Best Companies' Offerings in the Oncology Clinical Trial Monitor Market with Key Developments
Company Updates
| Company | Description |
| IQVIA Inc | Its offerings include integration of specialized therapeutic expertise with sophisticated risk-based monitoring (RBM) and real-world data analytics. |
| Charles River Laboratories | They facilitate a global network of clinical pathology labs to assist Phase I-III trials with hematology, biochemistry, and urinalysis under GCP standards. |
| ICON Plc | A firm explores a tech-enabled model by combining on-site, remote, and centralized monitoring. |
| Parexel International Corporation | Its offering encompasses Phase I to IV studies with over 2,500 sites and 80+ ex-agency regulators. |
| WuXi AppTec | They facilitate a complete set of various services, from early-stage discovery to late-stage clinical monitoring and bioanalysis. |
| Syneos Health | Their offering comprises clinical trial management, clinical research associates, and site monitoring leads. |
| Labcorp Drug Development | A company provides an end-to-end oncology clinical trial monitoring and assistance framework |
| PPD Inc. | It is exploring comprehensive, technology-enabled monitoring solutions for oncology clinical trials. |
| Medpace, Inc. | A firm is emphasizing vital study using centralized data reviews, on-site monitoring, and remote visits to confirm safety and data integrity. |
| Allucent | Their offerings include experienced monitors skilled in RECIST/CTCAE criteria, complex medical records, and precision patient recruitment. |
Market Growth
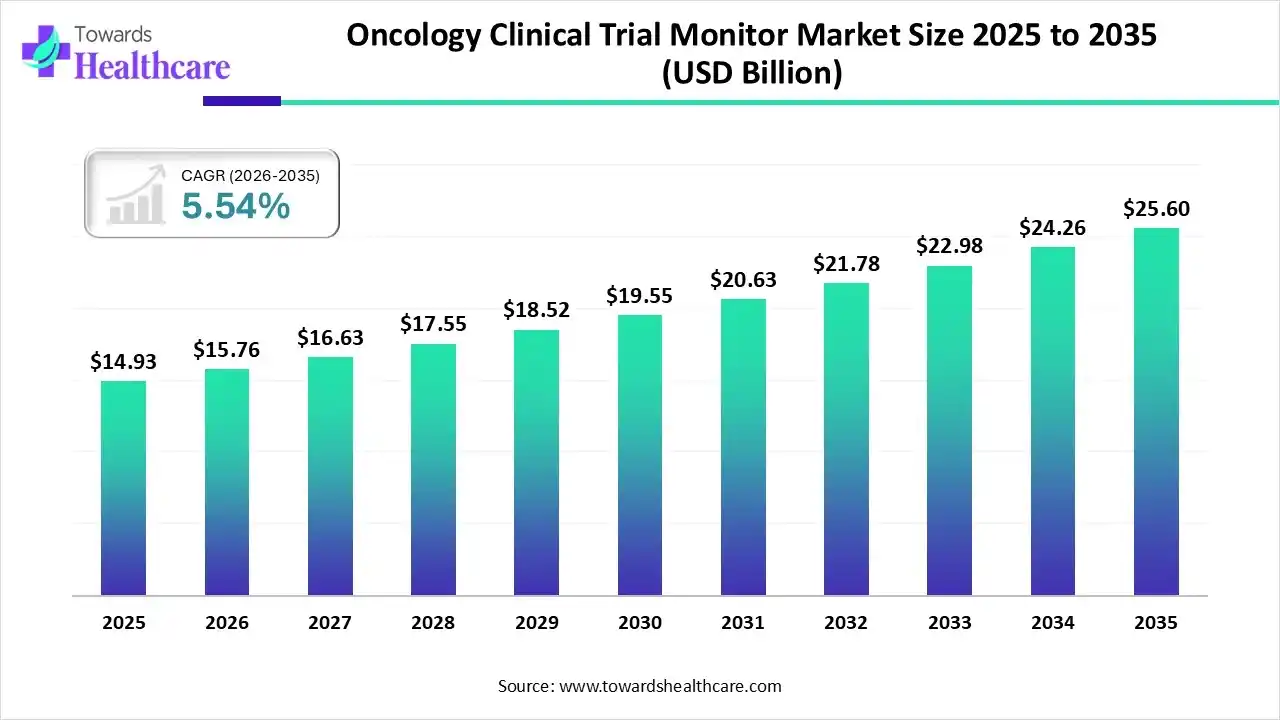
The global oncology clinical trial monitor market size was estimated at USD 14.93 billion in 2025 and is predicted to increase from USD 15.76 billion in 2026 to approximately USD 25.6 billion by 2035, expanding at a CAGR of 5.54% from 2026 to 2035.
What are the Key Developments in the Oncology Clinical Trial Monitor Market?
- In February 2026, Quest Diagnostics launched a novel blood test to evaluate measurable residual disease (MRD) in patients with the blood cancer myeloma.
- In February 2026, Invivoscribe unveiled the LeukoStrat KMT2A + MRD assay to foster high-sensitivity leukemia testing in clinical trials and patient management globally.
Step into the Oncology Clinical Trial Monitor Market dashboard, everything you need, all in one view - Access the Dashboard
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
