Latest Updates of Companies in the Induced Proximity Molecules Market

| Company | Headquarters | Latest Update |
| Kymera Therapeutics | United States | In April 2026, Kymera Therapeutics, Inc. announced that Gilead Sciences, Inc. had exercised its option to exclusively license KT-200, a first-in-class, oral CDK2 molecular glue degrader development candidate discovered and characterized by Kymera, under their strategic collaboration agreement. |
| Nurix Therapeutics | United States | In January 2025, Nurix Therapeutics, Inc., a clinical-stage biopharmaceutical company focused on the discovery, advancement, and commercialization of targeted protein degradation medicines, the next frontier in innovative drug design aimed at improving treatment choice for patients with cancer and inflammatory diseases, outlined significant objectives and anticipated milestones. |
| Monte Rosa Therapeutics | United States | In March 2026, Monte Rosa Therapeutics, Inc., a clinical-stage biotechnology company developing new molecular glue degrader (MGD)-based medicines, reported business highlights. |
| C4 Therapeutics | United States | In March 2026, C4 Therapeutics announced a partnership with Merck KGaA to develop targeted protein degraders for oncogenic proteins. |
| Bristol Myers Squibb | New Jersey | In March 2026, Evotec received a $10 million milestone payment from Bristol Myers Squibb following the initiation of a Phase 1 clinical study evaluating BMS. |
Recent Developments in the Induced Proximity Molecules Market
- In April 2026, an ad hoc announcement pursuant to Art. 53 LR Molecular Partners AG, a clinical-stage biotech company developing a novel class of custom-built protein drugs known as DARPin therapeutics, announced that Clare Fisher was elected by shareholders
- In November 2025, General Proximity, a biotechnology organization discovering and developing the next generation of induced proximity medicines, announced a strategic multi-target collaboration with Daiichi Sankyo through its Daiichi Sankyo Research Institute in Boston to apply General Proximity's proprietary OmniTAC discovery platform to oncology programs of interest to Daiichi Sankyo.
- In May 2025, VantAI, a frontrunner in generative AI and proximity-based therapeutics, announced that it had entered into a second expansion of its collaboration and license agreement with Blueprint Medicines, a worldwide biopharmaceutical organization, to prioritize targets and design and advance new therapies for areas of high medical need.
Supply Chain Analysis
R&D:
- R&D processes for induced proximity molecules include target selection and ligand discovery, structural biology and modeling, ternary complex assessment, proteomics and functional screening, and ADME and pharmacokinetic optimization.
- Key Players: Proximity Therapeutics & Triana Biomedicines
Manufacturing Processes:
- The major manufacturing processes for induced proximity molecules such as PROTACs, LYTACs, and molecular glues center on modular assembly, linker chemistry, and high-throughput purification.
- Key Players: Arvinas and Amgen
Patient Services:
- Patient services are specifically in oncology and neurodegeneration, involving specialized patient services to manage their unique catalytic, event-driven pharmacology.
- Key Players: Plexium Inc.
Market Growth
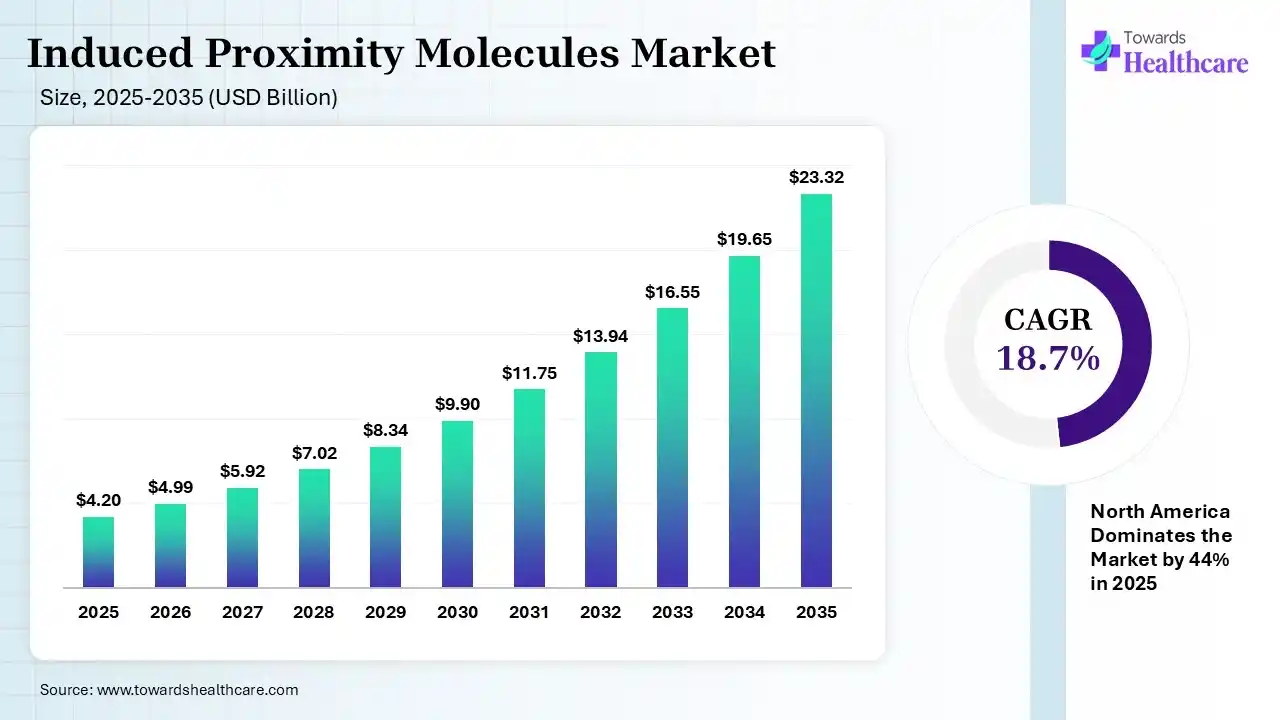
The global induced proximity molecules market size was estimated at USD 4.2 billion in 2025 and is predicted to increase from USD 4.99 billion in 2026 to approximately USD 23.32 billion by 2035, expanding at a CAGR of 18.7% from 2026 to 2035. The induced proximity molecules market is growing because these molecules have the strength to unlock a wide portion of the disease targets that were once thought inaccessible.
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
