Who are the Hereditary Angioedema Therapeutics Market Top Companies and What are Their Offerings?

| Companies | Headquarters | Hereditary Angioedema Therapeutics |
| Takeda Pharmaceuticals | Tokyo, Japan | Takhzyro, Firazyr, and Cinryze |
| BioCryst Pharmaceuticals | Durham, U.S. | Orladeyo |
| CSL Behring | King of Prussia, U.S. | Haegarda, Andembry, and Berinert |
| KalVista Pharmaceuticals | Cambridge, U.S. | Ekterly |
| Pharming Group | Leiden, Netherlands | Ruconest |
| Intellia Therapeutics | Cambridge, U.S. | Lonvo-z |
| Astria Therapeutics | Boston, U.S. | Navenibart |
| Ionis Pharmaceuticals | Carlsbad, U.S. | Dawnzera |
| Pharvaris | Zug, Switzerland | Deucrictibant |
| Sanofi | Paris, France | General HAE management portfolio |
What are the Recent Developments in the Hereditary Angioedema Therapeutics Market?
- In March 2026, the interim data from a Phase 1/2 trial of onvuzosiran (ADX-324) developed by Adarx Pharmaceuticals was presented at the American Academy of Allergy, Asthma & Immunology annual scientific meeting, held in Philadelphia, Pennsylvania. The data revealed that the products showed tolerance and lowered the kallikrein levels, which is a protein triggering HAE swelling attacks, and with the use of this product, the frequency of swelling attacks will be reduced.
- In February 2025, the initiation of the ALPHA-ORBIT Phase 3 clinical trial of navenibart was announced by Astria Therapeutics, Inc. The product is developed to offer rapid and sustained HAE attack prevention, which will be administered every 3 months (Q3M) and every 6 months (Q6M).
Hereditary Angioedema Therapeutics Market Value Chain Analysis
R&D
- The R&D of the hereditary angioedema therapeutics focuses on the development of long-acting monoclonal antibodies, oral on-demand treatments, and one-time CRISPR/RNA-based gene therapies.
- Key players: KalVista Pharmaceuticals, Astria Therapeutics, Intellia Therapeutics.
Clinical Trials and Regulatory Approvals
- The safety profile, symptom relief duration, reduction in monthly attack rates, and quality of life improvements are evaluated in the clinical trials and regulatory approvals of the hereditary angioedema therapeutics.
- Key players: KalVista Pharmaceuticals, Takeda Pharmaceuticals, and CSL Behring.
Packaging and Serialization
- The packaging and serialization of hereditary angioedema therapeutics involves the use of prefilled syringes, 2D DataMatrix barcodes, and tamper-evident seals.
- Key players: Takeda Pharmaceuticals, CSL Behring, BioCryst Pharmaceuticals.
Distribution to Hospitals, Pharmacies
- The specialty pharmacies and authorized wholesalers are responsible for the distribution of the hereditary angioedema therapeutics to the hospitals and pharmacies.
- Key players: Takeda Pharmaceuticals, CSL Behring, BioCryst Pharmaceuticals.
Patient Support and Services
- The home injection training, financial assistance, 24/7 clinical support, and insurance navigation are provided in the support and services of the hereditary angioedema therapeutics.
- Key players: Takeda Pharmaceuticals, CSL Behring, BioCryst Pharmaceuticals.
Global Market Growth
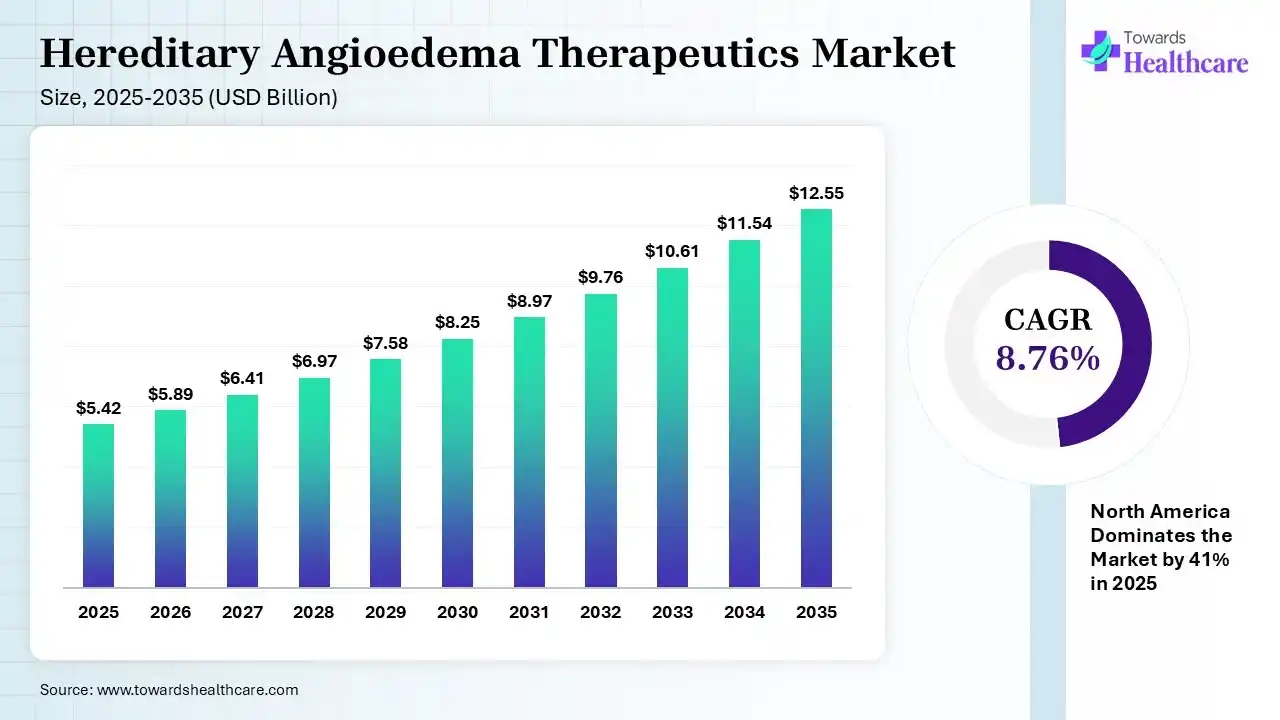
The global hereditary angioedema therapeutics market size was estimated at USD 5.42 billion in 2025 and is predicted to increase from USD 5.89 billion in 2026 to approximately USD 12.55 billion by 2035, expanding at a CAGR of 8.76% from 2026 to 2035.
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
