Explore the Top Companies in the Hemorrhoids Treatment Market
- RG Hospitals
- Abbott Laboratories
- AstraZeneca
- Boston Scientific
- Olympus Corporation
- Conmed
- PACE Hospitals
- Cook Medical
Hemorrhoids Treatment: Pain-Free Pathways
The hemorrhoids treatment market is experiencing robust growth, driven by rising prevalence due to sedentary lifestyles, aging populations, chronic constipation, pregnancy-related cases, and increasing availability of minimally invasive outpatient procedures with shorter recovery times. It includes pharmaceuticals, minimally invasive procedures, surgical interventions, medical devices, and supportive care products used to treat internal and external hemorrhoids. Therapies include OTC and prescription medications, non-surgical office-based procedures, surgical hemorrhoidectomy, stapled hemorrhoidopexy, lifestyle and dietary interventions, and supportive care products.
Market Growth
The global hemorrhoids treatment market size is calculated at USD 1.21 billion in 2025, grew to USD 1.28 billion in 2026, and is projected to reach around USD 2.18 billion by 2035. The market is expanding at a CAGR of 6.05% between 2026 and 2035.
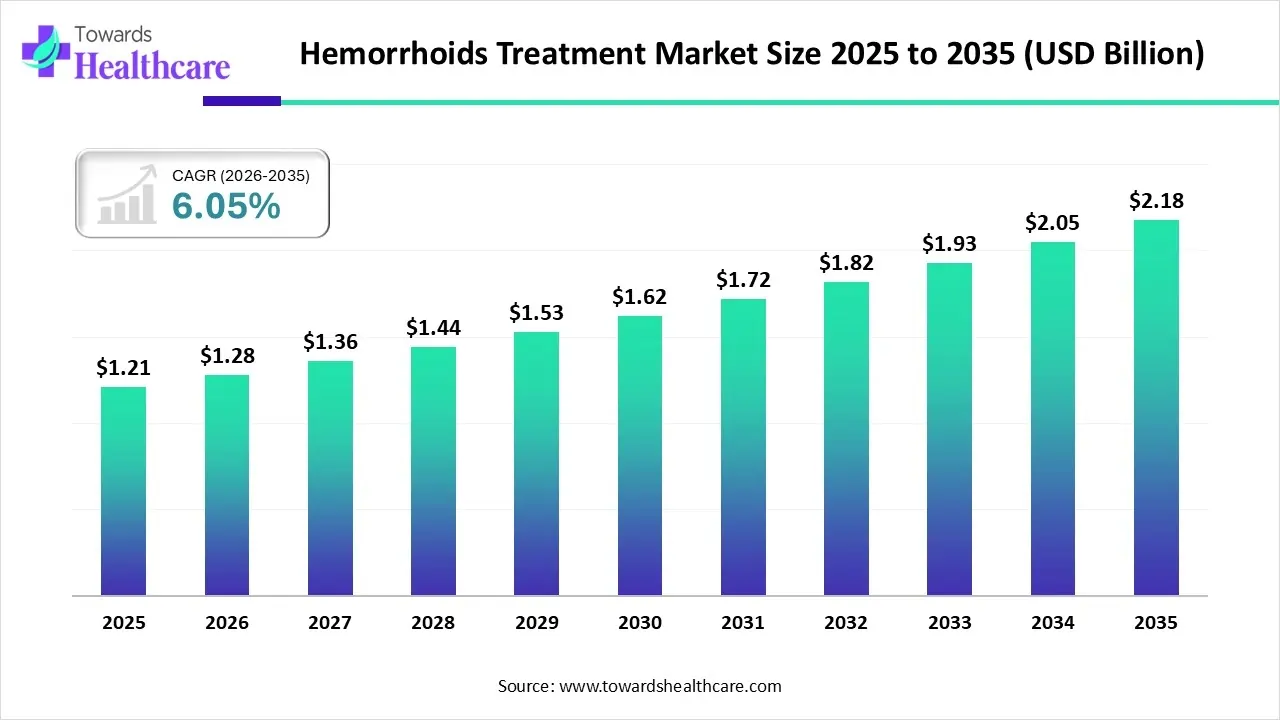
Hemorrhoids Treatment Market Outlook
- Industry Growth Overview: The market is expected to accelerate due to the increasing development of regenerative medicines and the availability of robotic-assisted surgeries. AI-based diagnostic tools also enable patients to diagnose hemorrhoids at an early stage.
- Major Investors: Private equity firms and venture capital investors provide funding to pharmaceutical and biopharmaceutical companies to conduct research and clinical trials related to hemorrhoid therapeutics. They also fund healthcare organizations to adopt advanced surgical tools.
- Global Expansion: Key players collaborate to expand their geographical presence and deliver their proprietary products to a wider consumer base across diverse locations. This strengthens a company’s position in the market.
Top Companies & Their Offerings in the Hemorrhoids Treatment Market
Johnson & Johnson
- Headquarters: New Jersey, United States
- Specialty: Surgical equipment
- Offerings: PROXIMATE PPH Hemorrhoidal Circular Stapler Set
Medtronic
- Headquarters: Dublin, Ireland
- Specialty: Surgical equipment
- Offerings: HET Bipolar System
Norwell Consumer Healthcare, Inc.
- Headquarters: Canada
- Specialty: OTC medication
- Offerings: Hemovel
Max Healthcare
- Headquarters: New Delhi, India
- Specialty: Treatment and patient care
- Offerings: Provides laser and RFA treatments
eXroid Technology Ltd.
- Headquarters: United Kingdom
- Specialty: Treatment and surgical care
- Offerings: Electrotherapy
Company Landscape
Company 1: Medtronic plc
Company Overview
- Company Overview: A global healthcare technology leader, providing a broad range of medical devices, therapies, and services. It is a major player in the Hemorrhoid Treatment Devices market, focusing on surgical and minimally invasive instruments.
Corporate Information (Headquarters, Year Founded, Ownership Type)
- Headquarters: Dublin, Ireland (Operational headquarters in Fridley, Minnesota, USA)
- Year Founded: 1949
- Ownership Type: Public (NYSE: MDT)
History and Background
- History and Background: Started in a garage in Minneapolis as a medical equipment repair shop. Grew to become a pioneer in implantable cardiac pacemakers and expanded into multiple therapeutic areas, becoming one of the largest medical technology companies globally.
Key Milestones/Timeline:
- Key Milestones/Timeline:
- 1949: Founded by Earl Bakken and Palmer Hermundslie.
- 2015: Completed the acquisition of Covidien, significantly expanding its surgical and medical supplies portfolio.
- 2024/2025: Continued focus on expanding its surgical innovations and minimally invasive therapy offerings within its portfolio.
Business Overview
- Business Overview: Provides technology and therapies to treat nearly 70 health conditions, including cardiac, surgical, neurological, and diabetes-related issues. The hemorrhoid treatment products fall under its Minimally Invasive Therapies Group.
Business Segments/Divisions:
- Cardiovascular Portfolio
- Neuroscience Portfolio
- Medical Surgical Portfolio (includes minimally invasive surgical products like staplers and energy devices relevant to advanced hemorrhoidectomy)
- Diabetes
Geographic Presence
- Geographic Presence: Operates in more than 150 countries, with a significant presence in North America, Europe, and the Asia Pacific.
Key Offerings:
- Surgical Stapling Systems: Used for procedures like Stapled Hemorrhoidopexy (PPH).
- Advanced Energy Devices (e.g., LigaSure): Used for vessel sealing and cutting in surgical hemorrhoidectomy, offering faster, less painful procedures.
- Minimally invasive tools for colorectal procedures.
End-Use Industries Served:
- Hospitals and Clinics
- Ambulatory Surgical Centers (ASCs)
- Physicians and Surgeons (Colorectal, General Surgery)
Key Developments and Strategic Initiatives
- Mergers & Acquisitions: Continual evaluation of tuck-in acquisitions to strengthen its Medical Surgical portfolio.
- Partnerships & Collaborations: Engages in collaborations with healthcare providers and group purchasing organizations (GPOs) to streamline technology adoption.
- Product Launches/Innovations: Ongoing development and launch of enhanced surgical instruments and single-use devices, often focused on improving patient outcomes and OR efficiency.
- Capacity Expansions/Investments: Continuous investment in manufacturing and R&D facilities globally to support its broad product portfolio.
- Regulatory Approvals: Secures necessary clearances (e.g., FDA, CE Mark) for its surgical devices, including updated clearances for specific colorectal applications in 2024.
- Distribution channel strategy: A mix of direct sales channels, distributors, and e-commerce platforms, particularly focusing on ASCs and major hospital networks.
Technological Capabilities/R&D Focus
- Technological Capabilities/R&D Focus: Focus on surgical visualization, advanced energy delivery, and robotics to make surgical procedures less invasive and more precise.
- Core Technologies/Patents: Patents related to surgical stapling technology, vessel sealing (LigaSure), and minimally invasive surgical tools.
- Research & Development Infrastructure: Over $2.7 billion spent annually on R&D, with centers of excellence worldwide.
- Innovation Focus Areas: Smart surgery, robotics (e.g., Hugo system), and improved tissue management in abdominal and colorectal surgery.
Competitive Positioning
- Strengths & Differentiators: Global scale, strong brand reputation in medical devices, comprehensive portfolio of advanced surgical instruments, deep hospital relationships.
- Market presence & ecosystem role: A dominant force in the high-value surgical devices market, setting standards for stapled hemorrhoidopexy and energy-based excision.
SWOT Analysis:
- Strengths: Global market leadership, strong R&D pipeline, diversified product offerings.
- Weaknesses: High cost of advanced devices, exposure to currency fluctuations.
- Opportunities: Growth in minimally invasive procedures (MIS), expansion in emerging markets (MEA, APAC).
- Threats: Intense competition from Johnson & Johnson and Boston Scientific, increasing regulatory scrutiny.
Recent News and Updates
- Press Releases: Focused on clinical data supporting its surgical products and expansions in its robotics platform (2024/2025).
- Industry Recognitions/Awards: Frequently recognized for innovation in surgical technology and corporate governance.
Company 2: Johnson & Johnson (J&J)
Company Overview
- Company Overview: A multinational corporation that focuses on pharmaceutical, medical device, and consumer health products. J&J's presence in hemorrhoid treatment is primarily through its Ethicon surgical technology division and its Consumer Health segment (e.g., OTC topical products).
Corporate Information (Headquarters, Year Founded, Ownership Type)
- Headquarters: New Brunswick, New Jersey, USA
- Year Founded: 1886
- Ownership Type: Public (NYSE: JNJ)
History and Background
- History and Background: Founded by three brothers (Robert Wood Johnson, James Wood Johnson, and Edward Mead Johnson) as a surgical dressings company. Pioneered sterile surgical supplies and became a diversified healthcare leader over the next century.
Key Milestones/Timeline
- Key Milestones/Timeline:
- 1886: Founded.
- 1999: Ethicon launched its PPH (Procedure for Prolapse and Hemorrhoids) stapler, a key product in the hemorrhoid surgical space.
- 2023/2024: J&J completed the separation of its Consumer Health business into a new publicly traded company, Kenvue, but still retains a significant presence in the medical device sector through Ethicon.
Business Overview
- Business Overview: Post-split, J&J's primary focus is on Pharmaceuticals and MedTech (Medical Devices). The hemorrhoid device market is addressed by the MedTech segment, specifically under Ethicon.
Business Segments/Divisions
- Business Segments/Divisions:
- Pharmaceuticals (Janssen)
- MedTech (Includes Ethicon, focusing on surgery)
Geographic Presence
- Geographic Presence: Operates and sells products in virtually every country globally, making it a truly multinational corporation.
Key Offerings
- Key Offerings:
- Ethicon Circular Staplers (e.g., PPH systems): Essential for Stapled Hemorrhoidopexy, a procedure for internal hemorrhoids.
- Surgical instruments and wound closure products: Used in traditional and advanced hemorrhoidectomy.
- OTC products (Historically, some brands are now under Kenvue, but the core MedTech presence remains).
End-Use Industries Served
- End-Use Industries Served:
- Hospitals and Clinics (Primary focus for Ethicon)
- Ambulatory Surgical Centers (ASCs)
- General, Colorectal, and Bariatric Surgeons
Key Developments and Strategic Initiatives
- Mergers & Acquisitions: Continued focus on strategic acquisitions within MedTech to enhance surgical portfolio, such as the acquisition of Abiomed in late 2022 to boost its Cardiovascular segment.
- Partnerships & Collaborations: Works with major institutions for clinical studies and training, maintaining a large network of surgical product adoption.
- Product Launches/Innovations: Ongoing innovation in surgical stapling and energy devices to improve tissue handling and reduce complications in procedures like PPH.
- Capacity Expansions/Investments: Major investments in digital surgery and robotic platforms to maintain a competitive edge.
- Regulatory Approvals: Continuous efforts to secure new clearances for its advanced surgical tools globally in 2024.
- Distribution channel strategy: Leverages a vast, established global distribution network, predominantly direct sales to hospitals for high-value surgical devices.
Technological Capabilities/R&D Focus
- Technological Capabilities/R&D Focus: Focus on digital surgery, advanced visualization, robotics, and stapling technology to make surgical procedures more predictable and less invasive.
- Core Technologies/Patents: Extensive patent portfolio covering surgical staplers, wound closure devices, and energy-based surgical instruments.
- Research & Development Infrastructure: R&D spending was over $15 billion in 2023, showcasing a massive commitment to innovation across all healthcare sectors.
- Innovation Focus Areas: Robotics (VELYS, Monarch), digital surgery ecosystems, and next-generation electrosurgery.
Competitive Positioning
- Strengths & Differentiators: Global brand recognition, massive R&D budget, dominant position in surgical staples (Ethicon), separation of Consumer Health simplifies focus on MedTech.
- Market presence & ecosystem role: A fundamental part of the global surgical ecosystem, often being the benchmark for quality and safety in stapled and energy-based surgical procedures.
SWOT Analysis
- SWOT Analysis:
- Strengths: Unmatched brand equity, diversified business model, leadership in surgical technology (Ethicon).
- Weaknesses: High operating scale complexity, potential litigation risk related to some legacy devices.
- Opportunities: Rapid adoption of digital surgery and robotics, market entry in developing economies.
- Threats: Aggressive competition from Medtronic and other surgical device makers, downward pressure on healthcare costs.
Recent News and Updates
- Press Releases: Announcements in 2024 focused on the performance of its MedTech segment and new regulatory approvals for its surgical platforms in the US and EU.
- Industry Recognitions/Awards: Regularly receives awards for innovation in medical technology and is highly rated for corporate responsibility.
Recent Developments in the Hemorrhoids Treatment Market
- In June 2025, a general surgeon at the Osmania General Hospital (OGH) developed an innovative painless hybrid laser sandwich procedure for hemorrhoids. The treatment is devised for people suffering from grade-3 and grade-4 hemorrhoids. The procedure offers the benefits of both conventional and laser surgery, providing patients with faster relief.
- In January 2025, Lux Hospitals launched the ENDOANAL Real-time Ultrasound-Guided Laser Procedure for treating Fistula in Ano. The procedure leverages real-time ultrasound guidance with advanced laser technology to provide highly effective treatment.
Partner with our experts to explore the Hemorrhoids Treatment Market at sales@towardshealthcare.com
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
