Top Vendors in the mRNA CDMO Service Market & Their Offerings with Major Investments
Company Profile
| Companies | Headquarters | Offerings |
| ApexBio Technology | Texas, USA | Provides custom mRNA synthesis, reagents, and early-stage support, including RNA design and IVT services, to accelerate discovery and research programs. |
| BioNTech SE | Mainz, Germany | A biotech leader in mRNA therapeutics and vaccines with in-house and partnered CDMO capabilities for development and manufacturing of mRNA products. |
| Biocina | South Australia, Australia | Offers end-to-end mRNA CDMO services, including process development, analytical support, and GMP manufacturing from preclinical through commercial scale. |
| Lonza Group AG | Basel, Switzerland | Major global CDMO with comprehensive mRNA services covering process optimization, analytical testing, large-scale GMP production, and regulatory support |
| Recipharm AB | Stockholm, Sweden | Provides CDMO services, including mRNA process development, formulation support, and GMP manufacturing from clinical stages to commercialization |
| Novo Holdings (Catalent, Inc.) | Florida, USA | Delivers end-to-end mRNA CDMO solutions, including formulation, fill-finish, and large-scale GMP manufacturing for vaccines and therapies. |
| Samsung Biologics | South Korea | Offers global CDMO services that include mRNA process development, GMP manufacturing, and analytical capabilities to support clinical and commercial mRNA programs. |
Major Investments in the mRNA CDMO Service Market
- In January 2025, the U.S. Department of Health and Human Services committed USD 590 million to Moderna to strengthen mRNA technology infrastructure and speed up the development of mRNA-based influenza pandemic vaccines.
- In October 2024, PopVax secured a USD 1.15 million grant from the Bill & Melinda Gates Foundation to support the development of a thermostable mRNA delivery formulation.
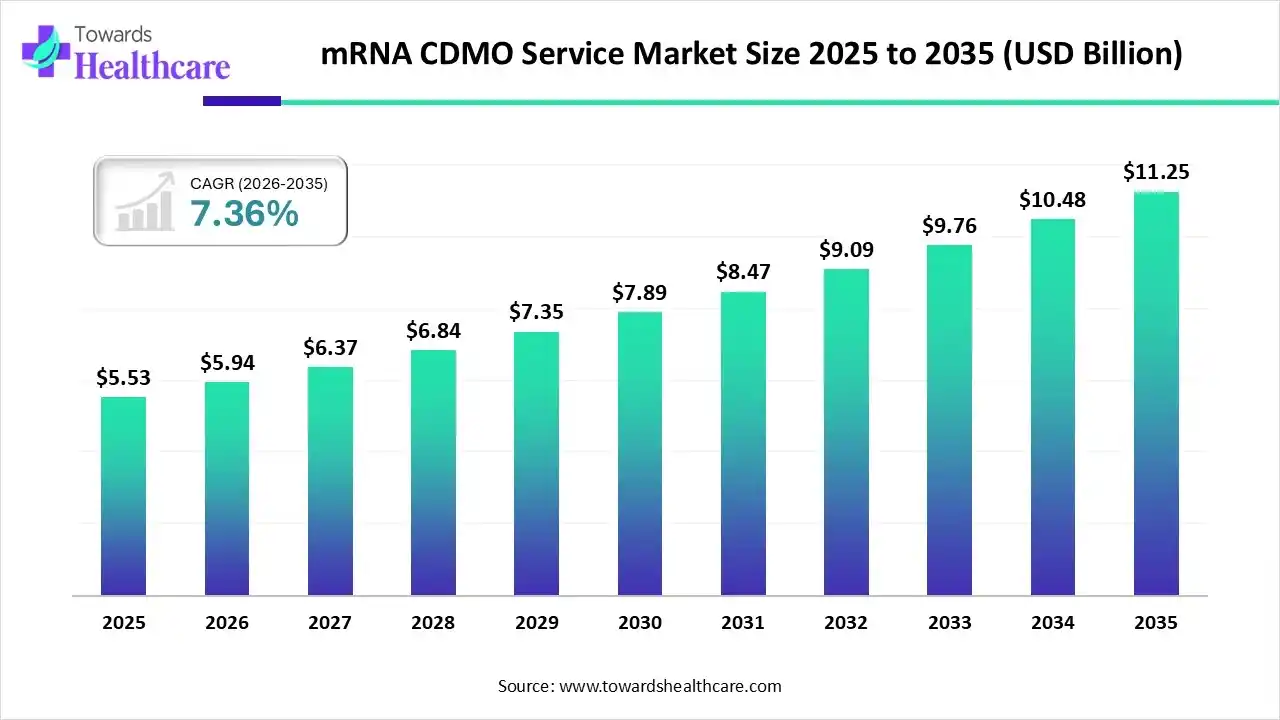
Market Forecast
The global mRNA CDMO service market size was estimated at USD 5.53 billion in 2025 and is predicted to increase from USD 5.94 billion in 2026 to approximately USD 11.25 billion by 2035, expanding at a CAGR of 7.36% from 2026 to 2035.
What are the Recent Developments in the mRNA CDMO Service Market?
- In May 2025, Aldevron announced it would produce a personalized mRNA-based CRISPR therapy for treating urea cycle disorder in infants. In collaboration with the Children’s Hospital of Philadelphia, the University of Pennsylvania, and Danaher subsidiaries Aldevron and IDT, the treatment is expected to be developed in six months—three times faster than typical gene-editing drug timelines.
- In November 2024, SyVento Biotech opened a new mRNA manufacturing facility in Poland covering over 7,000 m², featuring labs, production areas, offices, and shared spaces. This expansion marks a major step in the company’s growth, reinforcing its leadership in Europe’s mRNA manufacturing sector.
Now its easy to manage all your mRNA CDMO Service Market data under one place - Access the dashboard
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
