Meet the Top Companies of the Pharmaceutical CRO and CDMO Market and Explore their Company Profile
Company Profile
| Pharmaceutical CRO and CDMO | Headquarters | Services |
| IQVIA | North Carolina, U.S. | Phase 1-4 clinical trials and advanced technologies |
| ICON plc | Dublin, Ireland | Full-service clinical development, biosimilar research, and adaptive trial designs |
| Parexel | Massachusetts, U.S. | Phase 1-4 clinical development, AI-driven trial simulation, and post-market surveillance |
| Charles River Laboratories | Massachusetts, U.S. | Preclinical discovery, laboratory models, efficacy testing ad safety assessment |
| Medpace | Ohio, U.S. | Integrated full-service clinic trials and central laboratory services |
| Lonza Group | Basel, Switzerland | Biologic manufacturing, API synthesis, and cell and gene therapy development. |
| Samsung Biologics | Incheon, South Korea | Large-scale biologics manufacturing, small molecule substances, drug product filling, and monoclonal antibodies production |
| Catalent | New Jersey, U.S. | Oral, respiratory drug delivery technologies, cell and gene therapy manufacturing |
| Boehringer Ingelheim | Ingelheim, Germany | Contract manufacturing for biologics, small molecule chemical innovation, and process development |
| Thermo Fisher Scientific | Massachusetts, U.S. | End-to-end solutions, API manufacturing, sterile fill-finish, and clinical trials logistics |
Market Value Chain Analysis
R&D
- The R&D of the pharmaceutical CRO and CDMO focuses on enhancing drug discovery and optimizing their manufacturing using advanced platforms and AI-driven solutions.
- Key players: IQVIA, WuXi AppTec, Lonza.
Clinical Trials and Regulatory Approvals
- The drug safety, quality, efficacy, and manufacturing compliance are evaluated in the clinical trials and regulatory approvals of the pharmaceutical CRO and CDMO.
- Key players: ICON plc, Lonza, Catalent.
Formulation and Final Dosage Preparation
- The formulation and final dosage preparation in the pharmaceutical CRO and CDMO focus on the development of various formulations ranging from oral solids to sterile injectables by optimizing drug stability and delivery mechanisms.
- Key players: Lonza, Catalent, Patheon.
Packaging and Serialization
- The packaging and serialization of pharmaceutical CRO and CDMO products involves the use of tamper-evident designs and unique data track barcodes to maintain product integrity and supply chain traceability.
- Key players: Thermo Fisher Scientific, Catalent, PCI Pharma Services.
Patient Support and Services
- Decentralized trials platform, digital engagement tools, and home nursing are provided in the patient support and services of the pharmaceutical CRO and CDMOs.
- Key players: IQVIA, ICON plc, Parexel.
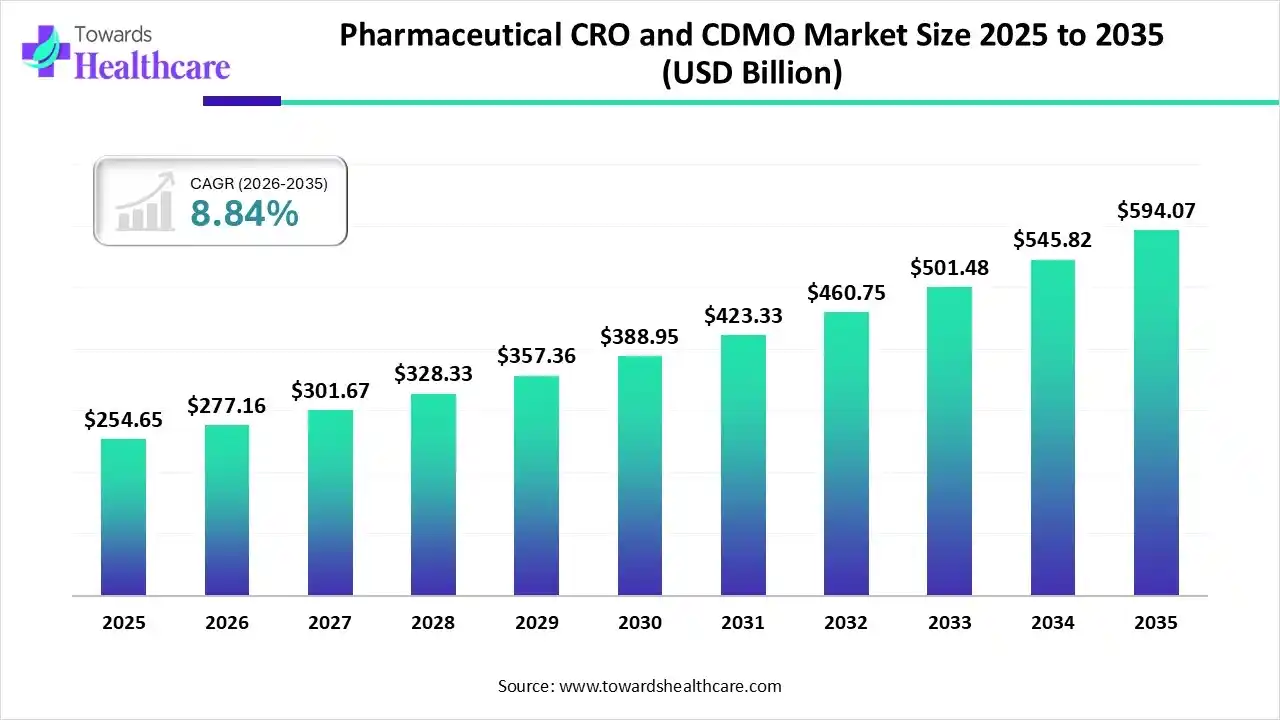
Market Growth
The global pharmaceutical CRO and CDMO market size was estimated at USD 254.65 billion in 2025 and is predicted to increase from USD 277.16 billion in 2026 to approximately USD 594.07 billion by 2035, expanding at a CAGR of 8.84% from 2026 to 2035.
Recent Developments in the Market
- In January 2026, a collaboration was formed between ST Pharm, which is a Korean CDMO, and a U.S.-based biotech company, where they will develop an oligonucleotide-based treatment for patients suffering from severe hypertriglyceridemia.
- In March 2025, a 'hybrid' CDMO model was launched by Shilpa Medicare, which is an Indian CDMO at DCAT 2025, where the company will offer traditional CDMO services and ready-to-license novel formulations.
Get complete Pharmaceutical CRO and CDMO Market data and deep insights under a single roof: Access the Dashboard
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
