Top Vendors in the Microbial CDMO Market & Their Offering
| Companies | Headquarters | Offerings |
| Lonza Group AG | Basel, Switzerland | Provides development and manufacturing services, including microbial expression systems and scalable GMP production, as part of its biologics CDMO portfolio |
| Fujifilm Diosynth Biotechnologies | Morrisville, North Carolina | Global CDMO that develops and manufactures biopharmaceuticals, including microbial-derived products, process development, and fillfinish services. |
| AGC Biologics | Washington, USA | Offers microbial fermentation development and GMP manufacturing, including protein expression, plasmid DNA, and other microbial biologics services. |
| Thermo Fisher Scientific | Massachusetts, USA | Through its Patheon brand, delivers microbial and biologics development, formulation, and manufacturing services across clinical and commercial stages. |
| Boehringer Ingelheim | Ingelheim, Germany | Offers contract development and manufacturing for biopharmaceuticals, including microbial and yeast fermentation production at global sites like Shanghai |
| WuXi Biologics | Jiangsu, China | A global CRDMO providing microbial and other biologics manufacturing, with facilities in China, Europe, and the U.S., supporting recombinant proteins, plasmid DNA, vaccines, etc |
Value Chain Analysis
R&D
- Microbial CDMOs focus on R&D to optimize bacteria, yeast, and fungi cultivation for recombinant proteins, vaccines, plasmid DNA, and metabolites. They enhance yield, strain stability, and scale processes from feasibility to GMP production.
- Key players: Catalent, Lonza, WuXi, and Samsung Biologics.
Clinical Trials
- Microbial CDMOs enable clinical development by producing live biotherapeutic products and recombinant proteins under strict cGMP standards.
- Key players: Lonza, Catalent, WuXi, and Samsung Biologics.
Market Growth
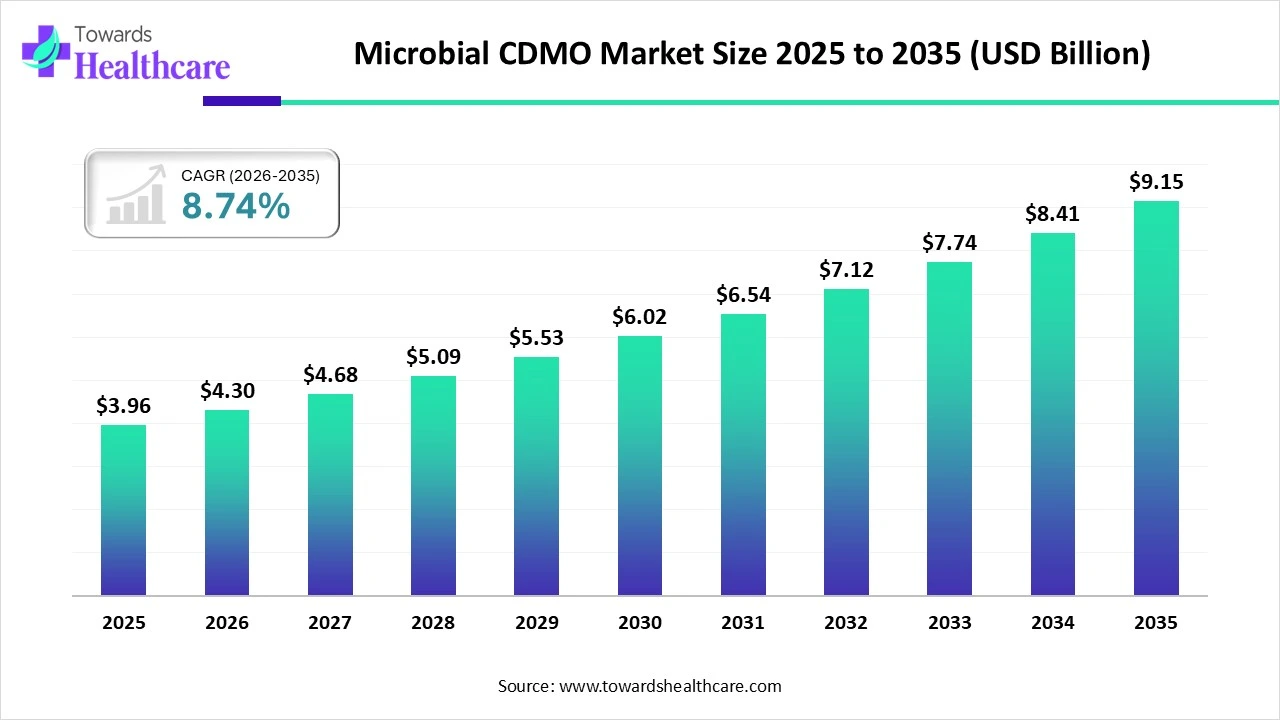
The global microbial CDMO market size was estimated at USD 3.96 billion in 2025 and is predicted to increase from USD 4.3 billion in 2026 to approximately USD 9.15 billion by 2035, expanding at a CAGR of 8.74% from 2026 to 2035.
What are the Recent Developments in the Microbial CDMO Market?
- In March 2025, Syngene acquired a biologics CDMO facility in Baltimore, boosting its bioreactor capacity, early-stage development, and global large-molecule biologics services.
- In December 2024, Lonza exited its capsules and health ingredients division to focus on CDMO operations, reorganizing into three units to boost biologics manufacturing and strengthen its market position.
Stop juggling reports. Manage your entire Microbial CDMO data in one seamless space - Access the Dashboard
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
