Clinical Trials Market Size, Company List and Regional Growth
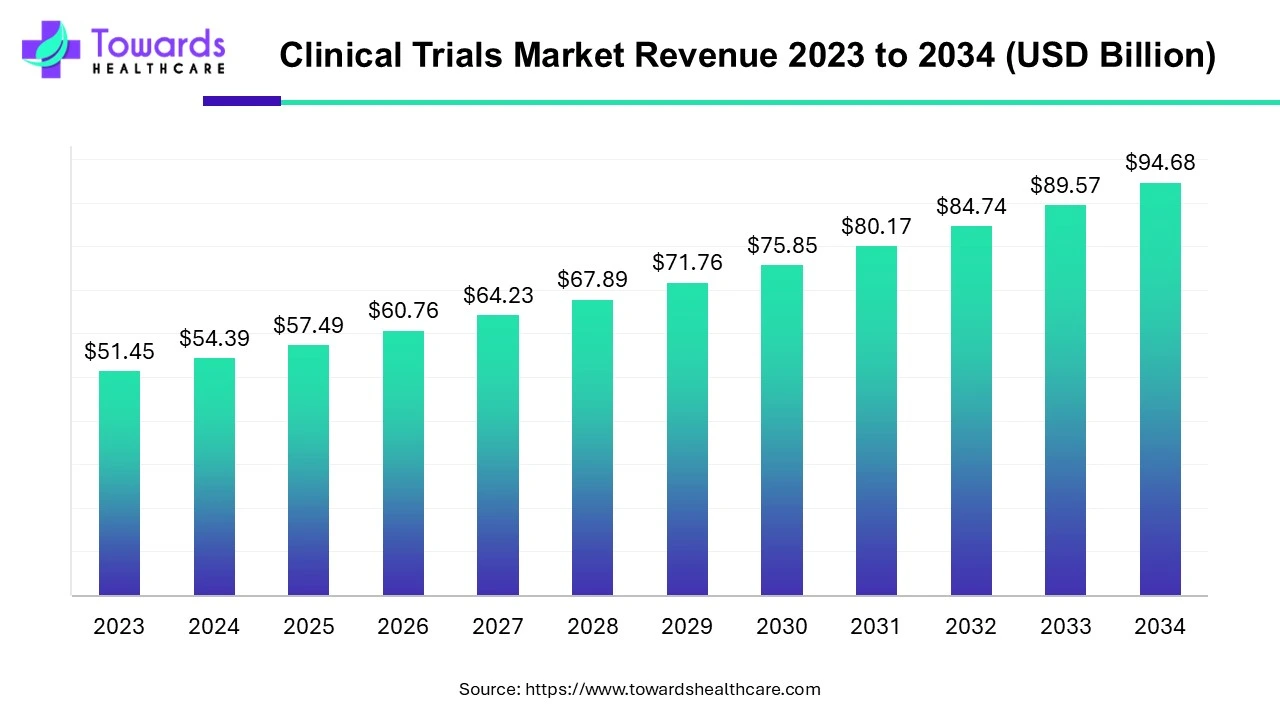
The global clinical trials market size is calculated at USD 98.91 billion in 2026 and is expected to be worth USD 174.18 billion by 2035, expanding at a CAGR of 5.7% from 2026 to 2035, as a result of growing demand for personalized medicines, rising investment in R&D and adoption of decentralized clinical trials.
Key Takeaways
- Clinical trials sector pushed the market to USD 92.88 billion by 2025.
- Long-term projections show USD 174.18 billion valuation by 2035.
- Growth is expected at a steady CAGR of 5.7% in between 2026 to 2035.
- North America dominated the clinical trials market by 60% in 2024.
- Asia Pacific is estimated to be the fastest-growing during the forecast period.
- By phase, the phase 3 segment led the market in 2024.
- The North America clinical trials market is set to grow from US$ 22.52 billion in 2025 to US$ 30.69 billion by 2034 at a CAGR of 3.50%.
- The Middle East and Africa clinical trials market, valued at US$ 3.81 billion in 2024, is projected to reach US$ 7.36 billion by 2034, growing at a CAGR of 6.80%.
- The Europe clinical trials market is projected to grow from US$ 14.25 billion in 2025 to US$ 21.73 billion by 2034 at a 4.8% CAGR.
- The Latin America clinical trials market is projected to grow from US$ 4.35 billion in 2024 to US$ 7.94 billion by 2034 at a 6.2% CAGR.
- The APAC clinical trials market is expected to grow from US$ 10.88 billion in 2024 to US$ 26.96 billion by 2034 at a 9.5% CAGR.
- By phase, the phase 2 segment is anticipated to grow at the fastest rate during the predicted time.
- By study design, the interventional study segment dominated the market in 2024 and is estimated to grow at the fastest rate during the forecast period.
- By intervention, the oncology segment led the market in 2024 and is estimated to achieve the fastest CAGR during the forecast period.
Key Indicators and Highlights
| Key Elements |
Scope |
| Market Size in 2025 |
USD 92.88 Billion |
| Projected Market Size in 2035 |
USD 174.18 Billion |
| CAGR (2025 - 2035) |
5.7% |
| Leading Region |
North America by 60% |
| Market Segmentation |
By Phase, By Study Design, By Indication, By Geography |
| Top Key Players |
Parexel, IQVIA, Charles River Laboratory, Omnicare, Kendle, Chiltern, Pharmaceutical Product Development, LLC |
41% of the clinical trial outcomes were submitted to the ClinicalTrials.gov federal database in the fiscal year 2022, with a rise from 34% in 2021
Clinical trials refer to a medical research study that evaluates the safety and effectiveness of new drugs, medical devices, diagnostic tools, or other medical interventions in humans. Clinical trials are conducted in phases, each with specific objectives and designs. Clinical trials are critical in advancing medical knowledge and improving patient care by identifying new and effective treatments, improving existing treatments, and identifying potential risks or side effects of medical interventions. The clinical trials market includes various participants, including pharmaceutical and biotechnology companies, contract research organizations (CROs), academic research institutions, regulatory bodies, and healthcare providers.
The government is exclusively making efforts to drive the clinical trials market growth. They are allocating new resources to data collection & scientific infrastructure, strengthening the protection of intellectual property, and affording access to patient populations. The R&D environment is also improving steadily across the globe. The world is hosting a growing pool of scientific & development expertise, adoption of advanced technology & quality clinical outsourcing providers. In addition, the various amendments made by the regulatory market to change the clinical trials evaluation standards is driving the market growth. Most countries are now focused on more efficient registration & approval processes with a raft of significant improvements introduced worldwide. Governments across the world are investing in new resources to support data collection and scientific infrastructure, which is helping to create a more efficient and effective clinical trial process. In addition, governments are taking steps to strengthen the protection of intellectual property, which is crucial for promoting innovation and attracting investment in the clinical trials market.
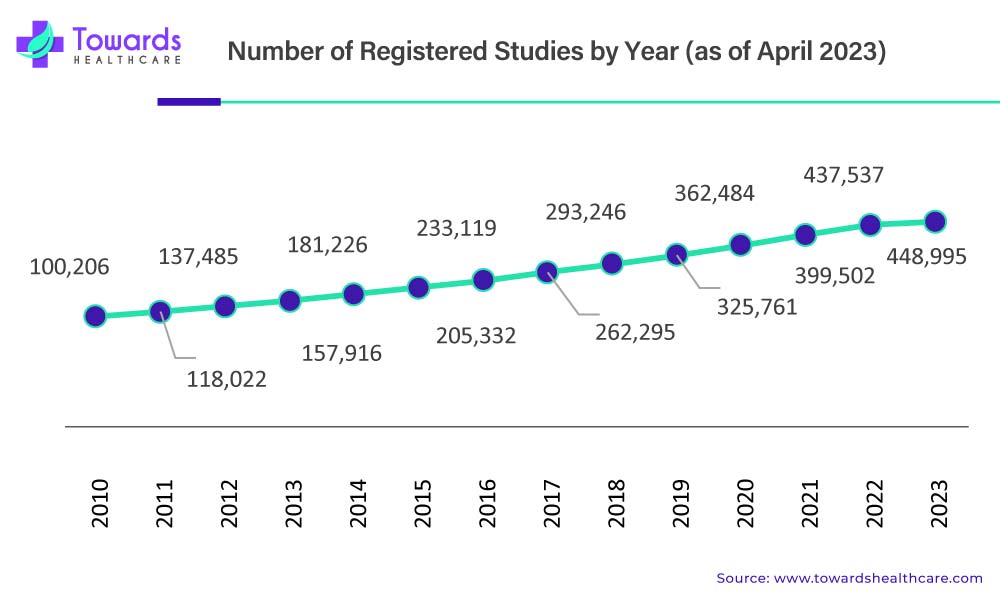
Furthermore, favorable government support and stringent regulations in clinical trials are anticipated to boost the demand. For instance, in March 2020, the FDA launched a Coronavirus Treatment Acceleration Program (CTAP) for possible therapies to speed up the development of treatment for the global disease caused by the coronavirus. Moreover, ClinicalTrials.gov has published a database of around 448,686 privately and publicly funded clinical studies around the globe in 221 countries. And according to them, there has been a significant increase in the number of clinical trials performed over the years.
The clinical trials market is driven by several factors, including the increasing prevalence of chronic diseases, the growing demand for personalized medicine, and the need for innovative and effective treatments. With the increasing focus on precision medicine and personalized healthcare, there is a growing demand for clinical trials that can identify biomarkers and other patient-specific factors that can be used to tailor treatments to individual patients. In addition, the clinical trials market is also being shaped by the increasing use of technology and data analytics to improve trial design and patient recruitment, as well as the use of virtual and decentralized clinical trials to reduce costs and increase patient access.
Another important factor driving the growth of the clinical trials market is the increasing access to patient populations. Governments are taking steps to improve patient access to clinical trials by funding programs that encourage patient participation and by creating incentives for healthcare providers to participate in clinical trials. The R&D environment is also improving steadily across the globe. Many countries are hosting a growing pool of scientific and development expertise, and the adoption of advanced technology is making it easier to conduct clinical trials and collect data. In addition, quality clinical outsourcing providers are becoming more widely available, making it easier for companies to conduct clinical trials in multiple locations around the world.
Patient-centric Approach is Leading the Way in Clinical Trails
A modern approach that focuses on placing the patient at the center of the trial design and execution, with the aim of improving patient outcomes and satisfaction. Patients are involved in the trial design process from the outset, providing input on study protocols, patient information materials, and other aspects of the trial. With a focus on patient engagement and retention throughout the trial, efforts are made to enlist a diverse and representative patient population. In order to execute patient-friendly trial designs, the needs and preferences of patients are kept in mind, with a focus on minimizing the burden and inconvenience for patients. In addition, patient advocacy groups are often involved in the clinical trial process, advocating for patient needs and interests and providing support and resources to patients throughout the trial. By executing patient-friendly trial designs, companies can improve patient recruitment and retention rates, leading to more accurate and reliable data.
Companies that prioritize patient engagement and satisfaction are more likely to build goodwill and positive relationships with patients, which can have long-term benefits for the company. Furthermore, reaching out to patients where they are while performing clinical trials is the future of clinical trials. Some of the ways in which the future trial model is seeking to meet patients where they are include:
Virtual and Decentralized Trials: With the help of technology, clinical trials can be conducted virtually or decentralized, enabling patients to participate from the comfort of their own homes. This can increase participation rates, particularly among patients who live far from trial sites, have mobility issues, or have other health concerns that make it difficult to travel to study centers.
Real-World Evidence: Real-world evidence (RWE) is data collected outside of traditional clinical trial settings, such as electronic health records, patient-generated health data, and other sources. By incorporating RWE into clinical trials, researchers can get a more comprehensive understanding of patient experiences and outcomes, which can help to improve trial design and recruitment.
Patient Engagement: Engaging patients in the trial process, by providing regular updates, soliciting feedback, and offering support, can help to build trust and increase patient participation in trials.
Growing Disease Variation And Prevalence
The growing prevalence of disease and incidence of new diseases is expected to give a further boost to the clinical trial market. For instance, according to the WHO statistics reported in September 2022, Non-communicable diseases (NCDs) kill 41 million people each year, equivalent to 74% of all deaths globally. In addition, according to WHO estimates for 2021, 1.5 million people were newly infected with HIV.
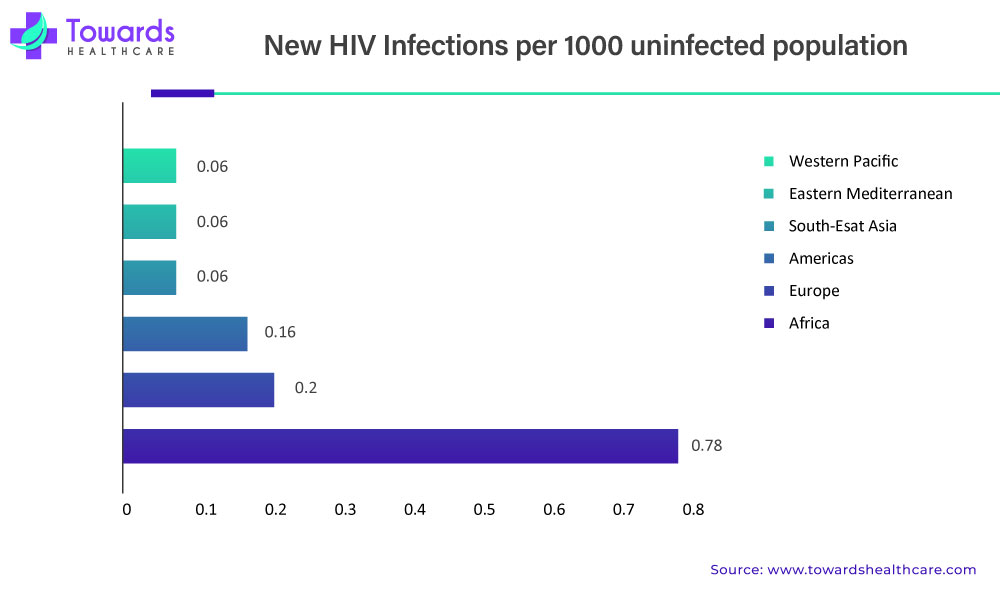
The worldwide population has varied disease profiles with emerging countries having the most diverse disease profile. This is expected to boost the clinical trial of new or rare diseases which otherwise would not have found any sponsors. More patients having a specific disease would act as a stimulus for biopharmaceutical companies to invest more in clinical trials for a disease segment. A diverse population would also mean easy recruitment of patients and a faster clinical trial process. Rare diseases are given the status of “Orphan disease” in U.S. and biopharmaceutical companies who sponsor clinical trials for orphan drugs would get incentives for the process. This trend is likely to have a positive impact on clinical trials for rare diseases thereby increasing the global clinical trials market.
In addition, advancements in medical technology and innovative research approaches have made it possible to conduct clinical trials for a wider range of diseases and conditions, including rare and orphan diseases that were previously difficult to study. This has opened up new opportunities for pharmaceutical companies and clinical research organizations to develop treatments for previously untreatable or poorly treated conditions. Moreover, the increasing prevalence of chronic diseases such as cancer, diabetes, and cardiovascular diseases has also contributed to the growth of the clinical trial market. The burden of these diseases on healthcare systems and patients is significant, and there is a growing need for innovative and effective treatments that can improve patient outcomes and quality of life.
As new diseases emerge and existing diseases evolve, there is a growing need for effective treatments that can improve patient outcomes and quality of life. This has led to an increase in demand for clinical trials that can test the safety and efficacy of new treatments. As a result, there has been a corresponding increase in the number of clinical trials being conducted globally, with many countries investing in research and development to address the growing demand.
Shift Towards Personalized Medicines
The paradigm shift towards personalized medicine is expected to have a positive impact on the clinical trial market. The classic clinical trial process is carried out on thousands of people while personalized medicine will focus only on the effect of drugs on individual patients for a specific period. Very few medicines in the development phase pass all phases of a clinical trial due to the traditional clinical trial approach. The mentality of “one size will fit all” is acting as a barrier for drugs that are currently in pipeline but would never see the day. The use of pharmacogenetics in the clinical trial process is expected to increase the number of drugs passing all phases of the clinical trial process. The shift towards personalized medicine is expected to increase the use of pharmacogenetics in the clinical trial phase thereby increasing the pipeline of drugs. This trend is expected to instigate biopharmaceutical companies to invest more in the clinical trial phase.
The use of digital health technologies, such as wearables, sensors, and mobile apps, has enabled researchers to collect large amounts of real-time patient data, which can be used to design more efficient and effective clinical trials. Digital health technologies have the potential to improve clinical trials in a number of ways such as improved patient monitoring, enhanced patient engagement, precise data collection, improved trial design, and faster recruitment. Digital health technologies have enabled decentralized clinical trials, where patients can participate in clinical trials from their own homes using mobile apps, wearables, and telemedicine. This has the potential to reduce the cost and time required for clinical trials and increase patient participation.
Regulators have recognized the potential of digital health technologies to improve clinical trials and have been increasingly accepting of digital health-tech results as evidence of improved treatment effectiveness. The vast amounts of data generated by digital health technologies can be analyzed using AI and machine learning algorithms to identify patterns and generate insights. This has the potential to improve the design and implementation of clinical trials, as well as the evaluation of treatment effectiveness.
Digital health technologies are enabling a more patient-centric approach to clinical trials, where patients have more control over their participation and can provide feedback on the design and implementation of clinical trials. This has the potential to improve patient engagement and retention in clinical trials. In addition, Regulators are increasingly using real-world evidence generated by digital health technologies to evaluate treatment effectiveness. This has the potential to reduce the cost and time required for clinical trials and enable faster regulatory approval of treatments. This has created opportunities for innovative companies in the digital health space to partner with pharmaceutical companies and CROs to conduct more efficient and effective clinical trials, ultimately leading to faster drug development and improved patient outcomes.
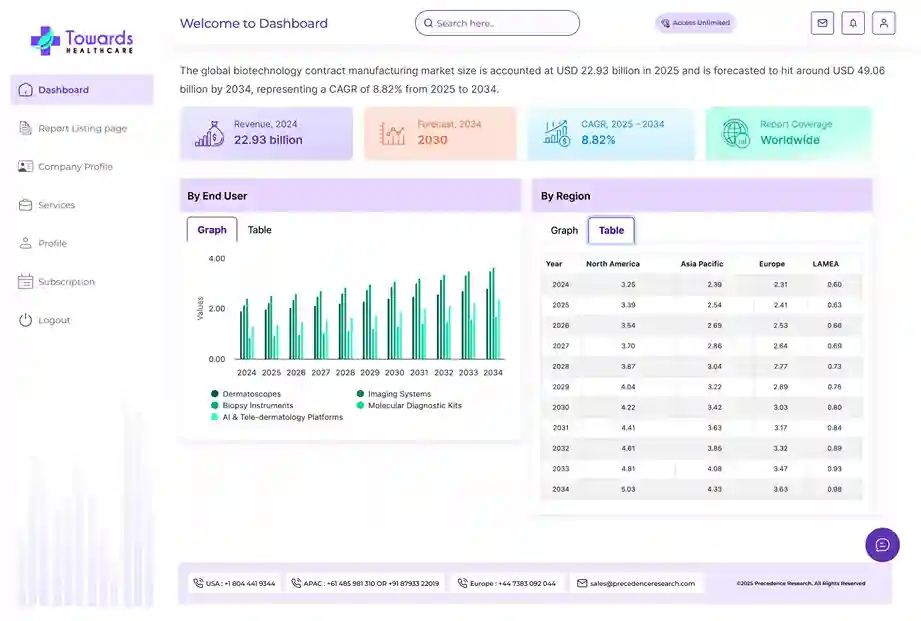
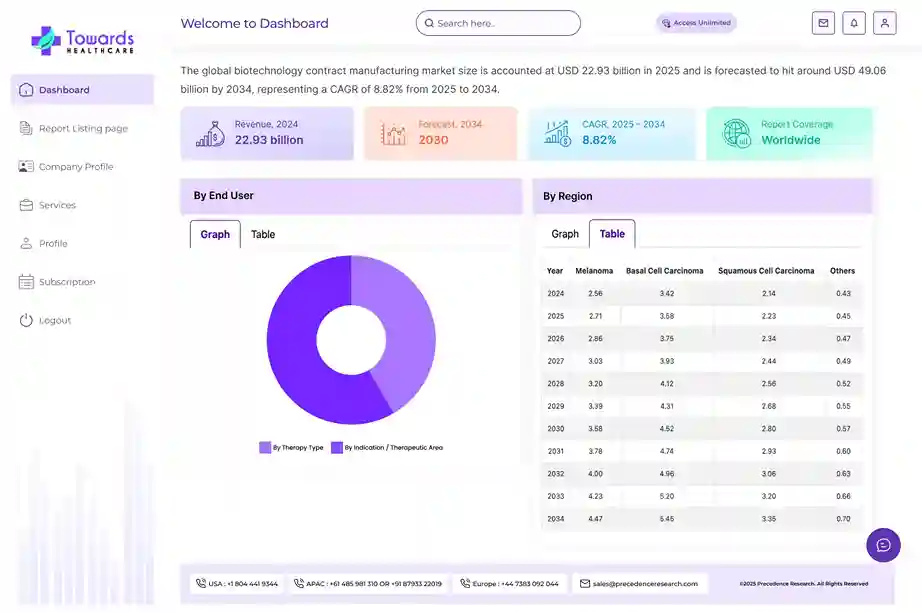
Segmentation outlook
Interventional Study is Projected to Dominate the Study Design Segment of the Clinical Trials Market Revenue
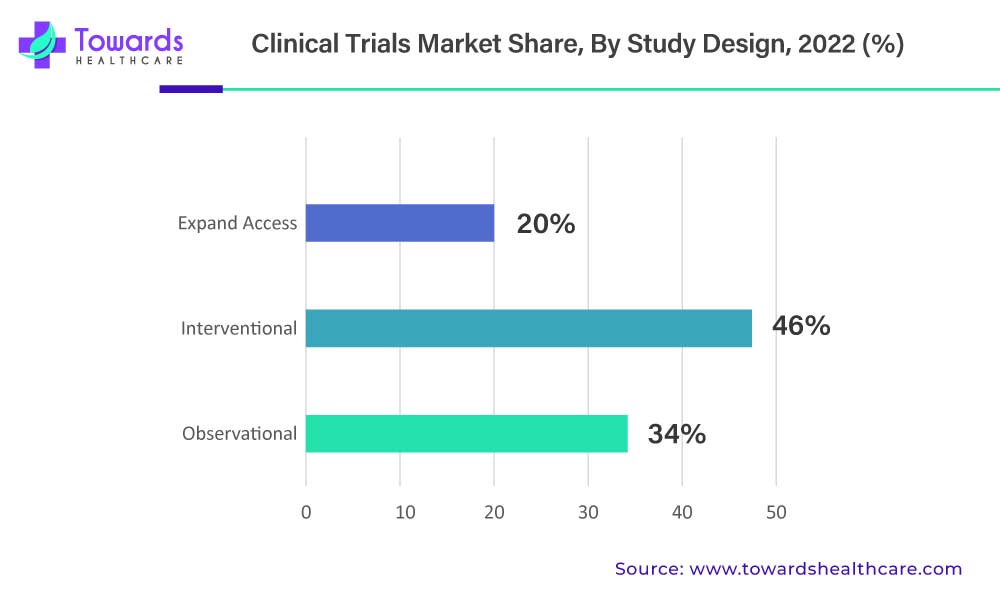
The requirement for clinical trials in developing diagnostic tests and vaccines for viral diseases such as SARS-CoV-2 has augmented the demand for clinical trials exponentially. Thus, the high incidence of novel viral diseases and ongoing technological improvements in clinical trials are major reasons for the high revenue share of interventional studies. The interventional study segment is expected to grow at a CAGR of 5.4% from 2022 to 2030. The interventional design segment accounted largest market share 46% in 2022.
Oncology is Projected to Dominate the Indication Segment of the Clinical Trials Market Revenue
Constant research on cancer treatment and increasing demand for precision medicine are the major reasons responsible for the high market share of oncology. The oncology segment was valued at USD 25 billion by 2030. The oncology segment accounted for a revenue share of 24% in 2021 and is expected to witness growth at a CAGR of 6.4% over the forecast period from 2022 to 2030.
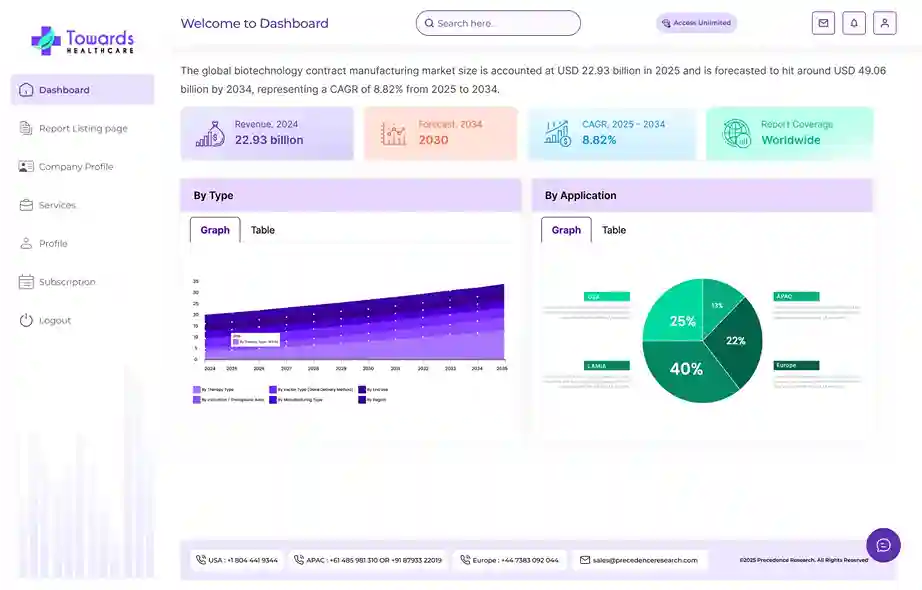
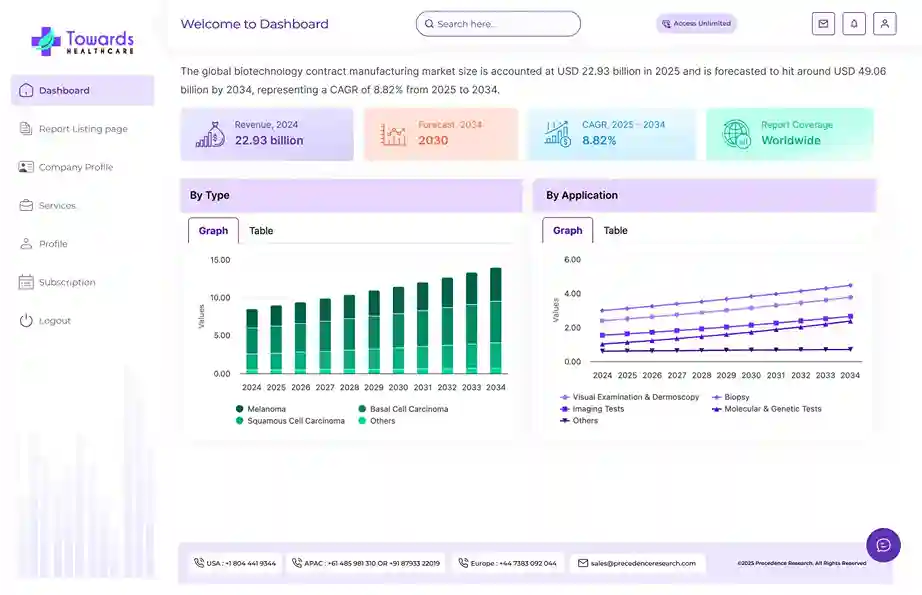
Regional Insights
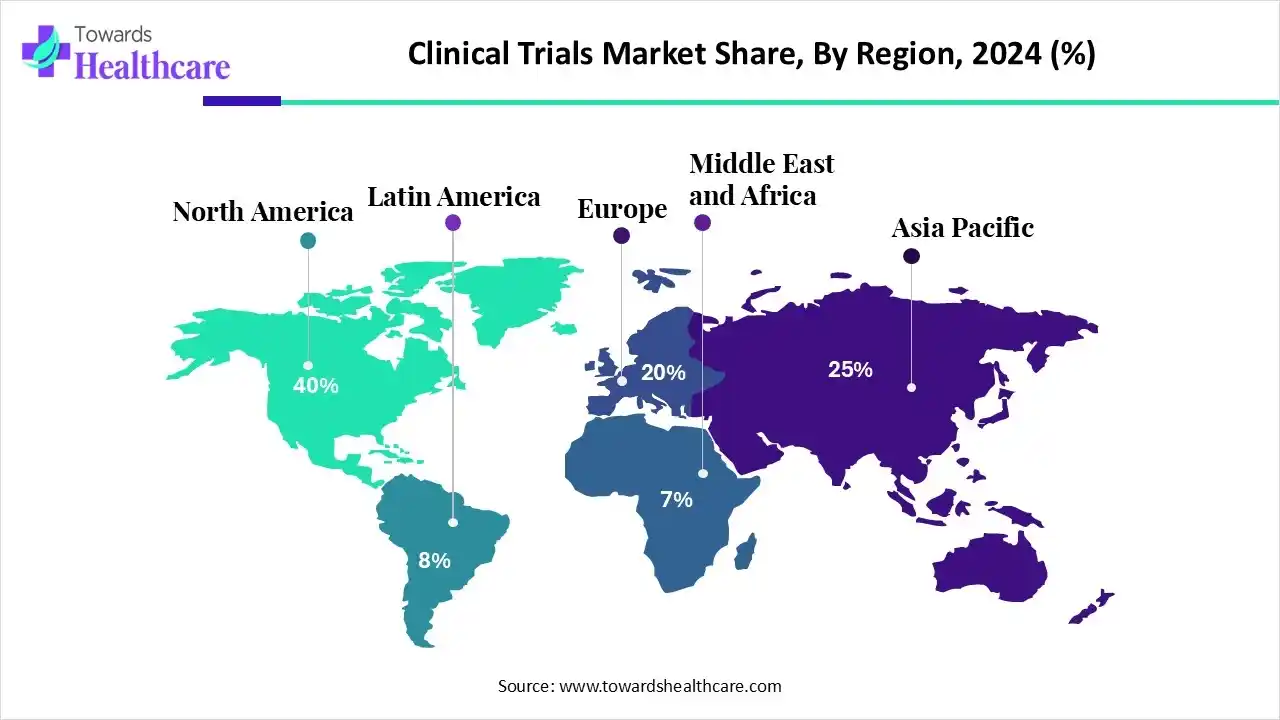
Increasing Investments Dominated North America
The North America clinical trials market is valued at US$ 55.48 billion in 2025, expected to grow to US$ 58.82 billion in 2026, and projected to reach approximately US$ 99.10 billion by 2035. This reflects a compound annual growth rate (CAGR) of 5.40% from 2026 to 2035.
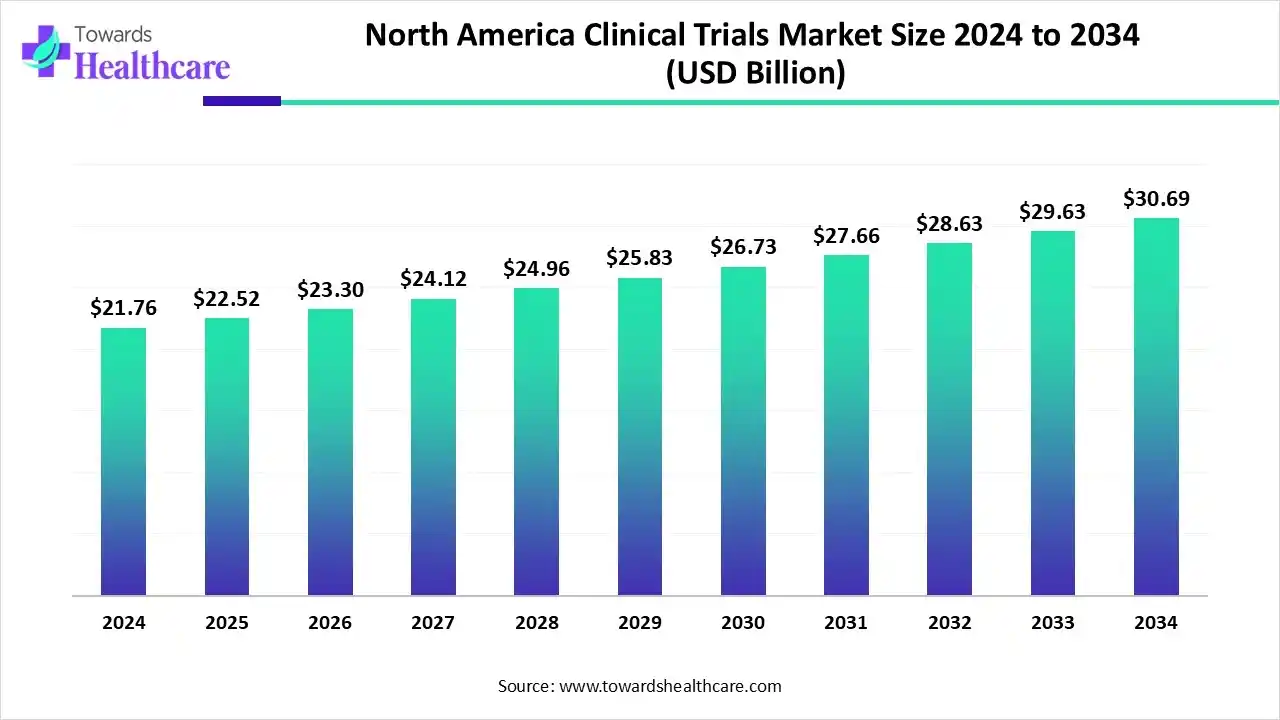
North America held the largest share of the clinical trials market in 2023. The increasing number of clinical trials, the presence of major pharmaceutical & biotechnology giants, technological advancements, and favorable scientific infrastructure drive the market. Clinical trials in the US are governed by the Food and Drug Administration. The US FDA ensures that clinical trials are designed, conducted, analyzed, and reported according to the laws. Health Canada is responsible for the conduct of clinical trials in Canada. The National Institute of Health invests most of its $48 billion budget in medical research for Americans. In August 2023, Project NextGen by the US Department of Health and Human Services granted $1 billion for COVID-19 vaccine clinical trials and $326 million for monoclonal antibodies.
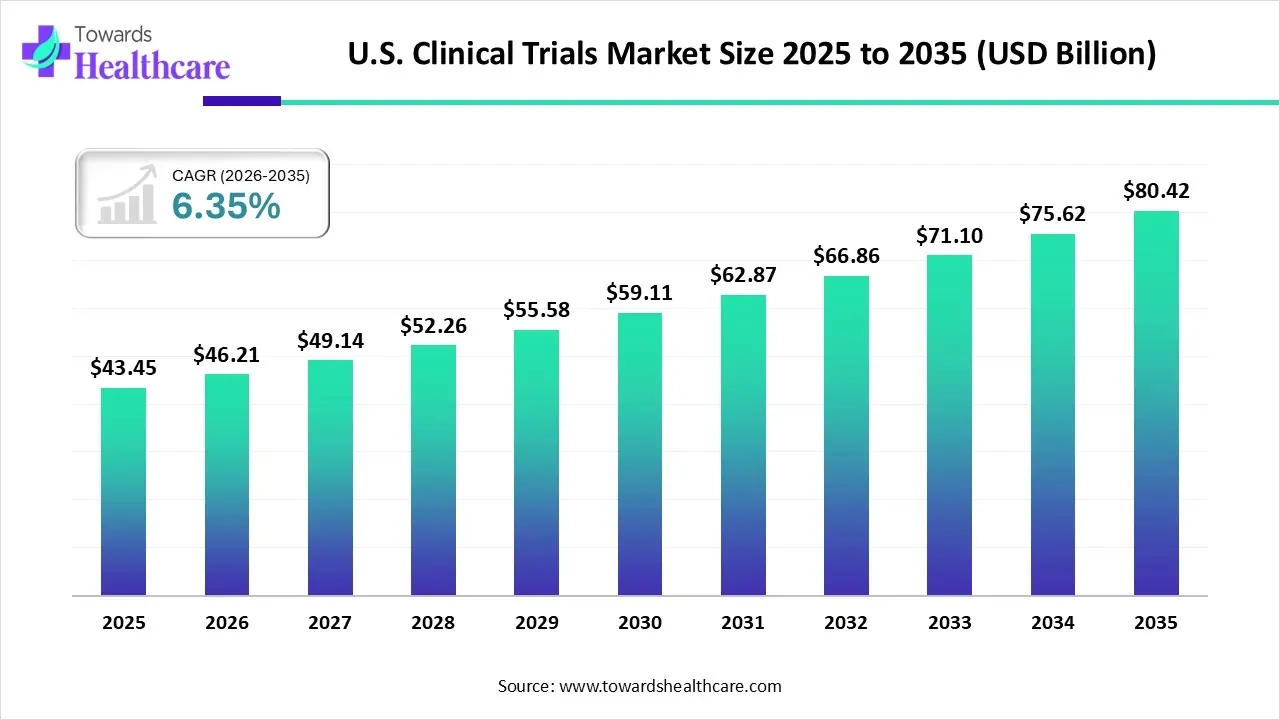
U.S. Clinical Trials Market Growth
The U.S. clinical trials market size is calculated at US$ 43.45 in 2025, grew to US$ 46.21 billion in 2026, and is projected to reach around US$ 80.42 billion by 2035. The market is expanding at a CAGR of 6.35% between 2026 and 2035.
Number of All Bio-Therapies in Clinical Trials, Per Million Population
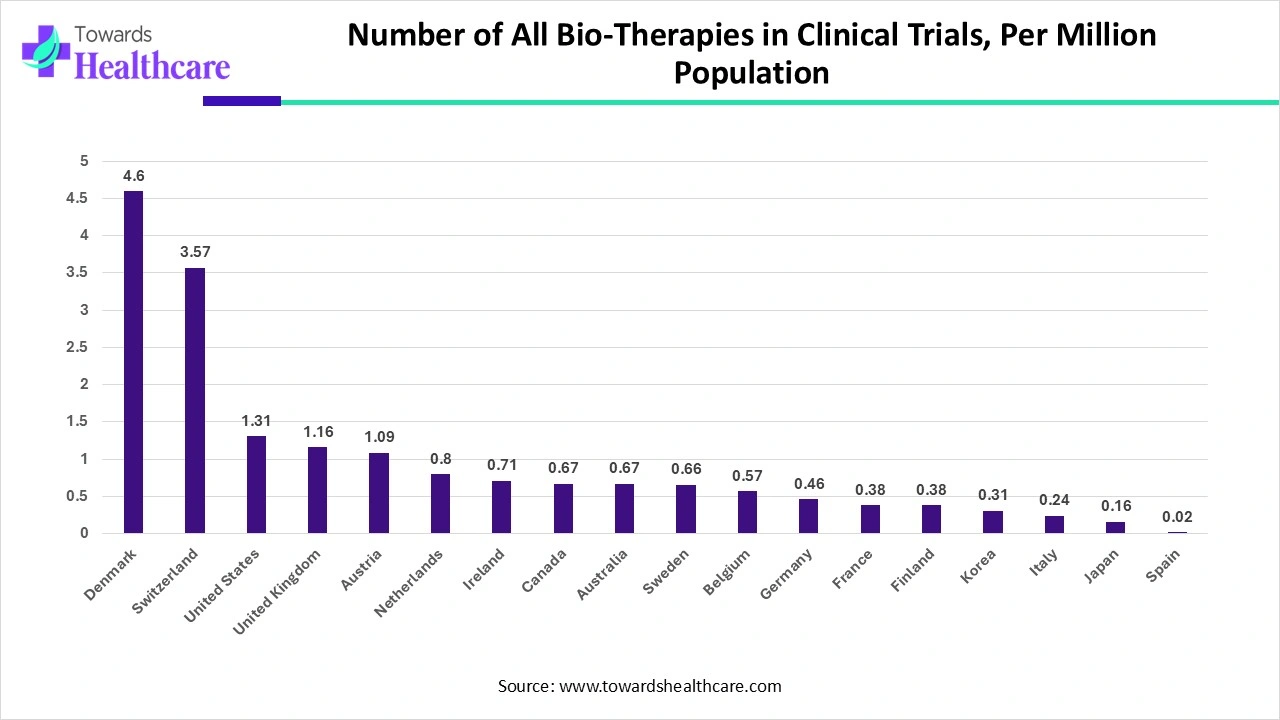
This table shows the number of bio-therapies being tested in clinical trials in various countries, normalized per million people in the population. Denmark leads by a large margin, with 4.60 bio-therapies per million, followed by Switzerland and the United States. Spain has the lowest number, with only 0.02 bio-therapies per million people.
Rising Number of Clinical Trials Drives Asia-Pacific
The APAC clinical trials market size was estimated at US$ 18.87 billion in 2025, projected to increase to US$ 20.42 billion in 2026 and reach US$ 41.33 billion by 2035, showing a healthy CAGR of 7.16% across the forecast years.
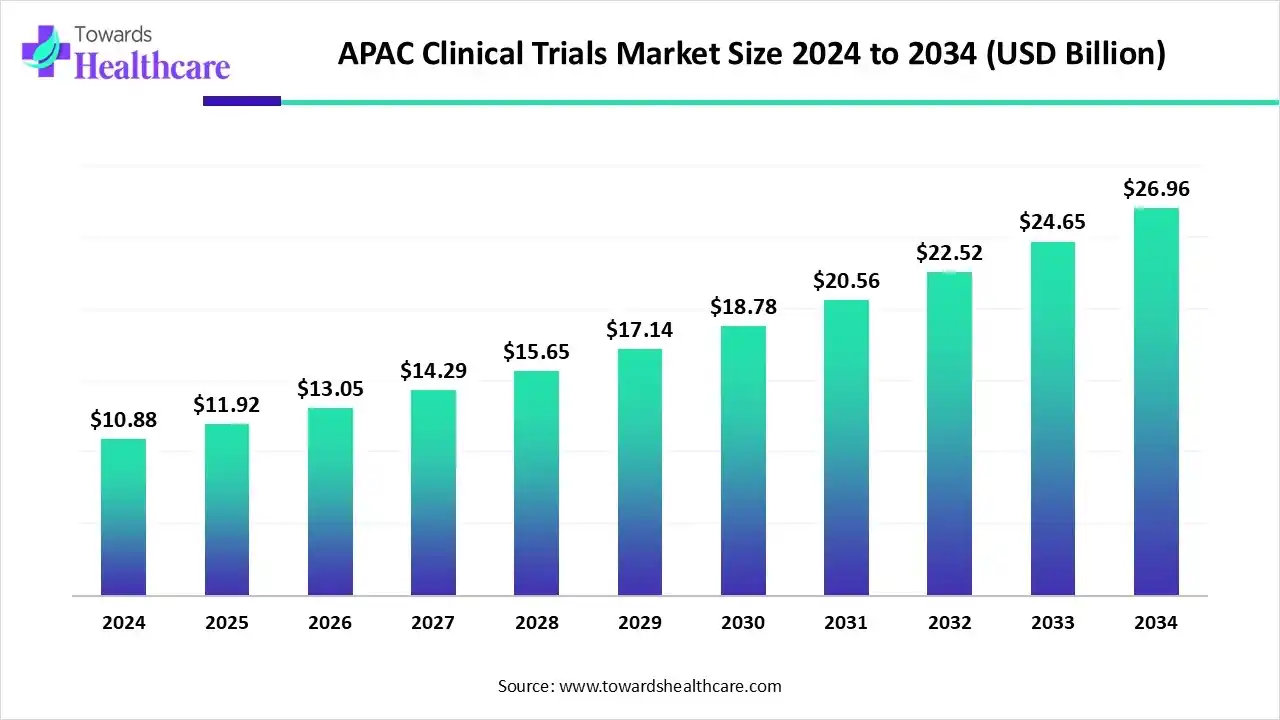
Asia-Pacific is anticipated to grow at the fastest rate in the market during the forecast period. The rising incidences of chronic disorders, expanding healthcare infrastructure, increasing number of clinical trials, and increasing investments & collaborations drive the market. Clinical trials are rapidly increasing in developing countries like India, Thailand, Vietnam, Philippines, etc. India reports a surge in phase 2 and 3 clinical trials from 2017 to 2023, growing from 15% to 18%. Similarly, in China, 4,300 clinical trial registrations were done in 2023, accounting for a 26.1% surge from 2022. China also reports an average consistent 16% growth in five-year clinical trials.
Number of Bio-Therapies and Experimental Bio-Therapies in Clinical Trials, December 2007
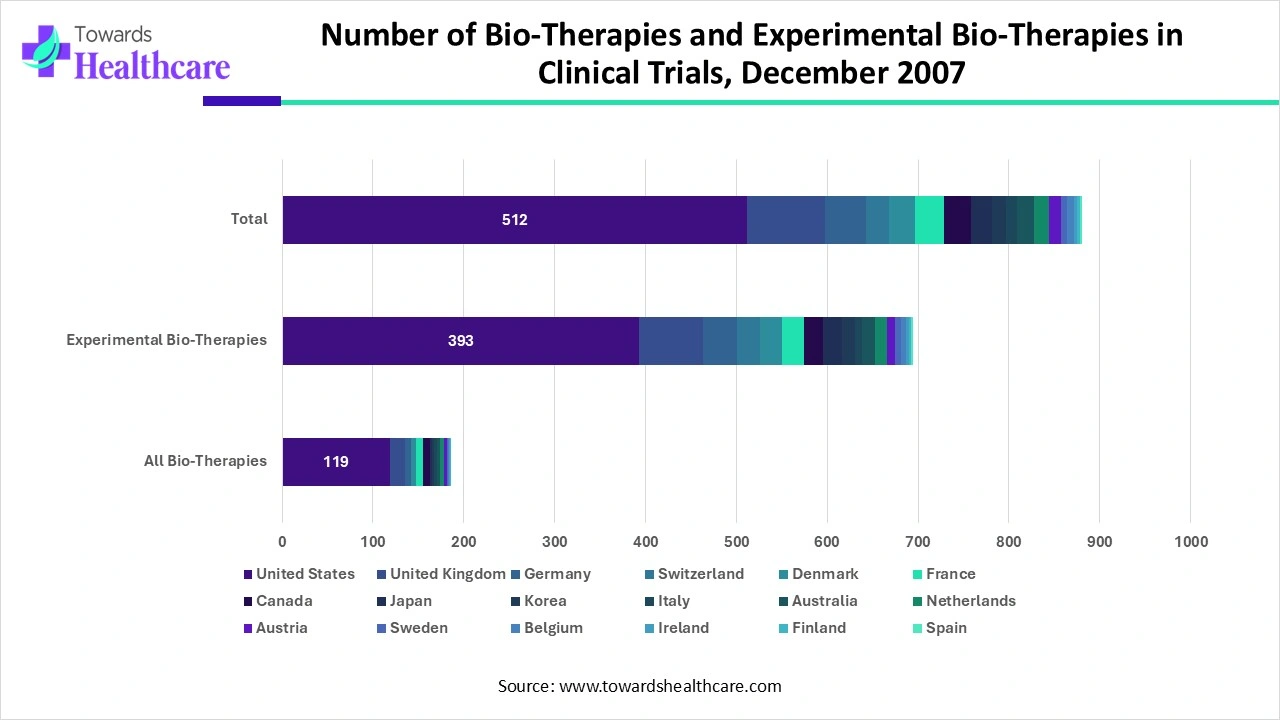
Oncology and Rare Diseases in Europe: Driving the Market
The Europe clinical trials market is valued at US$ 11.04 billion in 2025, increasing to US$ 11.65 billion in 2026, and is expected to reach approximately US$ 18.81 billion by 2035. This growth reflects a CAGR of 4.80% during the forecast period from 2026 to 2035.
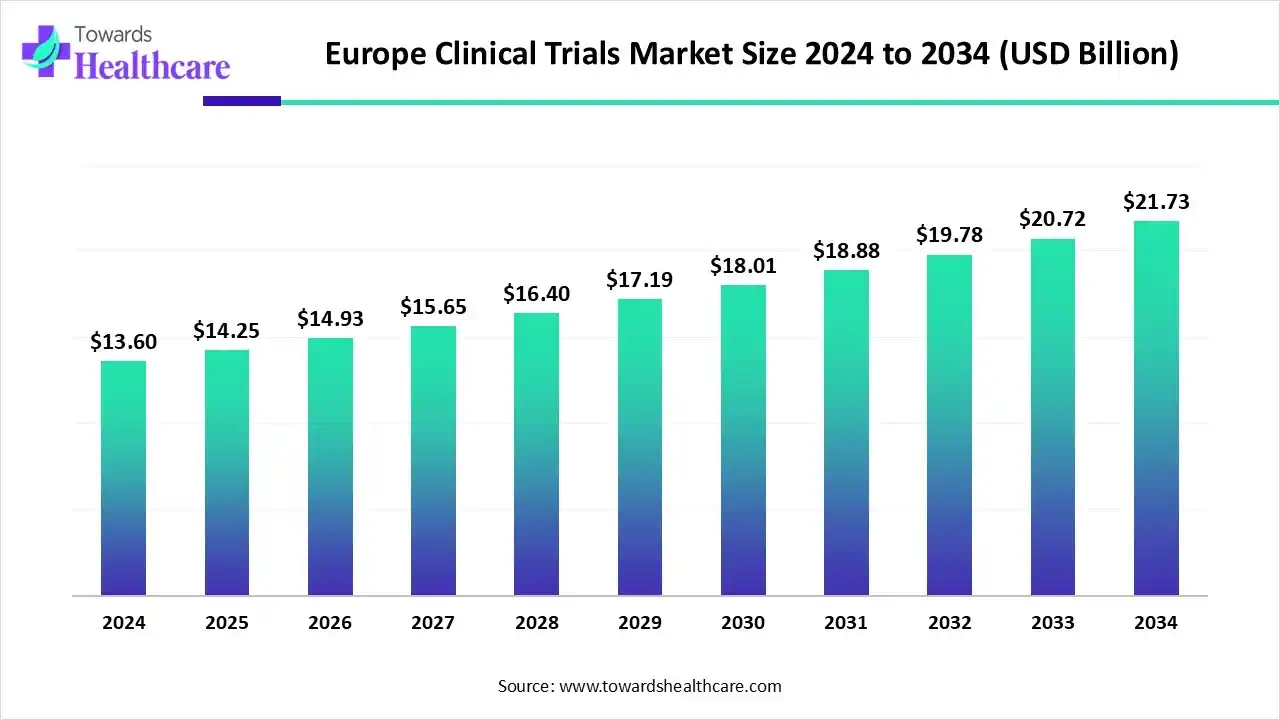
Europe is expected to grow at a notable rate in the foreseeable future in the clinical trials market because of its high-quality healthcare systems, diverse patient populations, and a strong regulatory framework. Europe, by offering access to specialized medical professionals, robust infrastructure, and systematic patient recruitment, facilitates efficient data collection. Oncology and rare diseases are conspicuously noticeable therapeutic areas for clinical trials in Europe, together with Central and Eastern Europe is rising as a preferred place for clinical trials owing to its increased and diverse patient populations, minimal costs, and clear regulatory frameworks. Furthermore, Europe is a major hub for both public and private funding of clinical research, contributing to its prominence in the global market.
Latin America Clinical Trials: PAHO Drives Growth, Ethics, and Global Collaboration
The Latin America clinical trials market is valued at US$ 4.67 billion in 2025 and is expected to grow to US$ 5.0 billion in 2026. It is projected to reach approximately US$ 9.17 billion by 2035, expanding at a compound annual growth rate (CAGR) of 6.30% from 2026 to 2035.
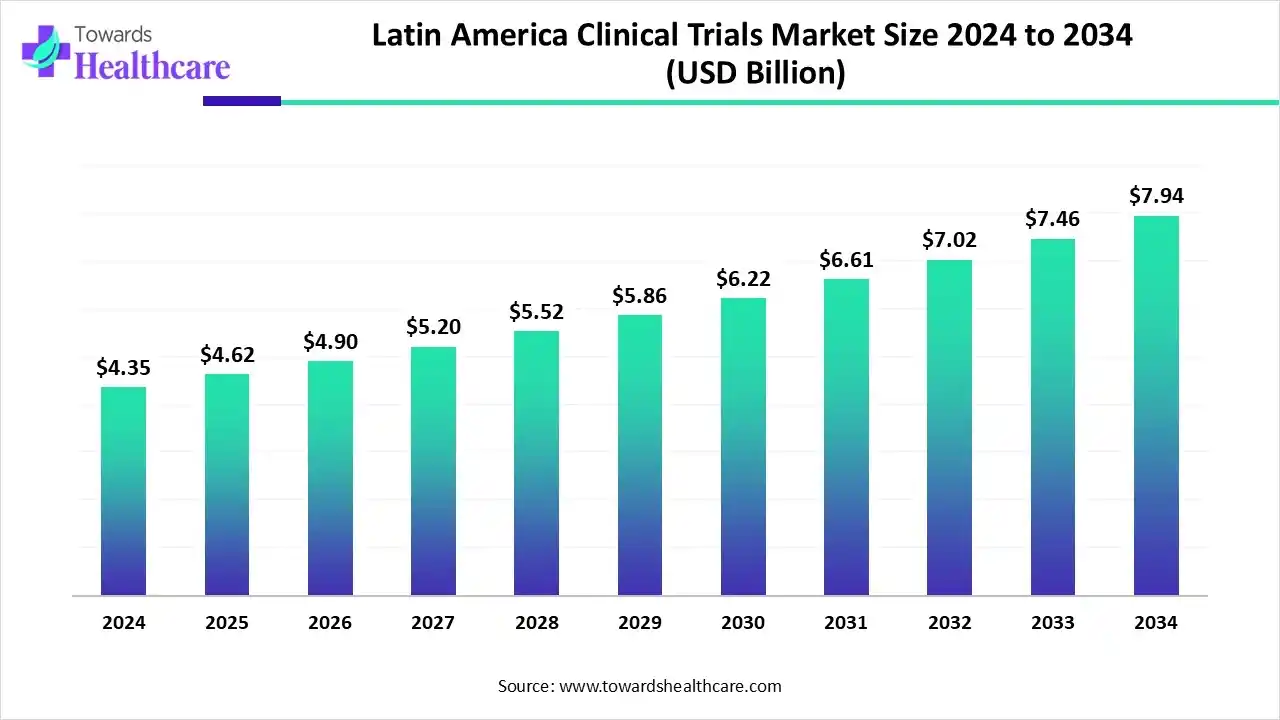
The Pan American Health Organization (PAHO) made efforts to expand the Latin America clinical trials market, improve clinical trials, offer high-quality results on health interventions, and improve research quality and coordination. The establishment of networks and collaboration supports the conduct of high-impact clinical trials and the expansion of research capacities. Advancing ethical and regulatory efficiency is of utmost importance to review and assess trials for their safety and effectiveness. The International Clinical Trials Registry Platform (ICTRP) of the World Health Organization (WHO) is a public platform that provides the required information on clinical trials conducted globally.
Middle East and Africa Clinical Trials Market Driven by Rising Chronic Diseases and Research Investments
The Middle East and Africa clinical trials market was valued at US$ 2.81 billion in 2025 and is expected to reach US$ 3.03 billion in 2026, progressing steadily to approximately US$ 5.79 billion by 2035. This growth reflects a compound annual growth rate (CAGR) of 6.80% over the forecast period.
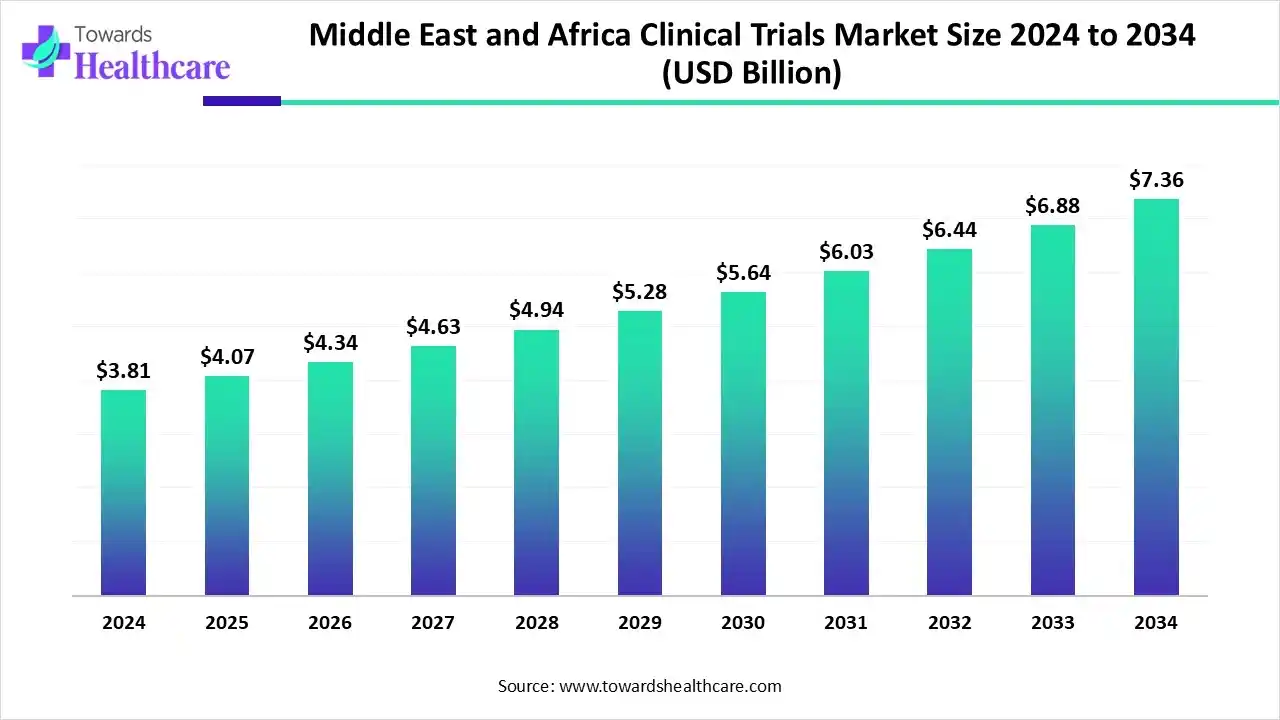
The Middle East and Africa clinical trials market is witnessing substantial growth, fueled by the increasing incidence of chronic and lifestyle-related diseases, expanding healthcare infrastructure, and favorable government policies. The region has become an attractive hub for global pharmaceutical and biotechnology companies, particularly due to South Africa’s strong presence offering advanced medical facilities, skilled clinical research professionals, and a diverse patient pool.
Solidarity Clinical Trials
The Solidarity Clinical Trials have used a randomized controlled trial design, which is considered the gold standard for evaluating the effectiveness of medical treatments. The use of this design has allowed for rigorous evaluation of different treatments for COVID-19. It is an international clinical trial launched by the WHO to find effective treatment against COVID-19. It included comparing four treatment options against the standard of care to evaluate their effectiveness against Coronavirus. Solidarity clinical trials were meant to discover if any of the drugs improve survival or slow down the progression of the disease. The trials had been conducted in a relatively short period of time, which has allowed for the rapid generation of evidence on the safety and efficacy of different treatments for COVID-19. This has been particularly important given the urgent need for effective treatments for the disease. During March, the funding for the Solidarity trial reached USD 108 million from 203,000 individual donations, governments, and charitable organizations with 45 countries involved in financing.
According to WHO, as of April 21st, 2020, around 100 countries participated in the trial and were working together to find effective therapeutics as early as possible. As per the WHO’s Director, by 18th March 2020, the ten countries that took part in the clinical trial are Thailand, Switzerland, Spain, South Africa, Norway, Iran, France, Canada, Bahrain, And Argentina.
Rapidly Evolving Clinical Trial Industry Over the Course of Two Years
There have been significant advancements in technology over the past two years that have impacted the clinical trial industry. For example, the use of artificial intelligence and machine learning algorithms for patient recruitment, data analysis, and personalized medicine is becoming more widespread. The use of artificial intelligence and machine learning algorithms has become increasingly popular in the industry. These technologies have enabled more efficient and effective patient recruitment, improved data analysis, and enabled personalized medicine. This has led to a more accurate and comprehensive analysis of trial data and improved patient outcomes. The COVID-19 pandemic has accelerated many of these changes and highlighted the importance of agility, flexibility, and patient-centricity in the clinical trial process. In addition, prominent market players have adopted several growth strategies in the clinical trials market in recent years.
Recent Updates
- In April 2023, the Melvin and Bren Simon Comprehensive Cancer Centre at Indiana University became the principal site for providing first-in-human clinical trials providing first-in-human clinical trials for patients with relapsed multiple myeloma. Patients who received higher doses of the immunotherapy known as REGN5459 had an overall response rate of 90.5 percent.
- In April 2023, at the American Association for Cancer Research (AACR) Annual Meeting 2023, researchers from The University of Texas MD Anderson Cancer Centre presented intriguing results from multiple clinical trials.
- From January 2022, the use of the new Clinical Trials Information System (CTIS) become compulsory in all EU and EEA countries. CTIS acts as a single point of contact for clinical trial data submission and evaluation.
Clinical Trials Market Companies
Major Market Segments Covered
By Phase
- Phase 1
- Phase 2
- Phase 3
- Phase 4
By Study Design
- Observational Study
- Interventional Study
- Expanded Access Study
By Indication
- Autoimmune/Inflammation
- Rheumatoid arthritis
- Multiple Sclerosis
- Osteoarthritis
- Irritable Bowel Syndrome (IBS)
- Others
- Pain Management
- Chronic Pain
- Acute Pain
- Oncology
- Blood Cancer
- Solid Tumors
- Other
- CNS Condition
- Epilepsy
- Parkinson's Disease (PD)
- Huntington's Disease
- Stroke
- Traumatic Brain Injury (TBI)
- Amyotrophic Lateral Sclerosis (ALS)
- Muscle Regeneration
- Others
- Diabetes
- Obesity
- Cardiovascular
- Others
By Geography
- North America
- Germany
- France
- United Kingdom
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- Southeast Asia
- Rest of Asia Pacific
- Latin America
- Brazil
- Rest of Latin America
- Middle East & Africa (MEA)
- GCC
- North Africa
- South Africa
- Rest of Middle East & Africa