Who are the iPSC Cell Therapies Market Top Vendors and What are Their Offerings?

FUJIFILM Cellular Dynamics (FCDI)
Headquarters: Madison, U.S.
iPSC Cell Therapies: Cardiology and neurology therapies
BlueRock Therapeutics (Bayer)
Headquarters: Boston, U.S.
iPSC Cell Therapies: Therapies for Parkinson’s disease, ophthalmology, and heart failure
Fate Therapeutics
Headquarters: San Diego, U.S.
iPSC Cell Therapies: Allogeneic NK CAR-T cell therapies
Cynata Therapeutics
Headquarters: Melbourne, Australia
iPSC Cell Therapies: Provides the Cymerus platform
Astellas Pharma (Ocata)
Headquarters: Tokyo, Japan
iPSC Cell Therapies: Regenerative ophthalmology via AIRM
Evotec SE
Headquarters: Hamburg, Germany
iPSC Cell Therapies: Provides an end-to-end iPSC platform
Lonza Group AG
Headquarters: Basel, Switzerland
iPSC Cell Therapies: Supports large-scale expansion and differentiation of iPSCs
REPROCELL Inc.
Headquarters: Yokohama, Japan
iPSC Cell Therapies: Provides StemRNA technology
Century Therapeutics
Headquarters: Philadelphia, U.S.
iPSC Cell Therapies: NK and T-cell therapies
Aspen Neuroscience
Headquarters: San Diego, U.S.
iPSC Cell Therapies: Personalized autologous iPSC-derived neuron replacement for Parkinson's disease
iPSC Cell Therapies Market Value Chain Analysis
R&D
- The R&D of the iPSC cell therapies focuses on the development of off-the-shelf, gene-edited allogeneic therapies.
- Key players: Fate Therapeutics, BlueRock Therapeutics, Century Therapeutics.
Clinical Trials and Regulatory Approvals
- The safety, functional tissue integration, genomic stability, tumorigenicity, and immunogenicity are evaluated in the clinical trials and regulatory approval of the iPSC cell therapies.
- Key players: Fate Therapeutics, BlueRock Therapeutics, Cynata Therapeutics.
Patient Support and Services
- Long-term safety monitoring, clinical trial navigation, and genetic counselling are provided in the patient support and services of the iPSC cell therapies.
- Key players: Fate Therapeutics, BlueRock Therapeutics, Aspen Neuroscience.
Growth Market
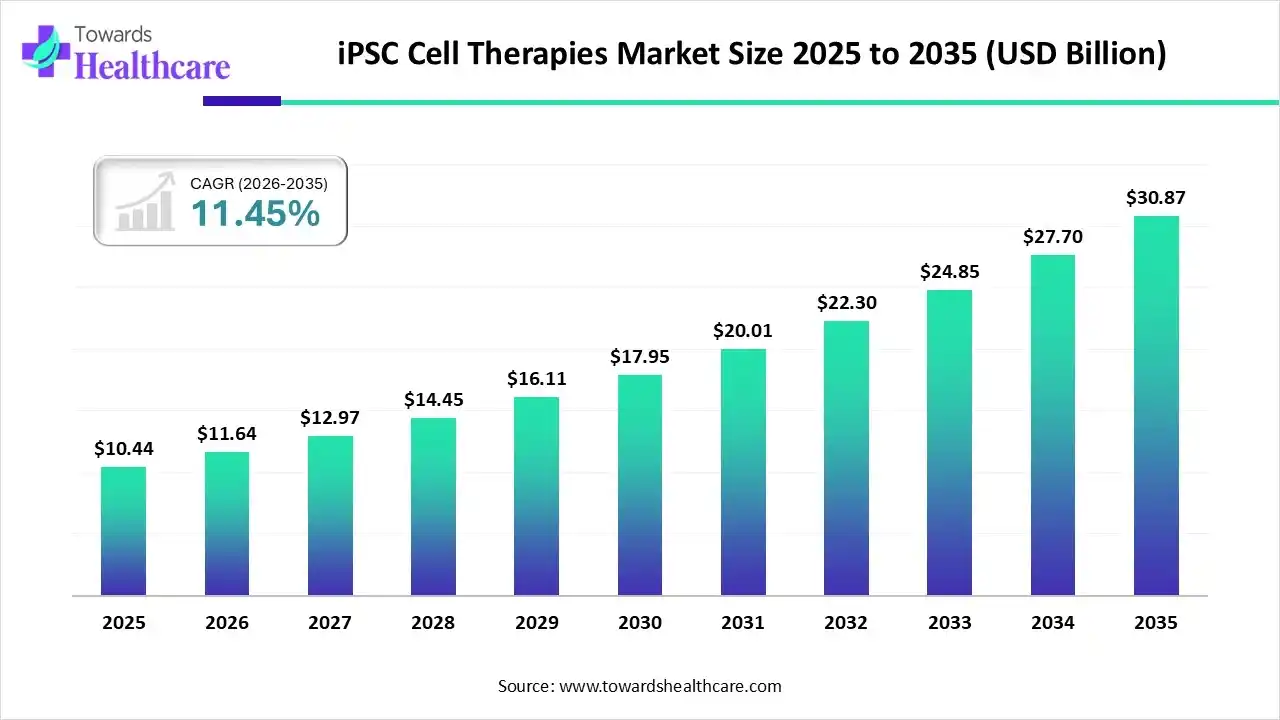
The global iPSC cell therapies market size was estimated at USD 10.44 billion in 2025 and is predicted to increase from USD 11.64 billion in 2026 to approximately USD 30.87 billion by 2035, expanding at a CAGR of 11.45% from 2026 to 2035.
What are the Recent Developments in the iPSC Cell Therapies Market?
- In May 2025, the off-the-shelf allogeneic iPSC-derived subtype-specific neural regenerative cell therapy developed for the treatment of spinal cord injury (SCI) by XellSmart Biopharmaceutical was announced to have received U.S. FDA and National Medical Products Administration (NMPA) of China approval for its Phase I clinical trial.
- In March 2025, the world’s first hospital-based autologous induced pluripotent stem cell (iPSC) foundry, utilizing an integrated, on-site facility to develop clinical-grade iPSCs and derivative therapies with the use of the Nebula platform, will be launched by the collaboration between Cellino and Mass General Brigham’s Gene and Cell Therapy Institute (GCTI).
Get easy access to iPSC cell therapy market insights, top vendors, and their therapies in one place: Access the dashboard
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
