Latest Updates of Key Players in the Humanized Monoclonal Antibody Market with Forecast

F. Hoffmann-La Roche Ltd
Headquarter: Switzerland
Latest Update: In December 2025, Roche announced that the U.S. Food and Drug Administration (FDA) had approved additional indications for its PATHWAY anti-HER2/neu (4B5) Rabbit Monoclonal Primary Antibody and VENTANA HER2 Dual ISH DNA Probe Cocktail tests.
AbbVie Inc.
Headquarter: United States
Latest Update: In October 2025, AbbVie announced it would unveil novel data from its vigorous antibody-drug conjugate (ADC) platform at the 2025 European Society for Medical Oncology (ESMO) congress.
Amgen Inc.
Headquarter: United States
Latest Update: In April 2025, Amgen announced that the U.S. Food and Drug Administration (FDA) had approved UPLIZNA as the first and only treatment for adults living with Immunoglobulin G4-related disease (IgG4-RD).
Johnson & Johnson
Headquarter: United States
Latest Update: In April 2025, Johnson & Johnson announced that the U.S. Food and Drug Administration (FDA) had approved IMAAVY, a human FcRn-blocking monoclonal antibody, for the management of generalized myasthenia gravis (gMG).
Bristol Myers Squibb
Headquarter: United States
Latest Update: In December 2025, Harbour BioMed announced a multi-year, global strategic partnership and license agreement with Bristol Myers Squibb to discover and develop next-generation many-specific antibodies.
Merck & Co., Inc.
Headquarter: United States
Latest Update: In October 2025, Merck announced it had initiated three Phase 2b trials evaluating the safety and efficacy of tulisokibart, an investigational humanized monoclonal antibody targeting tumor necrosis factor (TNF)-driven cytokine 1A (TL1A).
Value Chain Analysis: Humanized Monoclonal Antibody Market
R&D:
- Major R&D processes in a humanized monoclonal antibody involve basic research, humanization and engineering, upstream and downstream technology, and preclinical and clinical validation.
- Key Players: Johnson & Johnson and Merck & Co., Inc.
Manufacturing Processes:
- The manufacturing of humanized monoclonal antibodies (mAbs) is a complex, many-stage bioprocess that integrates genetic engineering, cell biology, and advanced purification processes.
- Key Players: F. Hoffmann-La Roche Ltd. and AbbVie Inc.
Patient Services:
- Major patient services includes in humanized monoclonal antibody treatment focus on improving access, ensuring safety during administration, offering education, and providing financial assistance for these advanced-cost and dedicated therapies.
- Key Players: Amgen Inc. and AstraZeneca PLC
Market Forecast
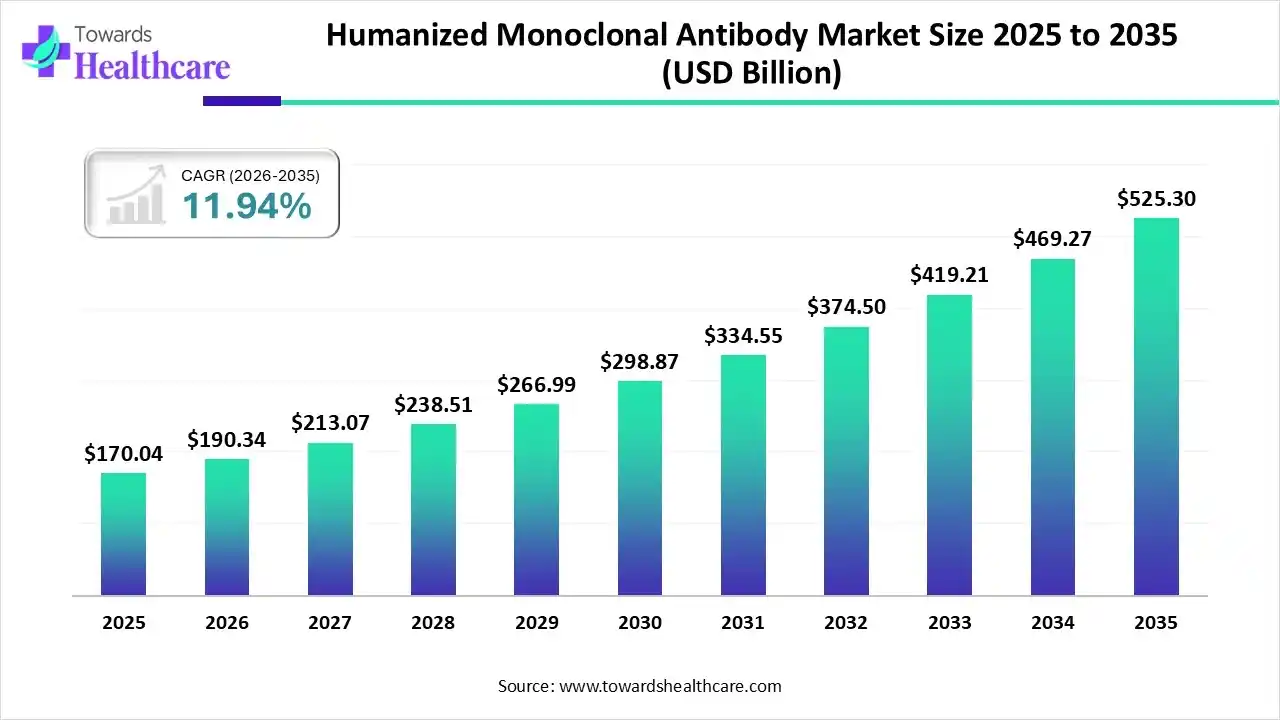
The global humanized monoclonal antibody market size was estimated at USD 170.04 billion in 2025 and is predicted to increase from USD 190.34 billion in 2026 to approximately USD 525.3 billion by 2035, expanding at a CAGR of 11.94% from 2026 to 2035.
Recent Developments in the Humanized Monoclonal Antibody Market
- In January 2026, Tonix Pharmaceuticals Holding Corp., a fully integrated, commercial biotechnology company, obtained data on TONMYA, which was examined as TNX-102 SL, at the 2026 Non-Opioid Pain Therapeutics Summit.
- In October 2025, Harbour BioMed announced the launch of its first fully human Generative AI HCAb Model powered by its Hu-mAtrIx AI platform, built upon the Harbour Mice platform.
- In December 2025, Mirum Pharmaceuticals, Inc., a leading rare disease company, announced its entry into a definitive agreement to acquire Bluejay Therapeutics, a privately held biotechnology company focused on viral and liver disease.
Become a valued research partner with us, please feel free to contact us at sales@towardshealthcare.com
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
