Who are the Medical Radioactive Iodine I-131 Market Top Vendors and What are Their Offerings?

Company Profile
| Companies | Headquarters | Medical Radioactive Iodine I-131 Solutions |
| Jubilant Draxlmage | Quebec, Canada | HICON Sodium Iodine I-131 |
| NTP Radioisotopes | Pretoria, South Africa | Fission produced I-131 |
| IRE-IRE ELiT | Fleurus, Belgium | Pharmaceutical-grade I-131 solutions |
| Curium Pharma | London, UK | Sodium iodine I-131 capsules and oral solutions |
| POLATOM | Otwock, Poland | Sodium iodine I-131 capsules and oral solutions |
| Isotope JSC | Moscow, Russia | Bulk I-131 isotopes |
| Lantheus Medical Imaging | Massachusetts, U.S. | Therapeutic I-131 radiopharmceuticals |
| Nordion Inc. | Ontario, Canada | Medical grade I-131 isotopes |
| International Isotopes Inc. | Idaho, U.S. | Sodium iodine I-131 kits |
| Cardinal Health | Ohio, U.S. | Distribution and radiopharmacy services for I-131 |
Market Value Chain Analysis
R&D
- The R&D of the medical radioactive iodine I-131 focuses on restoring iodine uptake in cancer treatments and the development of targeted radioimmunotherapies.
- Key players: AstraZeneca, Eli Lilly, Actinium Pharmaceuticals.
Clinical Trials and Regulatory Approvals
- The thyroid remnant ablation rates, disease-free survival, radiation exposure level, bone marrow suppression, and salivary gland toxicity are evaluated in the clinical trials and regulatory approvals of the medical radioactive iodine I-131.
- Key players: Jubilant Draxlmage, Curium Pharma, AstraZeneca.
Patient Support and Services
- Comprehensive radiation safety training, financial assistance, and medication management are provided in the patient support and services of the medical radioactive iodine I-131.
- Key players: Jubilant Draxlmage, Cardinal Health, Curium Pharma.
Market Forecast
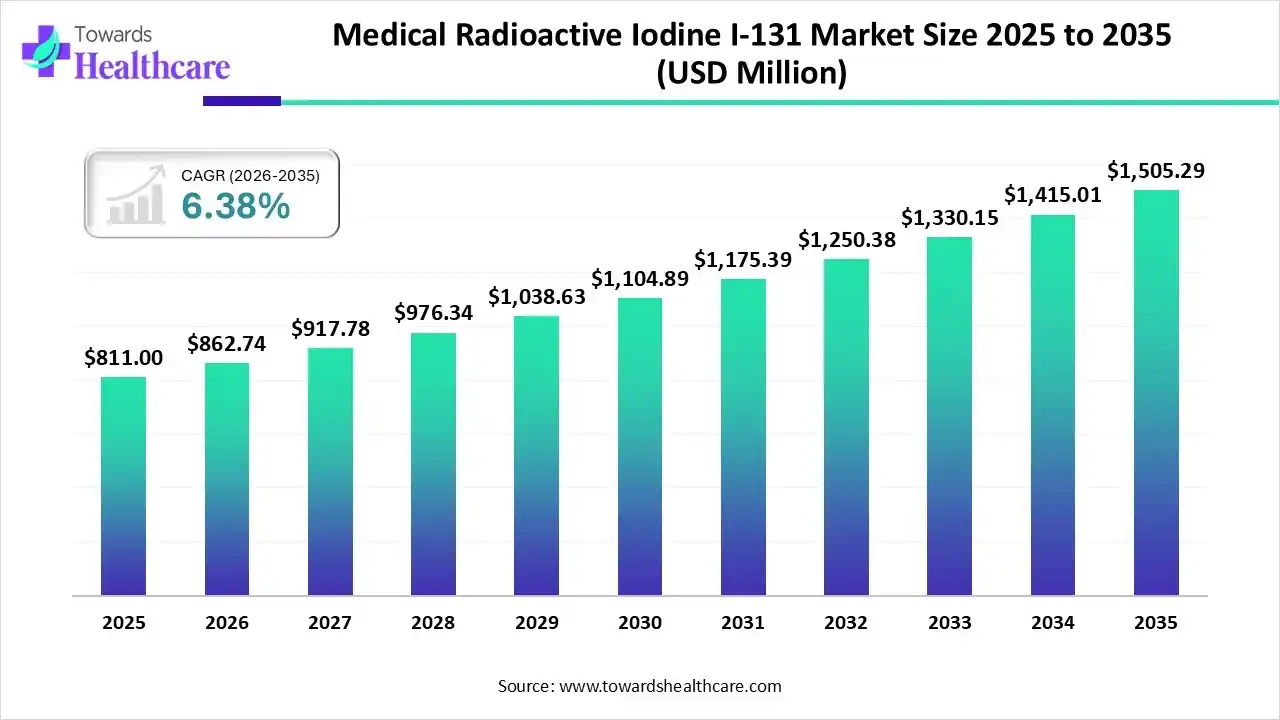
The global medical radioactive iodine I-131 market size was estimated at USD 811 million in 2025 and is predicted to increase from USD 862.74 million in 2026 to approximately USD 1505.29 million by 2035, expanding at a CAGR of 6.38% from 2026 to 2035.
What are the Recent Developments in the Market?
- In October 2025, the initiation of Phase II clinical trials of the combination of selpercatinib followed by 131I therapy for the treatment of RET fusion differentiated thyroid cancer was announced, where its efficacy and safety will be evaluated.
- In January 2025, to enhance the availability of the Iodine-131 diagnostic capsules to patients in the Nordic Region and in other European countries, a collaboration between Curium and Institute of Isotopes Co. Ltd. (Izotop) was announced.
Looking for a simple way to track medical radioactive iodine I-131 market reports? - Access the dashboard
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
