Who are the Pharmaceutical CMO and CDMO Market Top Vendors and What are Their Offerings?

Companies and Key Services
| Pharmaceutical CMO and CDMO | Headquarters | Key Services |
| Lonza Group | Basel, Switzerland | Services focused on small molecule API, biologics, cell, and gene therapies |
| Thermo Fisher Scientific | Waltham, U.S. | End-to-end clinical trial services for drug substances, sterile injectables, and oral solids |
| Catalent, Inc. | Bridgewater, U.S. | Advanced drug delivery and high-volume commercial manufacturing services |
| Samsung Biologics | Incheon, South Korea | Mass-scale biomanufacturing of mAbs and recombinant proteins |
| WuXi Biologics | Wuxi, China | Provides open access technology platforms for discovery, development, and manufacturing |
| Siegfried Holding | Zofingen, Switzerland | Integrated services for drug substances and drug products |
| Boehringer Ingelheim (BioXcellence) | Ingelheim, Germany | Services for mammalian cell culture and microbial fermentation |
| Recipharm AB | Stockholm, Sweden | Services for the development of inhalational products, sterile injectables, small molecules, and large molecules. |
| Fujifilm Diosynth Biotechnologies | Tokyo, Japan | Offer modular manufacturing with a focus on viral vectors, advanced therapies, and vaccines. |
| Piramal Pharma Solutions | Mumbai, India | Integrated manufacturing network for antibody-drug conjugate, sterile injectables, and high-potency APIs |
Market Value Chain Analysis
R&D
- The R&D of the pharmaceutical CMO and CDMO focuses on the utilization of specialized platforms, AI-driven process optimization, and continuous manufacturing for complex modalities.
- Key players: Lonza Group, Thermo Fisher Scientific, Catalent, Inc.
Clinical Trials and Regulatory Approvals
- The data integrity, stability, consistency, process validation, and good manufacturing practice (GMP) compliance are evaluated in the clinical trials and regulatory approvals of the pharmaceutical CMO and CDMO.
- Key players: Lonza Group, Thermo Fisher Scientific, Catalent, Inc.
Packaging and Serialization
- The packaging and serialization of the pharmaceutical CMO and CDMO involve the integration of track-and-trace technologies, automated aggregation systems, and tamper-evident designs.
- Key players: Thermo Fisher Scientific, Catalent, Inc., and Recipharm AB.
Patient Support and Services
- The direct-to-patient (DtP) clinical trial logistics, digital adherence tools, and home nursing coordination are provided in the patient support and services of the pharmaceutical CMO and CDMO.
- Key players: Thermo Fisher Scientific, Catalent, Inc., and Almac Group.
Market Growth
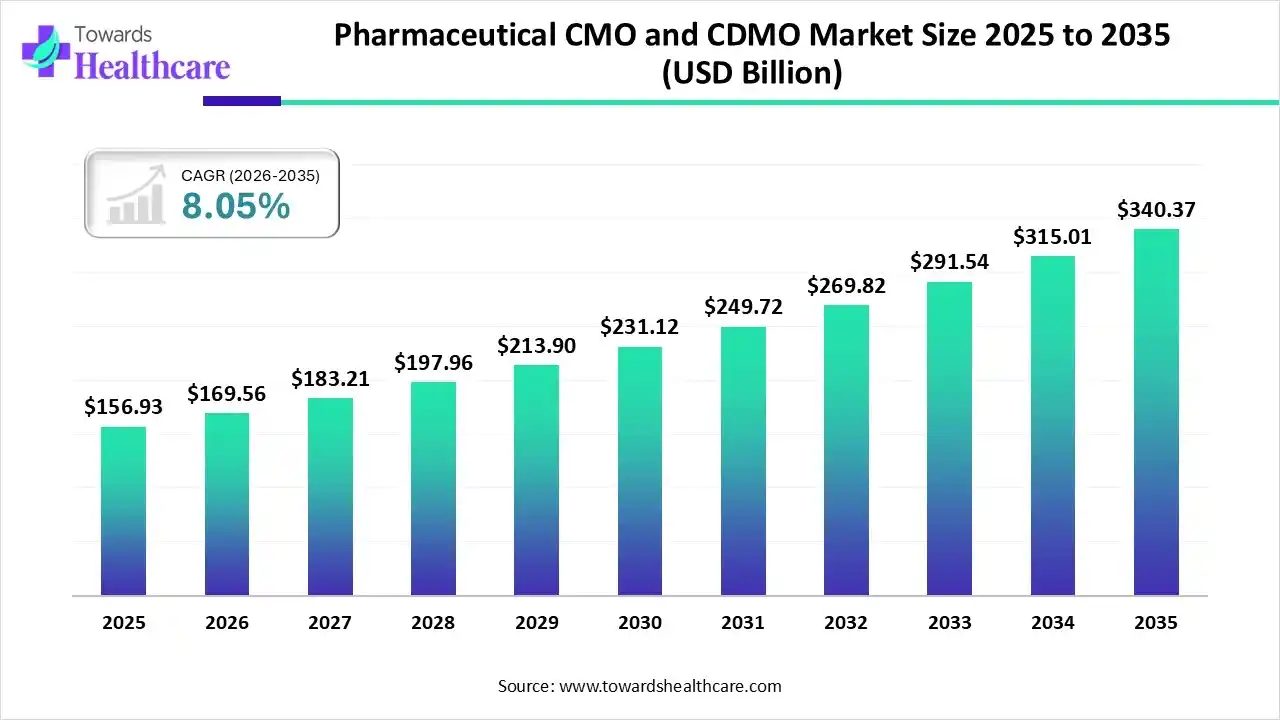
The global pharmaceutical CMO and CDMO market size was estimated at USD 156.93 billion in 2025 and is predicted to increase from USD 169.56 billion in 2026 to approximately USD 340.37 billion by 2035, expanding at a CAGR of 8.05% from 2026 to 2035.
What are the Recent Developments in the Market?
- In November 2025, a significant CDMO partnership between HRV Pharma and MetroChem API was announced, which will support the advanced API development in India, where it will provide scale-up and end-to-end GMP manufacturing and exclusive and semi-exclusive development for multiple strategic molecules.
- In March 2025, a 'hybrid' CDMO model was launched by Indian CDMO, Shilpa Medicare, at DCAT 2025, which will support ready off-the-shelf novel formulations for exclusive b2b licensing.
Turn complex Pharmaceutical CMO and CDMO Market data into clear, confident decisions - Access the Dashboard
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
