Latest Updates of Key Players in the Pharmaceutical Intermediate CDMO Market

| Company | Headquarters | Latest Update |
| Lonza Group | Switzerland | Lonza Group completed its strategic transformation into a pure-play CDMO by initiating the divestment of its Capsules & Health Ingredients (CHI) business. |
| Thermo Fisher Scientific | United States | Thermo Fisher Scientific has increased its bioprocess design center network in Asia, adding a novel facility in Hyderabad, India, and expanding existing centers in Incheon, Korea, and Singapore. |
| Catalent | United States | In October 2025, Catalent, a leading worldwide contract development and manufacturing organization (CDMO), stated that it would address recent inaccuracies in media coverage regarding the ownership of one of its former sites located in Bloomington, Indiana. |
| WuXi Biologics/AppTec | United States | The company continuously drives green technology inventions to provide advanced end-to-end Green CRDMO services for its worldwide partners |
| Samsung Biologics | South Korea | Samsung Biologics continued to increase its global CDMO business through long-term partnerships with pharmaceutical companies worldwide. |
| CordenPharma | Switzerland | CordenPharma is a CDMO partner supporting biotech and pharma innovators of multifaceted modalities in the advancement of their drug development. |
Get all Pharmaceutical Intermediate CDMO Market insights in one place; from data to competitor strategies. Access the dashboard and stay ahead
Supply Chain Analysis
R&D:
- Research and development (R&D) for pharmaceutical intermediate CDMO includes route scouting and design, process optimization, process safety assessments, scale-up studies, and flow chemistry development
- Key Players: Samsung Biologics
Manufacturing Processes:
- Manufacturing processes in a pharmaceutical intermediate CDMO involve a sequence of chemical synthesis, purification, and physical processing, ranging from pilot-scale to commercial-scale production.
- Key Players: Catalent and WuXi Biologics/AppTec
Patient Services:
- Patient services in clinical-stage biotechnology include improved drug formulation and palatability, enhanced adherence and compliance, safety and quality assurance, customization for rare diseases, and handling complex and potent drugs.
- Key Players: Lonza Group and Thermo Fisher Scientific
Market Growth
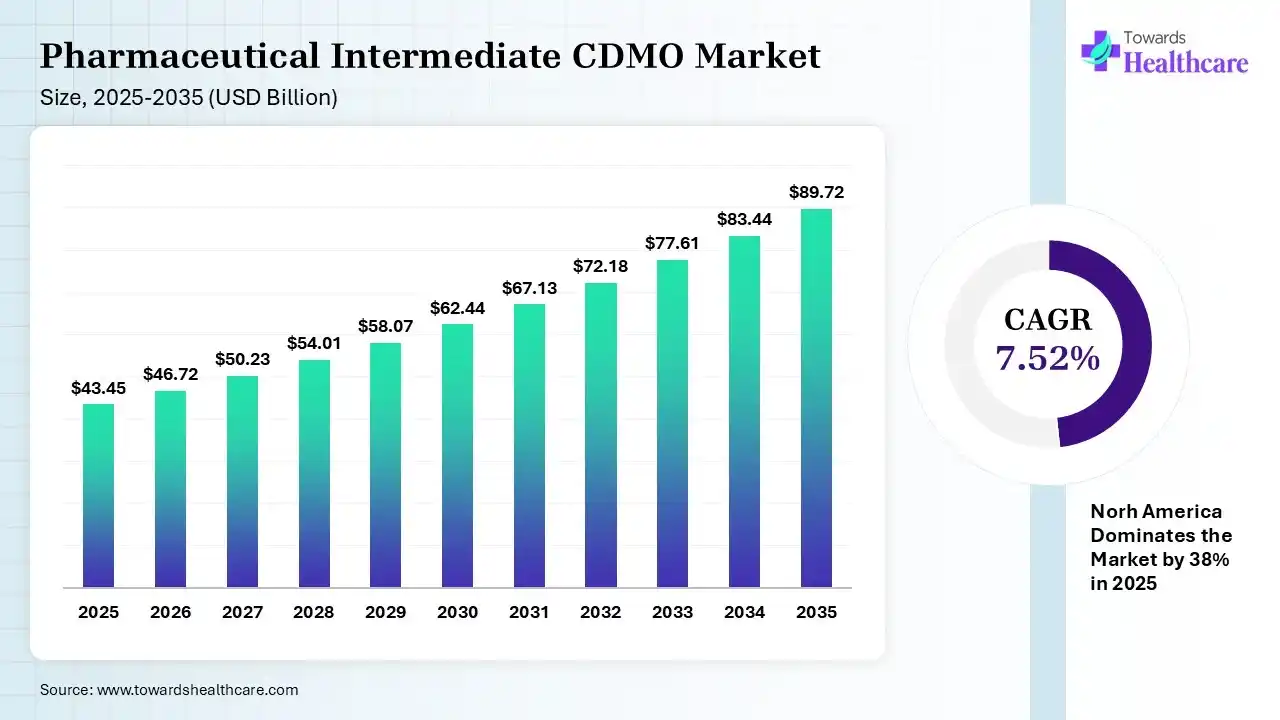
The global pharmaceutical intermediate CDMO market size was estimated at USD 43.45 billion in 2025 and is predicted to increase from USD 46.72 billion in 2026 to approximately USD 89.72 billion by 2035, expanding at a CAGR of 7.52% from 2026 to 2035.
The Pharmaceutical Companies Segment Led the Pharmaceutical Intermediate CDMO Market in 2025
| Segment | Share 2025 (%) |
| Pharmaceutical Companies (Dominant) | 60% |
| Biotechnology Companies (Fastest Growing) | 40% |
Explanation
- Pharmaceutical Companies dominate the market with 60% share due to their substantial demand for chemical intermediates in drug production and healthcare solutions.
- Biotechnology Companies represent 40% of the market and are the fastest growing due to the increasing need for specialized chemical solutions in biotech research and development.
The pharmaceutical companies segment dominated the market by 60% in 2025, as CDMOs speed up the drug advancement timeline, supporting healthcare organizations in bringing new drugs to market more rapidly. An efficient partnership with a CDMO leads to significant expense savings via optimized processes and economies of scale. A CDMO partner is a strategic decision that has a profound impact on the success of drug advancement and production. It allows them to focus on their core competencies, like scientific research and promotion.
Whereas the biotechnology companies segment is the fastest-growing in the market, as CDMO services provide scalability, expertise, cost savings, and speed, CDMOs allow biotech organizations to focus on novel strategies while trusting the production process to specialists. CDMOs streamline advancement timelines, enabling biotechs to transition significantly from clinical trials to commercial manufacturing.
Recent Developments in the Pharmaceutical Intermediate CDMO Market
- In October 2025, Terumo Corporation announced the successful completion of its acquisition of a Drug Product Plant and related Quality Control Laboratory operations from WuXi Biologics in Leverkusen, Germany, with the transaction officially closing.
- In October 2025, LOTTE BIOLOGICS and SK Pharmteco announced that they had signed a Letter of Intent (LOI) for a strategic partnership to strengthen their competitiveness in the global antibody-drug conjugate (ADC) market.
- In July 2025, ESTEVE acquired Regis Technologies, a United States-based Contract Development and Manufacturing Organization (CDMO), headquartered in Chicago. This strategic shift permits ESTEVE CDMO a physical presence in the United States, growing its contract advancement and manufacturing services for new small-molecule active pharmaceutical ingredients (APIs) in the entire drug development lifecycle from pre-clinical to commercial manufacturing.
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
