suffering from osteoarthritis
-
Model encourages small biotech firms to repurpose existing molecules
SWOT Analysis
Strengths:
-
Clinical focus on unmet needs
-
Repurposing reduces development risk
-
Publicly listed company (ASX)
Weaknesses:
-
No large commercial revenues yet
-
Success dependent on clinical trial outcomes
-
Limited diversification of pipeline
Opportunities:
-
Aging population increases OA market demand
-
Potential for licensing and partnerships
-
Apply repurposing model to other indications
Threats:
-
Competition from novel OA therapies
-
Regulatory or clinical trial failures
-
Funding constraints for small biotech
Recent Development / Innovation / Announcement
-
Focus on injectable PPS treatment for OA and other conditions
-
Emphasis on extracting value from under-utilized molecules
-
Investor communications highlight cost- and time-efficient development
Sosei Group Corporation
Overview
-
Headquarters: Tokyo, Japan (R&D in Cambridge, UK)
-
Established: 1990
-
Nature of Business: Global biopharmaceutical company focused on drug discovery and early development with partnerships and licensing
Top Products / Offerings
-
StaR® platform for GPCR-targeted drug discovery
-
Partnered drugs: Ultibro®, Seebri®, Enerzair®
-
Early-stage discovery and licensing services
Regional Impact
-
Japanese presence with global partnership footprint
-
Licensing model contributes to global therapeutic innovation
-
Supports treatments in neurology, immuno-oncology, gastroenterology, inflammation, and rare diseases
SWOT Analysis
Strengths:
-
Proprietary StaR® technology platform
-
Global partnerships with milestone and royalty revenue
-
Efficient early-stage R&D operations
Weaknesses:
-
Revenue dependent on milestone payments and royalties
-
Early-stage pipeline failure risk
-
Operating losses due to R&D investments
Opportunities:
-
Expansion in GPCR-targeted therapies
-
Licensing and emerging market expansion
-
Strategic acquisitions to accelerate growth
Threats:
-
Competition from biotech and large pharma
-
Pipeline failures could affect revenue
-
Currency and regulatory risks
Recent Development / Innovation / Announcement
-
FY 2023 revenue: JPY 12,766 million (US$90.8m), operating loss reflects R&D investment
-
Investing in program-centric R&D and specialty commercialization
-
Raised US$200 million in 2020 for strategic growth
Segue Therapeutics, LLC
Overview
-
Headquarters: New Orleans, Louisiana, USA
-
Established: 2016
-
Nature of Business: Early-stage biotech focusing on drug repurposing for diseases with high unmet need such as pancreatic cancer and fibrotic diseases
Top Products / Pipeline
-
Lead series: Type A – Series 2000 (fibroblast activation), Type B – Series 1000 (tumor invasion)
-
Platform: High-content imaging and phenotypic screening
-
Focus: Rapid repositioning of clinically safe molecules
Regional Impact
-
US-based, aiming to impact oncology and fibrosis therapies globally
-
Collaborates with academic institutions and pharma partners
-
Offers early-stage preclinical solutions with speed and cost efficiency
SWOT Analysis
Strengths:
-
Focused niche in drug repurposing
-
High-throughput screening technology
-
Experienced management team in oncology and fibrosis
Weaknesses:
-
Early-stage, dependent on partnerships and funding
-
Limited commercial track record
-
Pipeline concentrated on few indications
Opportunities:
-
High unmet needs in oncology and fibrosis
-
Potential for global licensing deals
-
Expand repurposing model across other disease areas
Threats:
-
Competition from established biotech and pharma
-
Regulatory approval risks
-
Market adoption uncertainty
Recent Development / Innovation / Announcement
-
Focused on pancreatic cancer and fibrosis treatment candidates
-
Platform enables faster identification of repurposed drugs
-
Active collaborations with US-based academic and clinical partners
Other Major Companies SWOT Analysis:
AbbVie Inc.
Company Overview:
Description: A research-based biopharmaceutical company focused on developing and commercializing advanced therapies in areas of high unmet medical need. Its substantial portfolio of existing small molecules and biologics positions it strategically for drug repurposing efforts, especially in immunology and oncology.
Corporate Information:
- Headquarters: North Chicago, Illinois, United States
- Year founded: 2013 (Spun off from Abbott Laboratories)
- Ownership type: Public (NYSE: ABBV)
History and Background:
Background: Formed to focus on proprietary pharmaceuticals, inheriting a rich pipeline and portfolio from Abbott Laboratories. Drug repurposing is a key strategy for life-cycle management and pipeline expansion, leveraging the known safety profiles of existing compounds.
Key Milestones/Timeline:
- 2024: Continued strong financial performance with total net revenues of $56.3 billion, reinforcing capacity for R&D investment.
- July 2024: Rob Michael assumed the role of CEO, following the retirement of Rick Gonzalez, maintaining leadership continuity and focus on innovation.
- 2025 (planned): Construction of a new $195 million active pharmaceutical ingredient (API) manufacturing facility in North Chicago to support next-generation medicines, including those in immunology and oncology.
Business Overview:
Focus: Discovery, development, manufacture, and commercialization of innovative medicines. Drug repurposing is integrated into the R&D strategy to accelerate development timelines and reduce risk, especially in complex diseases.
Business Segments/Divisions:
Pharmaceutical products: Focused on therapeutic areas including immunology, oncology, neuroscience, and eye care.
Allergan aesthetics: Products and services related to medical aesthetics.
Geographic Presence:
Global: Operations and sales in over 70 countries worldwide.
Key markets: United States (largest market), Europe, and other international regions.
Key Offerings:
Core repurposed potential: Leveraging established drugs like Humira (adalimumab) and compounds from its pipeline for new indications.
Therapeutic focus: Immunological diseases (Crohn's disease, psoriatic arthritis), oncology, and neuroscience disorders, where repurposing can address unmet needs.
End-Use Industries Served:
- Biopharmaceutical & pharmaceutical companies: Internal R&D and collaboration.
- Healthcare providers/hospitals & clinics: Distribution of therapeutic products.
- Clinical research organizations (CROs): Clinical trials for new indications.
Key Developments and Strategic Initiatives:
Strategic focus: Accelerating the use of artificial intelligence (AI) and data convergence to speed up drug discovery, including the identification of repurposing candidates.
R&D investment: Increased adjusted R&D investment to $10.8 billion in 2024.
Mergers & Acquisitions:
Recent focus: Acquisition of Cerevel Therapeutics and Immunogen (completed 2024) to bolster neuroscience and oncology pipelines, which also opens up new compounds for potential repurposing strategies.
Partnerships & Collaborations:
Academic/biotech collaborations: Engagements to leverage external data, genomics, and translational medicine expertise for drug discovery and repurposing insights.
Product Launches/Innovations:
Innovation focus: Advancing combination therapies in its pipeline, such as risankizumab (skyrizi) combinations for inflammatory conditions, representing a form of repositioning for existing assets.
Capacity Expansions/Investments:
2025 investment: $195 million investment in its North Chicago API manufacturing plant for expanding U.S. production capacity.
Regulatory Approvals:
2024 approvals: Continued expansion of indications for key immunology drugs (e.g., Skyrizi for ulcerative colitis, Rinvoq for pediatric patients), representing successful life-cycle management and indication expansion (a form of repurposing).
Distribution Channel Strategy:
- Direct sales force: Targeting healthcare professionals globally.
- Specialty pharmacies: For high-cost, complex biological treatments.
- Wholesalers and distributors: Broad reach across markets.
Technological Capabilities/R&D Focus:
- Focus: Precision medicine, genomics, data convergence, and the use of AI/machine learning (ML) to improve the probability of success in the drug pipeline, which is highly beneficial for repurposing identification.
- R&D investment: Substantial commitment to research and development ($10.8 billion adjusted in 2024).
Core Technologies/Patents:
- Core assets: Patents related to small molecules, biologics, and novel therapeutic modalities.
- Repurposing potential: Significant library of proprietary small molecules and biologics with known mechanisms and safety data.
Research & Development Infrastructure:
Infrastructure: Global R&D centers with capabilities in high-throughput screening, translational medicine, and clinical development.
Innovation Focus Areas:
- Therapeutic areas: Immunology, oncology, neuroscience, and eye care.
- Methodology: AI & data convergence to predict new indications for existing drugs.
Competitive Positioning:
Position: A top-tier biopharmaceutical company globally. Strong market leader in immunology and oncology. Drug repurposing is a key tool for extending the value of its diversified portfolio.
Strengths & Differentiators:
- Strengths: Robust product portfolio, particularly in immunology, strong financial base ($56.3 billion net revenue in 2024), and strategic acquisitions bolstering its pipeline.
- Differentiators: Scale of investment in R&D, advanced use of AI in discovery, and a large library of proprietary compounds.
Market Presence & Ecosystem Role:
Role: Major innovator and producer of specialized medicines. A significant end-user of drug repurposing technology for life-cycle extension and pipeline acceleration.
SWOT Analysis:
- Strengths: Strong global market presence, robust product portfolio (immunology, oncology), high R&D investment, strategic acquisitions.
- Weaknesses: High reliance on key blockbuster drugs like Humira, facing biosimilar competition, and integration risks from recent acquisitions.
- Opportunities: Growth in emerging markets, strategic M&A to expand pipeline, leveraging AI/ML for drug repurposing, and expansion of existing drug indications.
- Threats: Intense competition, patent expirations/biosimilar competition, regulatory challenges, and pricing pressures.
Recent News and Updates:
- August 2025: Announcement of $195 million investment to expand active pharmaceutical ingredient manufacturing in the U.S.
- 2024: Continued positive readouts and regulatory approvals for expanded indications of key immunology and oncology assets.
Press Releases:
- August 12, 2025: AbbVie announces $195 million investment to expand active pharmaceutical ingredient manufacturing in the U.S.
- 2024: Numerous releases detailing positive clinical trial data and regulatory milestones for pipeline and in-market assets.
Industry Recognitions/Awards:
General: Consistently recognized as a top global pharmaceutical company for r&d investment and innovation.
Novartis AG
Company Overview:
Description: A focused, innovative medicines company with a powerful research-driven pipeline. Drug repurposing is a crucial part of its strategy to deliver high-value medicines faster by leveraging existing compounds across its core therapeutic areas.
Corporate Information:
- Headquarters: Basel, Switzerland
- Year founded: 1996 (Merger of Ciba-Geigy and Sandoz)
- Ownership type: Public (six: NOVN, NYSE: NVS)
History and Background:
Background: Formed from the merger of two long-standing chemical and pharmaceutical companies. Following the 2023 spin-off of Sandoz (generics/biosimilars), Novartis is now a pure-play innovative medicines company, intensifying its focus on breakthrough R&D and new technology platforms, which include advanced methods for drug repurposing.
Key Milestones/Timeline:
- 2024: Delivered strong financial results with double-digit growth in sales (net sales $50.3 billion) and net profit, confirming the success of the pure-play medicines strategy.
- 2024: Ranked number one in the access to medicine index.
- 2023: Completion of the spin-off of Sandoz to become a fully focused innovative medicines company.
Business Overview:
Focus: Delivering high-value, innovative medicines to address high disease burden across four core therapeutic areas. Drug repurposing is supported by a large small-molecule and biologics library and is a cost-effective way to accelerate treatments.
Business Segments/Divisions:
Innovative medicines: This is the sole focus post-Sandoz spin-off, covering its branded pharmaceuticals.
Geographic Presence:
- Global: Products available in approximately 140 countries.
- Priority geographies: United States, China, Germany, and Japan.
Key Offerings:
- Core repurposed potential: Leveraging established small molecules and biologics like Entresto (sacubitril/valsartan), Cosentyx (Secukinumab), and Kisqali (Ribociclib) for new or expanded indications.
- Therapeutic focus: Cardiovascular-renal-metabolic, immunology, neuroscience, and oncology.
End-Use Industries Served:
- Biopharmaceutical & pharmaceutical companies: Internal use and R&D partnerships.
- Healthcare providers/hospitals & clinics: Major purchasers and dispensers of Novartis medicines.
- Universities & research institutions: R&D collaborations, including drug repurposing initiatives.
Key Developments and Strategic Initiatives:
- Strategic focus: Deepening focus on five technology platforms: chemistry, biotherapeutics (established), XRNA, radioligand therapy, and gene & cell therapy (emerging). Repurposing efforts are increasingly applied across these platforms.
- Digital/AI strategy: Scaling data science and technology, including the use of AI/ML to improve R&D productivity, which directly benefits drug repurposing project selection and validation.
Mergers & Acquisitions:
Recent focus: Value-creating, bolt-on acquisitions and early-stage deals to strengthen the pipeline, including in radioligand therapy (RLT) and AI capabilities relevant to discovery and repurposing.
Partnerships & Collaborations:
Academic/industry alliances: Network of over 300 academic and 100 industry alliances for mutual scientific interest, including repurposing collaborations (e.g., AI-driven drug discovery partnerships).
Product Launches/Innovations:
Innovation focus: Advancing its pipeline of over 30 potential high-value new medicines. Emphasis on life-cycle management for key brands through indication expansion (repurposing).
Capacity Expansions/Investments:
R&D investment: Ongoing significant investment in R&D, including advanced platforms (XRNA, RLT) and enhanced technical capabilities (e.g., si/XRNA manufacturing).
Capital allocation: Balanced approach with investments in organic business, including R&D and capes.
Regulatory Approvals:
2024 approvals: Reached key innovation milestones, including Scemblix FDA accelerated approval for 1L Ph+ CML-CP and Kisqali EC approval for hr+/her2- stage II and III EBC (expanded indications).
Distribution Channel Strategy:
- Direct sales/marketing: Focus on healthcare providers in priority geographies.
- Global supply chain: Robust manufacturing and logistics network to ensure access to medicines globally.
- Specialty distribution: For advanced therapy products like gene and cell therapies.
Technological Capabilities/R&D Focus:
- Focus: Technology-powered R&D engine focused on delivering transformative medicines. Prioritizing five technology platforms (chemistry, biotherapeutics, RNA, RTL, gene & cell therapy).
- Data science/AI: Significant investments in data science, technology, and AI to accelerate timelines and increase the probability of success, a core driver for repurposing.
Core Technologies/Patents:
- Core assets: Extensive patent portfolio covering small molecules, biologics, and platform technologies (RLT, xRNA).
- Repurposing advantage: Vast library of known compounds with established target and safety data for in silico (computational) repurposing.
Research & Development Infrastructure:
Infrastructure: Global biomedical research centers. Streamlined development portfolio with increased focus and enhanced competencies post-Sandoz spin-off.
Innovation Focus Areas:
- Disease areas: Cardiovascular-renal-metabolic, immunology, neuroscience, oncology.
- Approach: Delivering high-value medicines through technology leadership and R&D productivity improvement.
Competitive Positioning:
Position: Among the top global innovative medicines companies. Strong financial base and a focused strategy on breakthrough science. Its deep expertise across multiple therapeutic areas and technology platforms gives it a strong advantage in drug repositioning.
Strengths & Differentiators:
- Strengths: Focused innovative medicines strategy, strong pipeline, significant investment in advanced technology platforms, and a powerful global market presence ($50.3 billion net sales in 2024).
- Differentiators: Leadership in cutting-edge platforms (RLT, xRNA, cell & gene therapy) alongside a dedicated focus on leveraging data science for R&D productivity.
Market Presence & Ecosystem Role:
Role: Global leader in innovative pharmaceuticals. Active in shaping the future of medicine through investment in new modalities and efficient drug discovery/repurposing models.
SWOT Analysis:
- Strengths: Strong focus on innovative medicines, robust pipeline, leadership in advanced technology platforms, strong financial performance, strong global presence.
- Weaknesses: Integration and focus challenges from previous organizational complexity, ongoing risk of patent expirations on legacy products, and high drug pricing scrutiny.
- Opportunities: Expansion of advanced therapy platforms (RLT, xRNA), significant untapped market potential in cardiovascular disease (e.g., Leqvio), leveraging AI for accelerated drug repurposing.
- Threats: Increasing generic and biosimilar competition, regulatory hurdles, potential negative clinical trial outcomes for key pipeline assets, and global pricing pressures.
Recent News and Updates:
- 2025 guidance: Net sales expected to grow mid- to high-single digit in 2025, with core operating income expected to grow high single to low double-digit.
- 2024: Continued strong momentum in sales growth, driven by key in-market assets like Entresto and Cosentyx.
Press Releases:
2024: Multiple media releases detailing strong financial results, positive clinical trial data, and regulatory approvals across its core therapeutic areas.
Industry Recognitions/Awards:
- 2024: Ranked number one in the access to medicine index.
- General: Consistently recognized for scientific leadership and R&D pipeline strength.
Key Companies and Their contributions and offerings
- Algernon Pharmaceuticals- It has provided drug repurposing of Repirinast for chronic kidney disease (CKD) and N, N-Dimethyltryptamine (DMT) for stroke and traumatic brain injury (TBI).
- Biovista- A major player offered its core services in systematic drug repositioning (SDR) to biopharmaceutical companies,
- Celentyx Ltd- It facilitates bespoke drug discovery services aimed at human immunology, such as the evaluation of existing drugs for potential use in new disease areas (drug repurposing).
- ChemBio Discovery, Inc.- A vital company provides consultancy and contract research services in computational drug discovery and development, which encompass assistance with drug repurposing.
- Chord Therapeutics SA- It has revealed that CRD1 (cladribine), which was being repurposed for the treatment of rare neuroinflammatory diseases
The Drug Repurposing Market: Value Chain Analysis
R&D
This leverages the identification of candidate drugs (through computational or experimental methods), evaluation of their potential in preclinical models, the conduct of clinical trials, and finally, post-market monitoring.
Key Players: BenevolentAI, Recursion Pharmaceuticals, and Healx, etc.
Clinical Trials & Regulatory Approvals
This includes testing of existing drugs for new uses, often fast-tracking them by studying their known safety profiles, and ultimately getting approved by various regulatory bodies.
Key Players: Brigham and Women's Hospital, Karolinska Institutet, etc.
Patient Services & Support
The market provides innovative treatments to patients with rapid and more cost-effective results by determining new uses for existing drugs.
Source- Bristol Myers Squibb (BMS), Cipla, Dr Reddy, etc.
What are the Transforming Developments in the Drug Repurposing Market?
- In October 2025, Nuformix launched a £228,081 open offer to escalate drug repurposing initiatives.
- In January 2025, ParityQC, a quantum architecture company, and the University of Hamburg unveiled the QuADro (Quantum Algorithms for Drug Repurposing) project, which focused on drug development and repurposing using quantum computing.
Partner with our experts to explore the Drugs Repurposing Market at sales@towardshealthcare.com
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
