Who are the Inhalable Biologics Market Top Vendors and What are Their Offerings?

Company Profile
| Companies | Headquarters | Inhalable Biologics |
| AstraZeneca | Cambridge, UK | Inhaled monoclonal antibodies and biologics |
| GlaxoSmithKline (GSK) | London, UK | Ellipta dry powder inhaler platform |
| MannKind Corporation | Danbury, U.S. | Offers Afrezza |
| Lonza Group | Basel, Switzerland | Provides inhalable biological proteins |
| Cipla | Mumbai, India | Offer inhaler devices and biosimilars |
| Vectura Group | Chippenham, UK | Inhaled drug delivery for biologics and complex molecules |
| Aradigm Corporation | Hayward, U.S. | Inhaled liposomal formulations for severe lung infections and cystic fibrosis |
| Teva Pharmaceutical Industries | Tel Aviv, Israel | Digihaler smart devices |
| Inovio Pharmaceuticals | Plymouth Meeting, U.S. | DNA-based inhalable vaccines and therapies |
| Spero Therapeutics | Massachusetts, U.S | Inhaled antibiotics and biologics |
Inhalable Biologics Market Value Chain Analysis
R&D
- The R&D of the inhalable biologics focuses on the development of advanced particles and technosphere dry powder technology.
- Key players: AstraZeneca, GlaxoSmithKline, MannKind Corporation.
Clinical Trials and Regulatory Approvals
- The device-drug deposition patterns, bioequivalence of inhalable biologics, and local lung mucosal safety are evaluated in the clinical trials and regulatory approvals of the inhalable biologics.
- Key players: AstraZeneca, GlaxoSmithKline, MannKind Corporation.
Patient Support and Services
- The digital health platforms and connected smart inhalers are provides for the patient support and services for inhalable biologics.
- Key players: AstraZeneca, GlaxoSmithKline, Teva Pharmaceutical Industries.
Market Growth
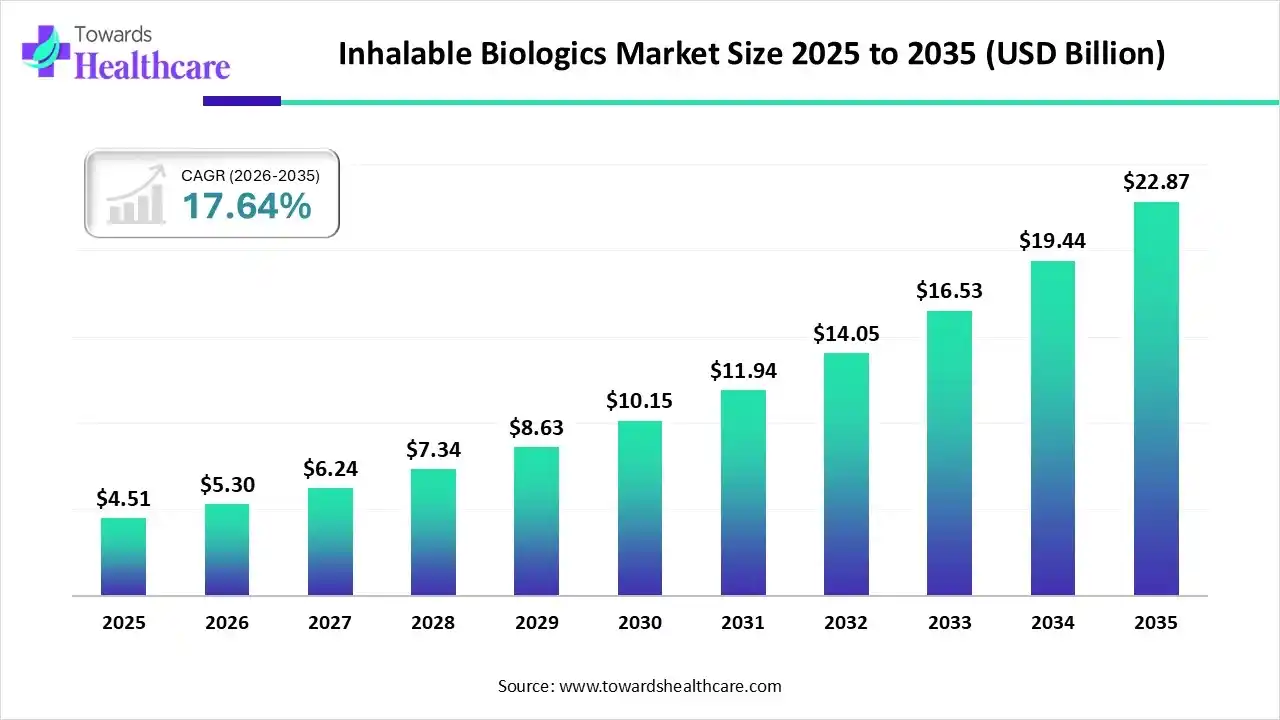
The global inhalable biologics market size was estimated at USD 4.51 billion in 2025 and is predicted to increase from USD 5.3 billion in 2026 to approximately USD 22.87 billion by 2035, expanding at a CAGR of 17.64% from 2026 to 2035.
What are the Recent Developments in the Inhalable Biologics Market?
- In June 2025, Quattrii, the ‘first ever’ dry powder inhaler (DPI) engine, delivering large volumes of biologic and mRNA molecules, with approximate 70% of the drug in a single inhalation, was launched by Cambridge Healthcare Innovations (CHI).
- In March 2025, the phase-2 clinical trial of the next-generation, inhaled COVID-19 vaccine was announced to have been initiated by McMaster University, where they will identify the safety of the vaccine in a broad study group.
Now managing your entire Inhalable Biologics Market data is simple - everything in one place. Access the Dashboard
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
