
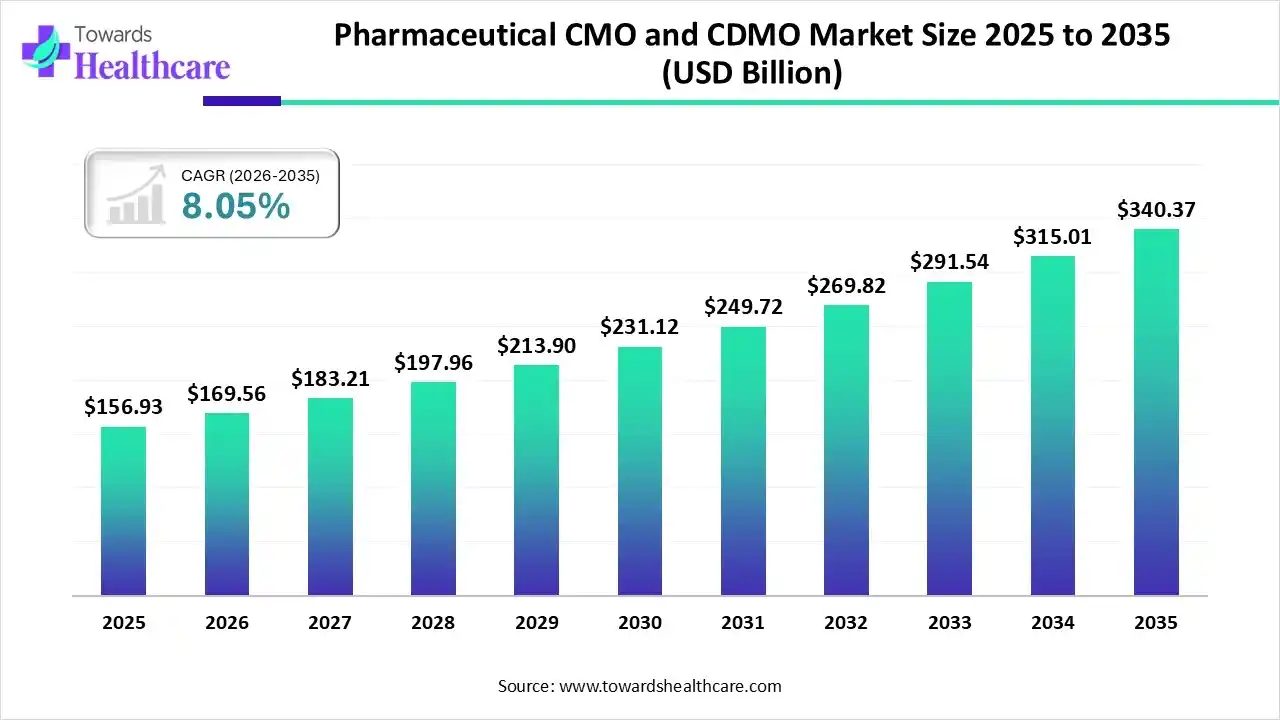
The global pharmaceutical CMO and CDMO market size was estimated at USD 156.93 billion in 2025 and is predicted to increase from USD 169.56 billion in 2026 to approximately USD 340.37 billion by 2035, expanding at a CAGR of 8.05% from 2026 to 2035.
The growing advancements in advanced therapies, vaccines, and biologics are increasing the demand for pharmaceutical CMO and CDMO services. Increasing outsourcing trends, growing innovations, and technological advancements are also promoting the market growth.
The pharmaceutical CMO and CDMO market is driven by the increasing complexity of the modern drug pipeline. The pharmaceutical CMO and CDMO refer to the specialized service providers offering outsourced drug manufacturing and/or development services from R&D to commercialization. They offer affordable services, fastest drug development, and regulatory expertise, which attract the industry.
AI offers a wide range of applications, such as optimization of drug formulations, process optimization, and faster product development timelines in the pharmaceutical CMO and CDMO market. Moreover, its real-time data analysis, predictive maintenance, and batch optimization help in maintaining product quality and reducing the chance of errors. Additionally, its quality control, regulatory compliance, automated documentation, inventory management, and evidence-based insights are also increasing its demand.
Escalation of Complex Dosage Forms
The growing diseases and R&D activities are promoting the development of orphan drugs, personalized treatment, and high-value niche therapies, which are increasing the demand for specialized manufacturing services, leading to a rise in the demand for pharmaceutical CMOs and CDMOs.
Increased Biologics and Biosimilar Manufacturing
The growing health awareness is increasing the adoption of biologic, biosimilar, and gene and cell therapies, which is increasing the collaborations with the pharmaceutical CMO and CDMOs to leverage their services and regulatory expertise.
Increased Advanced Technology Adoption
To enhance operational efficiency, regulatory compliance, and maintain product quality, the pharmaceutical CMOs and CDMOs are utilizing advanced technologies, promoting automation, continuous manufacturing, predictive analytics, etc.
| Table | Scope |
| Market Size in 2026 | USD 169.56 Billion |
| Projected Market Size in 2035 | USD 340.37 Billion |
| CAGR (2026 - 2035) | 8.05% |
| Leading Region | North America by 41% |
| Historical Data | 2020 - 2023 |
| Base Year | 2025 |
| Forecast Period | 2026 - 2035 |
| Measurable Values | USD Millions/Units/Volume |
| Market Segmentation | By Service Type, By Product Type, By End-User, By Region |
| Top Key Players | Lonza Group, Thermo Fisher Scientific, Catalent, Inc., Samsung Biologics, WuXi Biologics, Siegfried Holding, Boehringer Ingelheim (BioXcellence), Recipharm AB, Fujifilm Diosynth Biotechnologies, Piramal Pharma Solutions |
| Segments | Shares % |
| API Manufacturing | 44% |
| Finished Dosage Formulation | 31% |
| Packaging | 17% |
| Others | 8% |
Why Did the API Manufacturing Segment Dominate in the Market in 2025?
The API manufacturing segment led the pharmaceutical CMO and CDMO market by 44% share in 2025, as they are essential for the production of drugs. The growth in the demand for generic and branded drugs also increased the use of these services. Moreover, growth in the investments and outsourcing trends also increased their use.
Finished Dosage Formulation
The finished dosage formulation segment is expected to show the highest growth during the upcoming years, due to the growing development of vaccines and other injectable products. Additionally, expanding development of modified release and combination products is also increasing the demand for finished dosage formulation services.
| Segments | Shares % |
| Small Molecule | 63% |
| Biologics | 37% |
Which Product Type Segment Held the Dominating Share of the Market in 2025?
The small molecule segment held the dominating share of the pharmaceutical CMO and CDMO market by 63% in 2025, due to growth in their approval rates. Increased rate of generic small molecule production, along with increased outsourcing, led to growth in the pharmaceutical CMO and CDMO collaborations.
Infographics
Biologics
The biologics segment is expected to expand rapidly in the market during the predicted time, due to growing demand for targeted therapies. Similarly, increasing development of monoclonal antibodies and specialized therapies is also increasing the use of pharmaceutical CMO and CDMO services, where the growing clinical trials are also increasing their demand.
| Segments | Shares % |
| Pharmaceutical Companies | 58% |
| Biotechnology Companies | 31% |
| Others | 11% |
What Made Pharmaceutical Companies the Dominant Segment in the Market in 2025?
The pharmaceutical companies segment registered its dominance over the global pharmaceutical CMO and CDMO market by 58% share in 2025, due to growth in the manufacturing process. The increased R&D activities and investments also increased the partnership with the pharmaceutical CMO and CDMOs to leverage their advanced technologies and expertise.
Biotechnology Companies
The biotechnology companies segment is expected to show the highest growth during the upcoming years, due to growing advancements in biologics, orphan drugs, and personalized therapies development. The expanding startup ecosystems and funding are also increasing the collaboration with the pharmaceutical CMOs and CDMOs.
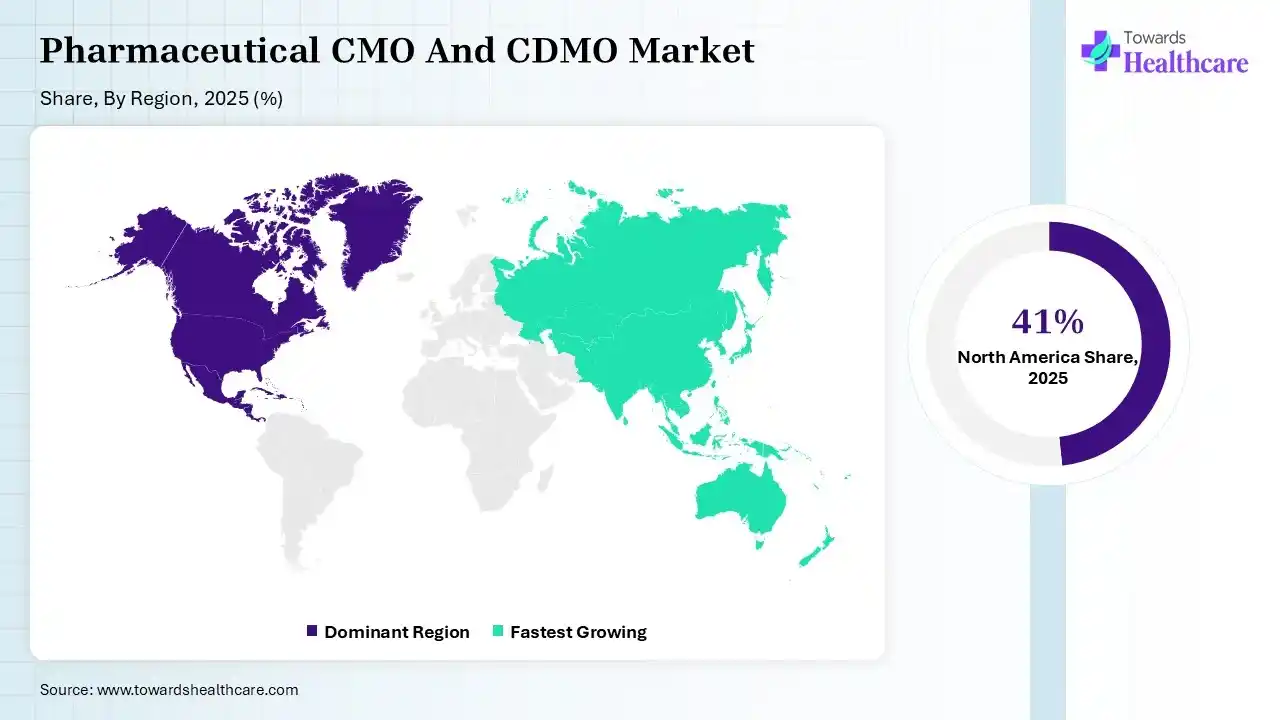
North America dominated the pharmaceutical CMO and CDMO market by 41% share in 2025, due to the presence of well-developed pharmaceutical and biotechnology companies, which actively participated in the clinical trials and R&D activities. This increased the demand for pharmaceutical CMO and CDMO to leverage their services, advanced facilities, and technologies, which contributed to the market growth.
U.S. Market Trends
The U.S. is a home for top pharmaceutical and biotechnology companies, which are increasing the advancements in R&D and clinical trials, leading to a growth in the reliance on the pharmaceutical CMO and CDMOs. Their advanced manufacturing facilities, stringent regulatory compliance, and advanced technologies are also increasing their collaborations.
Asia Pacific is expected to host the fastest-growing pharmaceutical CMO and CDMO market by 22% share during the forecast period, due to expanding manufacturing capabilities and increasing generic drug production. Additionally, expanding industries, growing demand for biologics, vaccines, and increasing clinical trials are also driving the demand for pharmaceutical CMO and CDMO, enhancing the market growth.
India Market Trends
India is experiencing a rapid growth in the biotech sector, driving the development of biological, advanced therapies, biosimilars, and specialty drugs, which is leading to a growth in strategic partnerships with the pharmaceutical CMO and CDMOs. Growing government initiatives, generic products development, and affordable manufacturing services are also increasing their acceptance rates.
Europe is expected to grow significantly in the pharmaceutical CMO and CDMO market by 30% share during the forecast period, due to the presence of robust R&D ecosystems, advanced pharmaceutical and biotech hubs, and stringent regulations, which encourage the use of pharmaceutical CMO and CDMOs. The growing demand for cell and gene therapies is also driving their demand, promoting market growth.
UK Market Trends
The UK consists of a well-established pharmaceutical and biotechnology hub, which is increasing the number of innovations. The universities and research institutes are also increasing their R&D activities focused on the development of small molecules and biologics, which is leading to new collaborations with the pharmaceutical CMOs and CDMOs.
R&D
Clinical Trials and Regulatory Approvals
Packaging and Serialization
Patient Support and Services

| Pharmaceutical CMO and CDMO | Headquarters | Key Services |
| Lonza Group | Basel, Switzerland | Services focused on small molecule API, biologics, cell, and gene therapies |
| Thermo Fisher Scientific | Waltham, U.S. | End-to-end clinical trial services for drug substances, sterile injectables, and oral solids |
| Catalent, Inc. | Bridgewater, U.S. | Advanced drug delivery and high-volume commercial manufacturing services |
| Samsung Biologics | Incheon, South Korea | Mass-scale biomanufacturing of mAbs and recombinant proteins |
| WuXi Biologics | Wuxi, China | Provides open access technology platforms for discovery, development, and manufacturing |
| Siegfried Holding | Zofingen, Switzerland | Integrated services for drug substances and drug products |
| Boehringer Ingelheim (BioXcellence) | Ingelheim, Germany | Services for mammalian cell culture and microbial fermentation |
| Recipharm AB | Stockholm, Sweden | Services for the development of inhalational products, sterile injectables, small molecules, and large molecules. |
| Fujifilm Diosynth Biotechnologies | Tokyo, Japan | Offer modular manufacturing with a focus on viral vectors, advanced therapies, and vaccines. |
| Piramal Pharma Solutions | Mumbai, India | Integrated manufacturing network for antibody-drug conjugate, sterile injectables, and high-potency APIs |
Strengths
Weaknesses
Opportunities
Threats
By Service Type
By Product Type
By End-User
By Region