May 2026

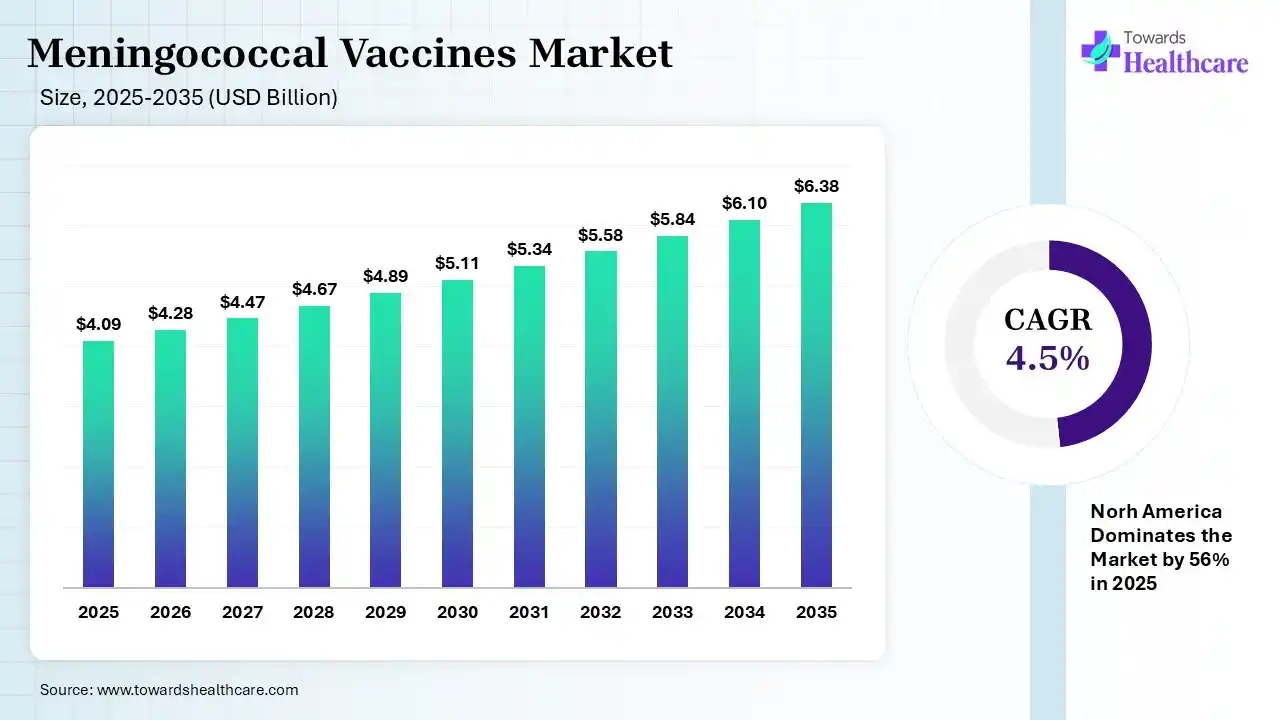
The global meningococcal vaccines market size was estimated at USD 4.09 billion in 2025 and is predicted to increase from USD 4.28 billion in 2026 to approximately USD 6.38 billion by 2035, expanding at a CAGR of 4.5% from 2026 to 2035.
The rise in the outbreaks of meningitis cases is increasing the adoption of the meningococcal vaccines globally. The growing R&D activities, government initiatives, technological advancements, and new vaccine launches are also enhancing the market growth.
The meningococcal vaccines market is driven by rising meningococcal disease incidences, growing government immunization initiatives, and expanding technological advancements. The meningococcal vaccines encompass vaccines developed to provide protection against Neisseria meningitidis, which is a bacterium responsible for meningitis and septicaemia. These vaccines help in preventing meningitis, septicaemia, their outbreaks, and protect the high-risk population, preventing further complications.
The use of AI in the development of meningococcal vaccines is increasing as it helps in predicting the target protein and antigen, promoting the development of optimized vaccines. It also helps in detecting adverse effects by determining the risk and offering safety monitoring, where it also provides the development of personalized vaccination strategies by identifying high risk population. It also helps in clinical trials optimization and forecasting outbreaks.
Increased Disease Awareness
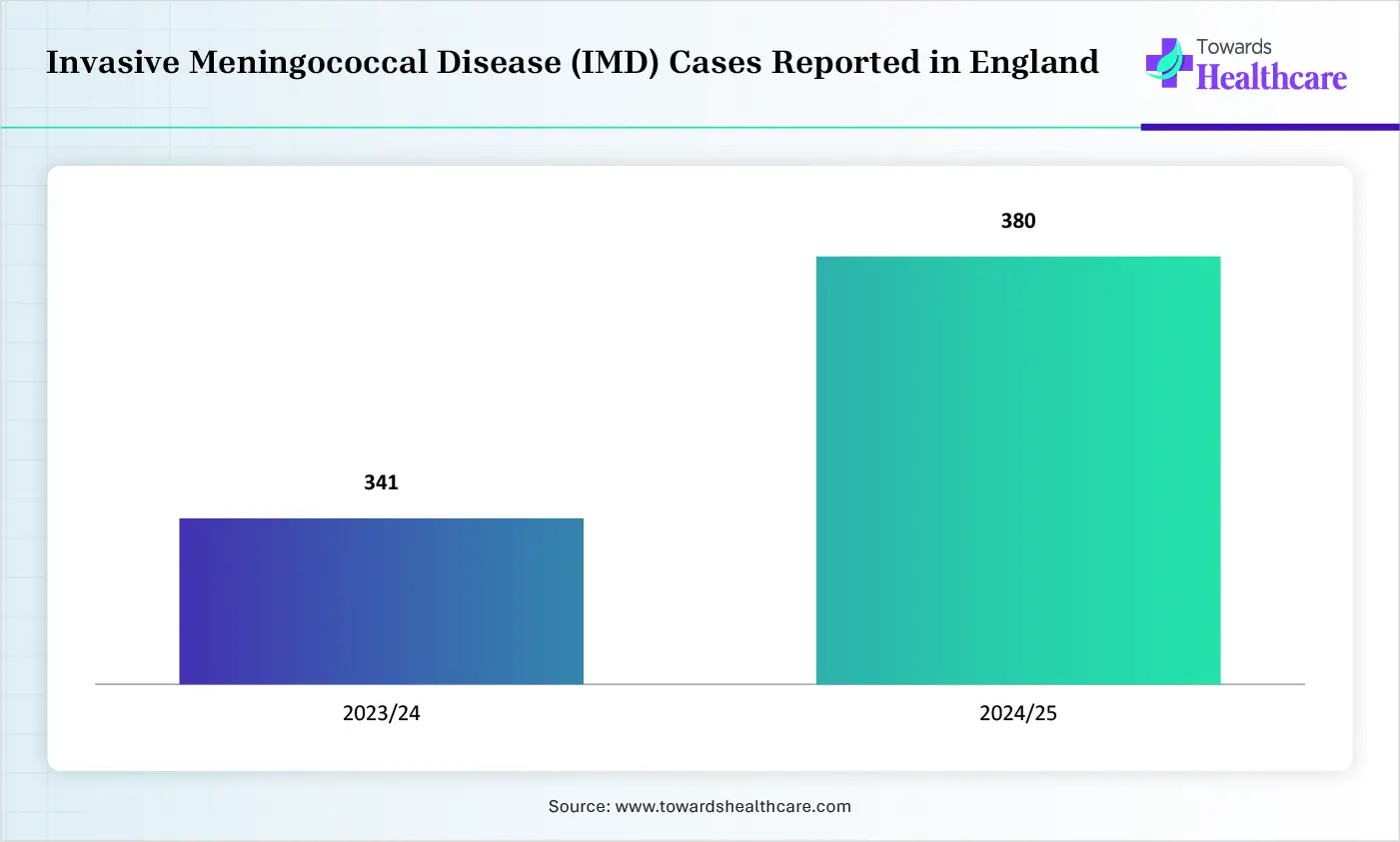
The growing educational campaign and increasing meningitis outbreaks are increasing health awareness, driving the demand and adoption of the meningococcal vaccines, as well as promoting the development of new vaccines.
Growing Infant Immunization
Increasing vaccination programs, growing regulatory approvals of the meningococcal vaccines, and government immunization campaigns are also increasing their use to provide long term protection to the infants.
Expanding Innovation
The companies are developing various multivalent vaccines, where the growing R&D activities are also increasing the development of next-generation vaccines and new delivery systems to improve vaccine efficacy.
| Table | Scope |
| Market Size in 2026 | USD 4.28 Billion |
| Projected Market Size in 2035 | USD 6.38 Billion |
| CAGR (2026 - 2035) | 4.5% |
| Leading Region | North America by 56% |
| Historical Data | 2020 - 2023 |
| Base Year | 2025 |
| Forecast Period | 2026 - 2035 |
| Measurable Values | USD Millions/Units/Volume |
| Market Segmentation | By Type, By Brand, By Serotype, By Age Group, By Sales Channel, By Region |
| Top Key Players | GSK (GlaxoSmithKline), Pfizer, Sanofi, Serum Institute of India, Merck & Co. (MSD), Walvax Biotechnology, Bio-Med Pvt. Ltd., CanSino Biologics (CanSinoBIO), Incepta Vaccine Ltd., Hilleman Laboratories |
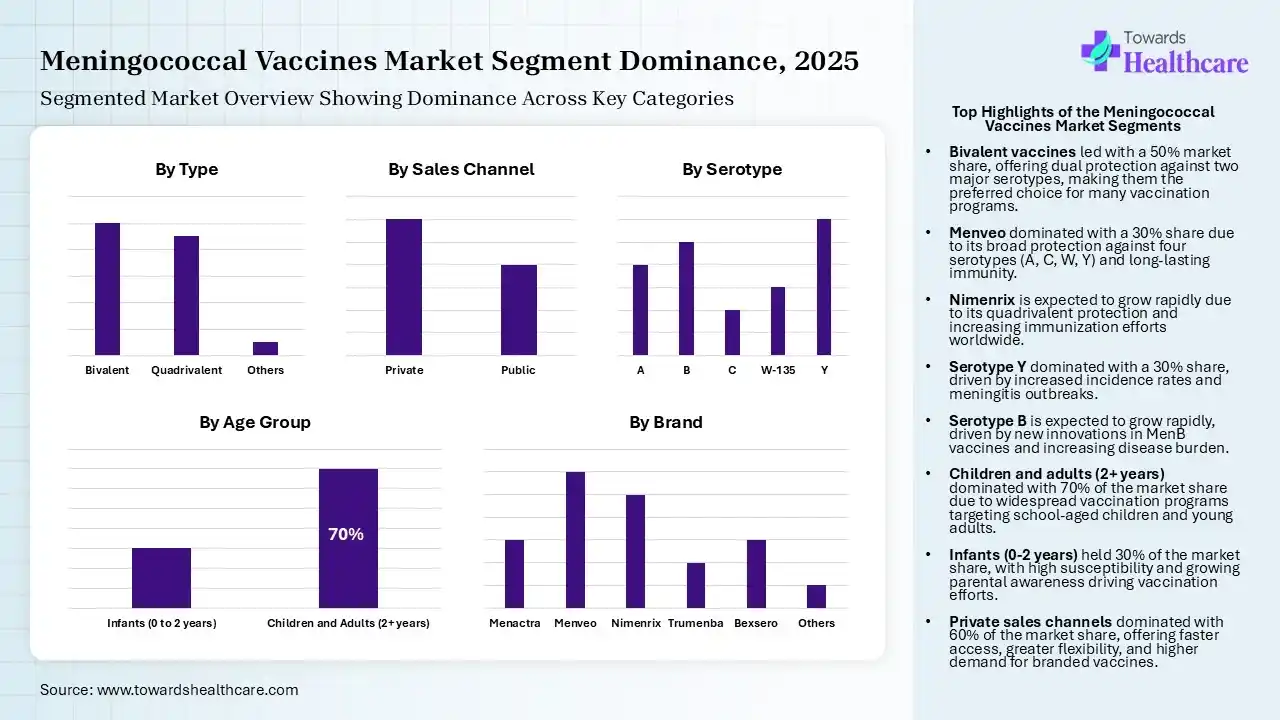
Why Did the Bivalent Segment Dominate in the Meningococcal Vaccines Market in 2025?
| Segment | Share 2025 (%) |
| Bivalent | 50% |
| Quadrivalent | 45% |
| Others | 5% |
Explanaion
The bivalent segment led the market by 50% in 2025 and is expected to show the highest growth over the forecast period, driven by its increased use against serotypes A and C. Growth in vaccination programs also increased its use and production rates, and its affordability also enhanced adoption rates.
How Menveo Segment Dominated the Meningococcal Vaccines Market in 2025?
| Segment | Share 2025 (%) |
| Menactra | 15% |
| Menveo | 30% |
| Nimenrix | 25% |
| Trumenba | 10% |
| Bexsero | 15% |
| Others | 5% |
Explanation
The Menveo segment held the largest revenue share of the market by 15% in 2025, due to its ability to target four different serotypes, that is, A, C, W, and Y. Its long-lasting immunity also increased its use, where its good tolerability and safety profile also increased its use, which promoted its production rates.
Nimenrix
The Nimenrix segment is expected to grow at the fastest CAGR during the predicted time, driven by its quadrivalent action against A, C, W, and Y serotypes. The growing immunization programs, disease outbreaks, government support, and increasing health awareness are also driving their demand.
Which Serotype Type Segment Held the Dominating Share of the Meningococcal Vaccines Market in 2025?
| Segment | Share 2025 (%) |
| A | 20% |
| B | 25% |
| C | 10% |
| W-135 | 15% |
| Y | 30% |
Explanation
The Y segment held the dominating share of the market by 20% in 2025, due to growth in their incidence rates, which increased the demand for their effective vaccinations. The growth in the immunization programs also recommended the use of ACWY vaccines such as Menveo and Nimenrix to control their outbreaks.
B
The B segment is expected to gain the highest share during the upcoming years, due to growing innovations of MenB vaccines and increasing disease burden. This is increasing the adoption of various meningococcal vaccines, where the growing health awareness, regulatory approvals, and vaccinations are also increasing their demand.
What Made Children and Adults (2 years and above) the Dominant Segment in the Meningococcal Vaccines Market in 2025?
| Segmnet | Share 2025 (%) |
| Infants (0 to 2 years) | 30% |
| Children and Adults (2+ years) | 70% |
Explanation
The children and adults (2 years and above) segment contributed the biggest revenue share of the market by 30% in 2025, due to growth in the incidence of invasive meningococcal disease (IMD) in children. This increased the use of meningococcal vaccines in children and adults to reduce the outbreak risk, where these vaccinations were also supported by the government, schools, and colleges.
Infants (0 to 2 years)
The infants (0 to 2 years) segment is expected to witness the fastest growth during the upcoming years, due to their high susceptibility and growing parental awareness. This is increasing the use of various meningococcal vaccines for routine immunization to provide protection and prevention of outbreaks.
Why the Private Segment Dominated the Meningococcal Vaccines Market?
| Segment | Share 2025 (%) |
| Private | 60% |
| Public | 40% |
Explanation
The private segment accounted for the highest revenue share of the market by 60% in 2025 and is expected to expand rapidly during the upcoming years, driven by its affordability and dispensing of high-quality or branded vaccines. Moreover, the presence of a wide range of vaccines and faster access also increased their use.
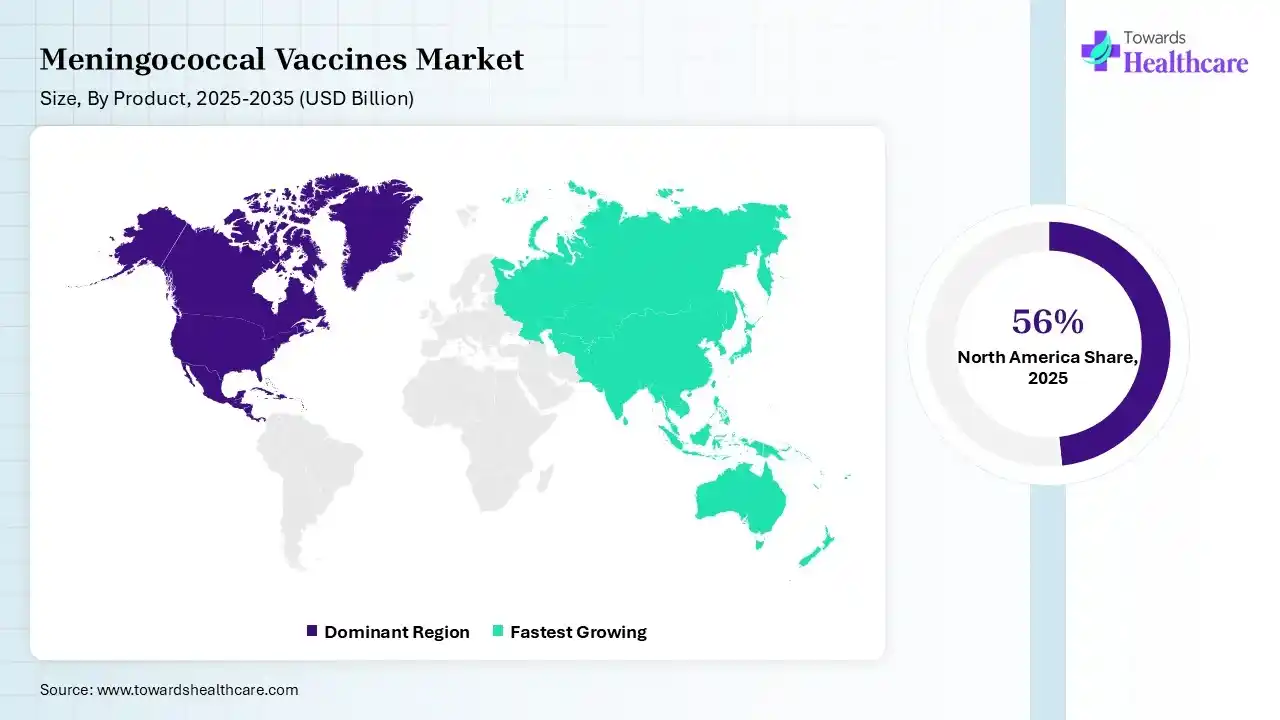
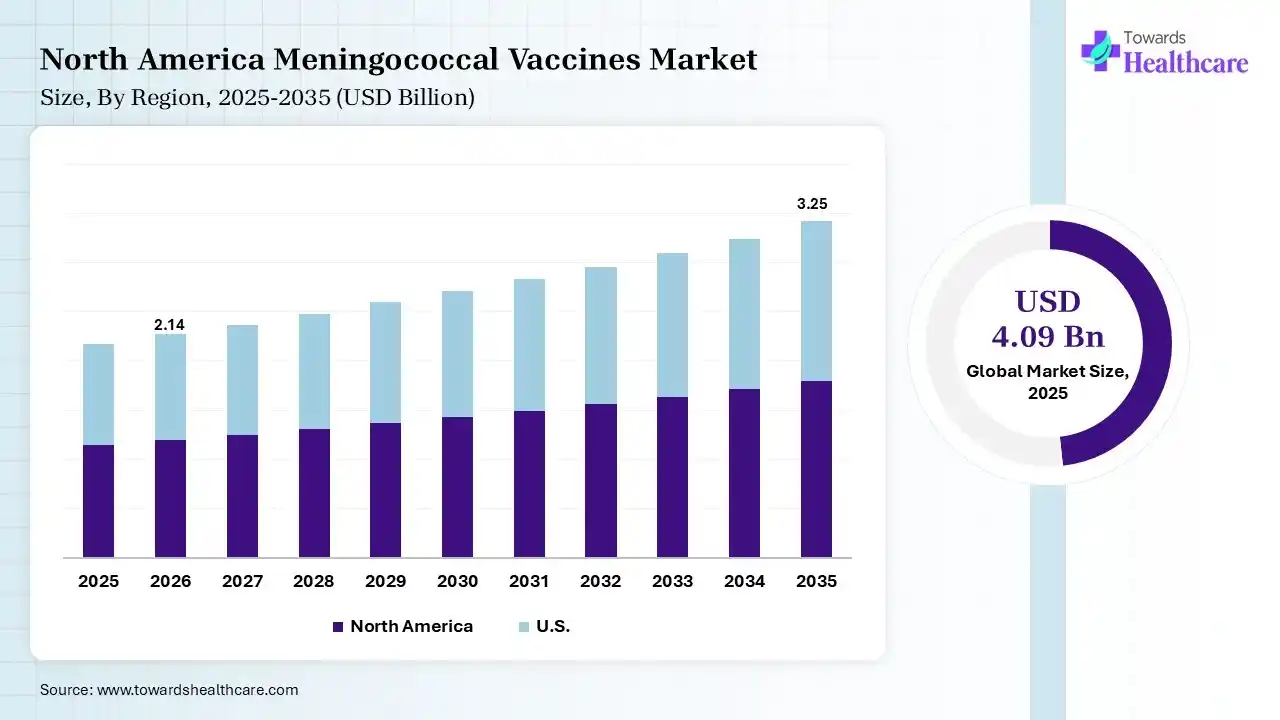
North America dominated the meningococcal vaccines market by 56% in 2025, due to the growth in disease awareness, where the growth in healthcare spending also increased their production rates. The presence of the advanced healthcare sector also increased its availability, where the rise in immunization programs also contributed to the market growth.
U.S. Market Trends
The growing health campaign in the U.S. is increasing meningococcal awareness, driving the use of meningococcal vaccines. The growing insurance coverage and outbreaks of the disease are also increasing the vaccination program, promoting their use, where the growing vaccine approvals are also creating new opportunities for their advancements.
Asia Pacific is expected to host the fastest-growing meningococcal vaccines market during the forecast period, due to expanding immunization programs and government initiatives. The rapid urbanization and increasing disease awareness are also increasing the development and adoption of meningococcal vaccines, enhancing the market growth.
India Market Trends
The expanding immunization programs in India are increasing the development and launch of new meningococcal vaccines, where the growing disease awareness is also increasing their demand. Additionally, growing healthcare investments and expanding vaccine manufacturing capabilities are also enhancing their innovations.
Europe is expected to grow significantly in the meningococcal vaccines market during the forecast period, due to the growing focus on outbreak preparedness and the presence of well-established companies, which are increasing the adoption of various meningococcal vaccines. Growing immunization programs are also promoting the market growth.
UK Market Trends
The growing disease awareness due to expanding immunization programs and educational campaigns across the UK are increasing the adoption of the meningococcal vaccines. The growing disease outbreaks, R&D activities, and government funding are also propelling the development of new vaccines.
R&D
Clinical Trials and Regulatory Approvals
Patient Support and Services

| Companies | Headquarters | Meningococcal Vaccines |
| GSK (GlaxoSmithKline) | London, UK | Penmenvy, Menveo, and Bexsero |
| Pfizer | New York, U.S. | Penbraya, Nimenrix, and Trumenba |
| Sanofi | Paris, France | MenQuadfi and Menactra |
| Serum Institute of India | Pune, India | MenFive and MenAfriVac |
| Merck & Co. (MSD) | New Jersey, U.S. | Mencevax |
| Walvax Biotechnology | Kunming, China | MenMCV4 and MenMCV2 |
| Bio-Med Pvt. Ltd. | Ghaziabad, India | Meningococcal Polysaccharide |
| CanSino Biologics (CanSinoBIO) | Tianjin, China | Menphencia and Menhycia |
| Incepta Vaccine Ltd. | Dhaka, Bangladesh | Incepta Meningococcal Vaccine |
| Hilleman Laboratories | Singapore | Low-cost MenACYWX Conjugate Vaccine |
Strengths
Weaknesses
Opportunities
Threats
By Type
By Brand
By Serotye
By Age Group
By Sales Channel
By Region
May 2026
May 2026
May 2026
May 2026