Meningococcal Vaccines Market Top Vendors and Their Offerings

From stats to strategies, see the whole Meningococcal Vaccines Market story; click to access
| Companies | Headquarters | Meningococcal Vaccines |
| GSK (GlaxoSmithKline) | London, UK | Penmenvy, Menveo, and Bexsero |
| Pfizer | New York, U.S. | Penbraya, Nimenrix, and Trumenba |
| Sanofi | Paris, France | MenQuadfi and Menactra |
| Serum Institute of India | Pune, India | MenFive and MenAfriVac |
| Merck & Co. (MSD) | New Jersey, U.S. | Mencevax |
| Walvax Biotechnology | Kunming, China | MenMCV4 and MenMCV2 |
| Bio-Med Pvt. Ltd. | Ghaziabad, India | Meningococcal Polysaccharide |
| CanSino Biologics (CanSinoBIO) | Tianjin, China | Menphencia and Menhycia |
| Incepta Vaccine Ltd. | Dhaka, Bangladesh | Incepta Meningococcal Vaccine |
| Hilleman Laboratories | Singapore | Low-cost MenACYWX Conjugate Vaccine |
Meningococcal Vaccines Market Value Chain Analysis
R&D
- The R&D of the meningococcal vaccines focuses on the development of pentavalent (MenABCWY) combination vaccines and affordable multivalent conjugate vaccines.
- Key players: GSK, Pfizer, Serum Institute of India.
Clinical Trials and Regulatory Approvals
- The safety and immunogenicity are evaluated in the clinical trials and regulatory approvals of the meningococcal vaccines.
- Key players: GSK, Pfizer, Sanofi.
Patient Support and Services
- The educational resources and financial assistance programs are provided in the patient support and services of the meningococcal vaccines.
- Key players: GSK, Pfizer, Sanofi.
Market Growth
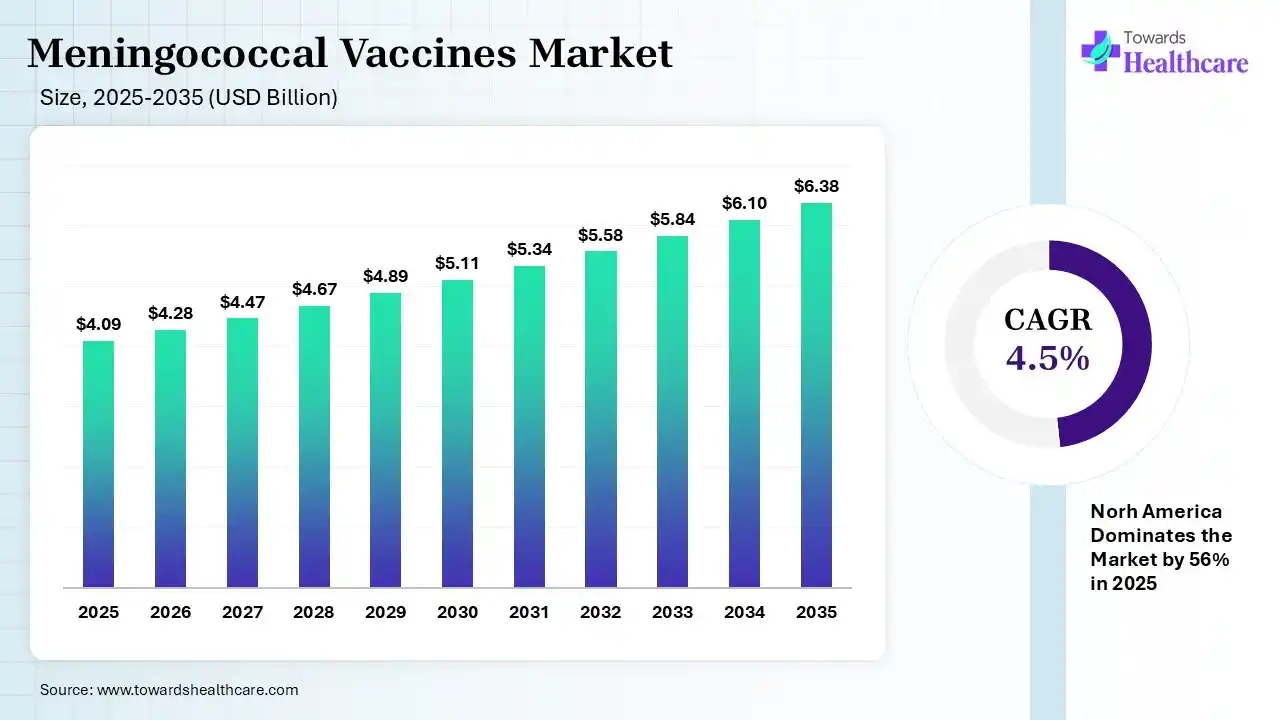
The global meningococcal vaccines market size was estimated at USD 4.09 billion in 2025 and is predicted to increase from USD 4.28 billion in 2026 to approximately USD 6.38 billion by 2035, expanding at a CAGR of 4.5% from 2026 to 2035.
What are the Recent Developments in the Meningococcal Vaccines Market?
- In April 2025, Penbraya, a combination of Trumenba targeting meningococcal group B and Nimenrix targeting meningococcal groups A, C, W-135, and Y, was announced to have received FDA approval against these five meningococcal serogroups, leading to invasive meningococcal disease (IMD), which was announced by Pfizer Inc.
- In March 2026, the "Drug Clinical Trial Approval Notice" issued by the National Medical Products Administration was received by Chengdu Kanghua Biological Products Co., Ltd. for its ACYW135 group meningococcal conjugate vaccine, authorizing the launch of clinical trials against epidemic cerebrospinal meningitis/meningococcal, caused by meningococcal groups A, C, Y, and W135.
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
