Latest Updates of Companies in the Metastatic Breast Cancer Market
| Company | Headquarters | Latest Update |
| Roche Holding AG | United States | In December 2025, Roche announced today that the U.S. Food and Drug Administration (FDA) has approved additional indications for its PATHWAY anti-HER2/neu (4B5) Rabbit Monoclonal Primary Antibody and VENTANA HER2 Dual ISH DNA Probe Cocktail tests. |
| Pfizer Inc. | United States | In March 2026, Pfizer Inc. announced positive topline results from the randomized Phase 2 FOURLIGHT-1 study assessing atirmociclib in combination with fulvestrant, versus fulvestrant or everolimus plus exemestane, in people with hormone receptor (HR)-positive, human epidermal growth factor receptor 2-negative (HER2-) advanced or metastatic breast cancer. |
| Eli Lilly and Company | United States | Eli Lilly and Company attained significant milestones in metastatic breast cancer (MBC) management, highlighted by the FDA approval of Inquiry. |
| AstraZeneca | United Kingdom | Silence Therapeutics intends to advance siRNAs to address unmet patient needs in a broad range of diseases with a genetic basis. |
| Novartis AG | United Kingdom | In December 2025, AstraZeneca Pharma India Ltd. (AZPIL), a worldwide, science-based biopharmaceutical company, announced that the Central Drugs Standard Control Organisation (CDSCO) had granted regulatory approval to import, sell, and distribute Datopotamab Deruxtecan in India |
Recent Developments in the Metastatic Breast Cancer Market
- In March 2026, Pfizer Inc. announced positive topline results from the randomized Phase 2 FOURLIGHT-1 study evaluating atirmociclib in combination with fulvestrant, versus fulvestrant or everolimus plus exemestane, in patients with hormone receptor-positive, human epidermal growth factor receptor 2-negative (HER2-) advanced or metastatic breast cancer who had received prior cyclin-dependent kinase (CDK) 4/6 inhibitor-based treatment.
- In December 2025, AstraZeneca and Daiichi Sankyo’s Enhertu, in combination with pertuzumab, has been approved in the US for the 1st-line management of adult patients with unresectable or metastatic HER2-positive breast cancer, as determined by a Food and Drug Administration (FDA)-driven test.
Supply Chain Analysis
R&D:
- Research and development (R&D) in RNA interference therapy includes molecular profiling and genomic evolution research, liquid biopsies and patient-based organoids, targeted therapy advancement, pre-metastatic niche and microenvironment studies, clinical trials, and real-world evidence.
- Key Players: Novartis and Sanofi
Manufacturing Processes:
- Manufacturing processes for RNA interference therapy involve epithelial-mesenchymal transition (EMT), intravasation, circulation and survival, pre-metastatic niche creation, extravasation and colonization, and metabolic reprogramming
- Key Players: Eli Lilly and AstraZeneca
Patient Services:
- Patient services involve palliative care, pain management, psychosocial support, nutritional counselling, integrative therapies, and patient navigation to clinical trials.
- Key Players: Roche and Pfizer
Market Growth
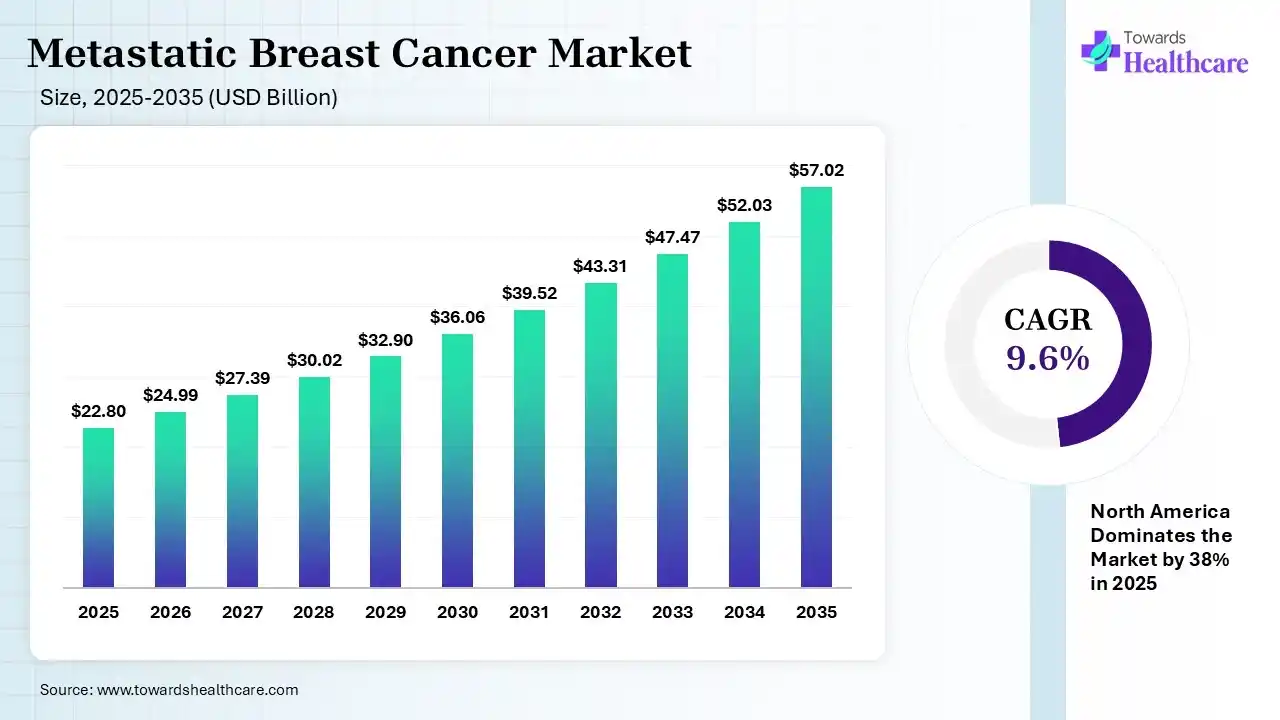
The global metastatic breast cancer market size was estimated at USD 22.8 billion in 2025 and is predicted to increase from USD 24.99 billion in 2026 to approximately USD 57.02 billion by 2035, expanding at a CAGR of 9.6% from 2026 to 2035. The metastatic breast cancer market is growing because metastatic breast cancer is a recurrent cancer, meaning it’s a cancer that came back after treatment and is affecting tissue and organs located far from the original breast cancer.
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
