Latest Updates of Key Players in the Pancreatic Cancer Precision Medicine Market
Company Background
| Company | Headquarters | Latest Update |
| AstraZeneca plc | United Kingdom | AstraZeneca Plc expects profit to increase further this year, driven by sales of its cancer drugs, as it works to offset the patent expiry of a blockbuster diabetes medicine. |
| Pfizer Inc. | United States | Pfizer Inc. presents data in its portfolio of potential advanced cancer medicines at the 2025 American Society of Clinical Oncology. |
| F. Hoffmann-La Roche Ltd. | Switzerland | Recent cancer developments are transforming the strength to identify and treat cancer, enabling more effective care. |
| Novartis AG | Switzerland | Novartis is reimagining cancer care with RLT for patients with advanced cancers. By harnessing the power of targeted radiation and applying it to advanced cancers, RLT is designed to deliver treatment straight to target cells anywhere in the body. |
| Merck & Co., Inc. | United States | Merck announced the presentation of novel oncology data in more than 12 tumor types at ASCO 2025. |
| Amgen Inc. | United States | Amgen is advancing a pan-KRAS inhibitor, AMG 410, designed to target multiple mutations (both G12C and G12D), as well as wild-type forms of the KRAS protein. |
Value Chain Analysis
R&D:
- R&D processes in pancreatic cancer precision medicine include recognizing actionable targets, enhancing early detection, and disabling drug resistance.
- Key Players: CSL Vifor and Daiichi Sankyo Company, Ltd.
Manufacturing Processes:
- Major manufacturing processes involve miniaturized, 3D, and simplified versions of a patient's tumour that replicate its molecular, histopathological, and healthcare characteristics.
- Key Players: Pharmacosmos A/S, and Fresenius Kabi AG
Patient Services:
- Major services include free tumor biomarker testing, genetic counseling for inherited mutations, and personalized reports that classify specific therapies or clinical trials.
- Key Players: Sanofi
Market Growth
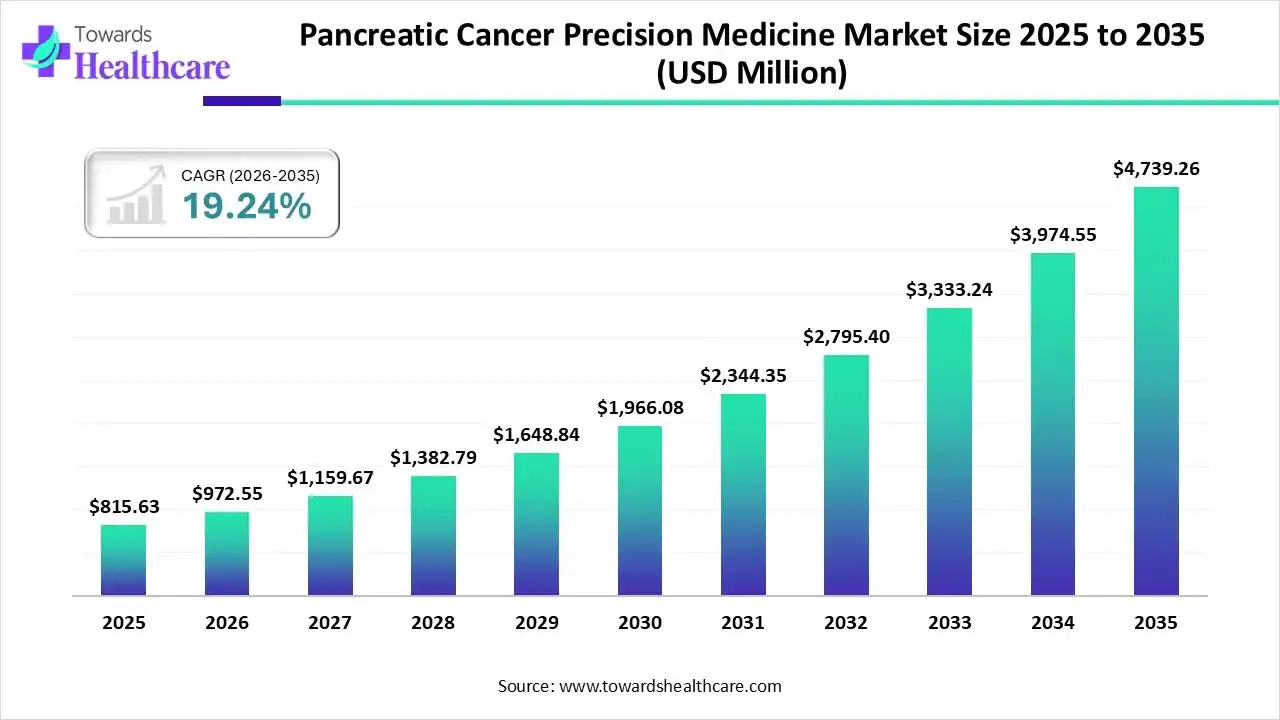
The global pancreatic cancer precision medicine market size was estimated at USD 815.63 million in 2025 and is predicted to increase from USD 972.55 million in 2026 to approximately USD 4739.26 million by 2035, expanding at a CAGR of 19.24% from 2026 to 2035.
Recent Developments in the Pancreatic Cancer Precision Medicine Market
- In February 2026, the FDA approved Optune Pax with gemcitabine/nab-paclitaxel, applying alternating electric fields to disrupt mitosis via abdominal arrays, expanding noninvasive, home-driven treatment delivery for locally progressive pancreatic cancer.
- In February 2026, SOFIE Biosciences, an established U.S. manufacturer and developer of radiopharmaceuticals, today announced that the first patient has been dosed in the second of its two Phase 3 clinical trials evaluating FAPI-74, a fluorine-18 labeled radiopharmaceutical targeting Fibroblast Activation Protein, as a novel diagnostic for patients with Pancreatic Ductal Adenocarcinoma.
- In October 2025, Takeda announced that it had entered into a license and collaboration agreement with Innovent Biologics for the development, manufacturing, and commercialization of two late-stage oncology medicines, IBI363 and IBI343, worldwide outside of Greater China.
From market growth to key vendors, track it all effortlessly - Access the Dashboard
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
