March 2026

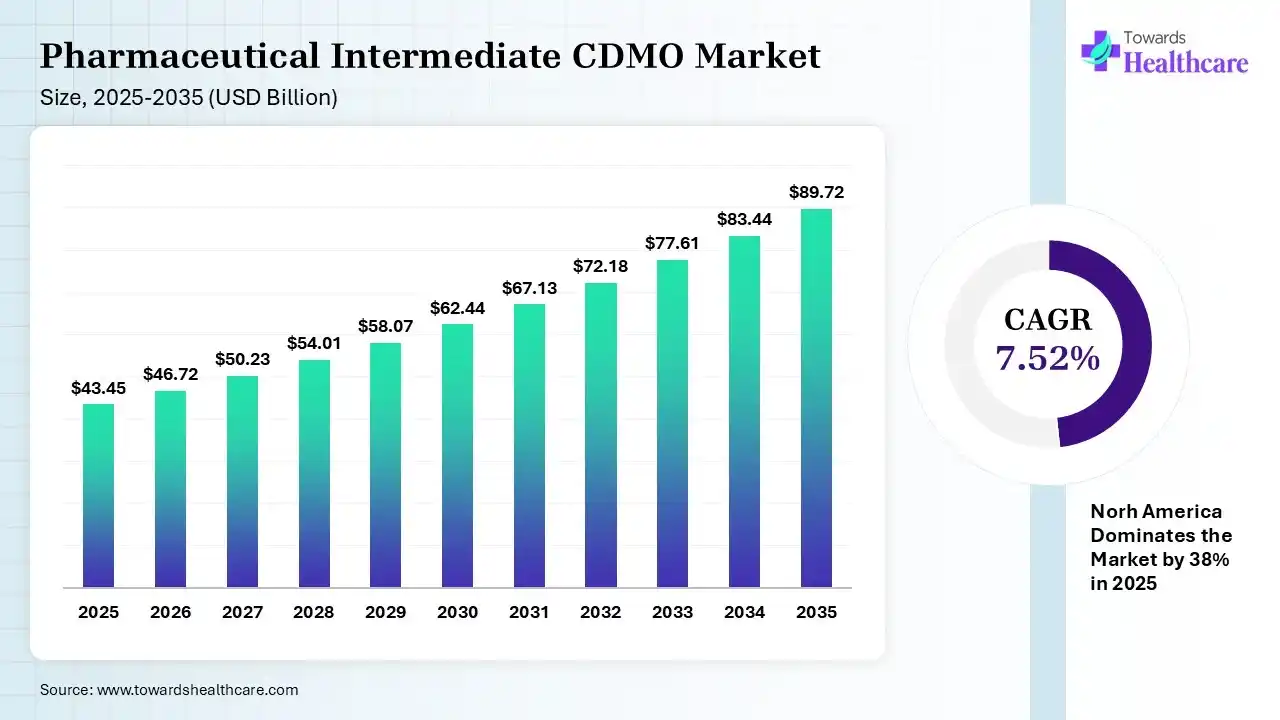
The global pharmaceutical intermediate CDMO market size was estimated at USD 43.45 billion in 2025 and is predicted to increase from USD 46.72 billion in 2026 to approximately USD 89.72 billion by 2035, expanding at a CAGR of 7.52% from 2026 to 2035.
The pharmaceutical intermediate CDMO market is growing because of the increasing applications in cGMP manufacturing of intermediates, clinical trial material development, and therapeutic specialization such as oncology, cardiovascular, CNS, and biologics and vaccines production.
The pharmaceutical intermediate CDMO market is growing, as it offers end-to-end services, from drug development via commercial production, for healthcare and biotech organizations. By speeding up the drug development technology, lowering expenses, and allowing more effective production of healthcare products, CDMOs are becoming crucial to the pharmaceutical industry. Collaborating with a CDMO supports any pharmaceutical organization or biotech firm in various ways. CDMOs offer regulatory expertise and quality control support, supporting clients in satisfying regulatory authorities and maintaining the highest standards throughout production. A CDMO partnership proves advantageous for a broad range of organizations in the pharmaceutical sector and healthcare space.
AI-based technology in pharmaceutical intermediate CDMO is transforming CDMOs with smarter healthcare manufacturing, enhancing manufacturing, compliance, logistics, and sustainability. AI-based technology has transformed the large molecule CDMO sector by improving drug discovery and advancement processes, increasing manufacturing efficiency, and improving quality control. AI-based technology is used to optimize molecular synthesis, predicting the standard chemical routes for intermediates.
AI-based tools such as Elsevier’s Reaxys analyze massive datasets to identify efficient pathways, slashing advancement time. AI-based platforms analyze raw material availability, vendor performance, and shipping data to manage challenges in the supply chain. AI-based technology is playing a transformative role in the healthcare intermediate CDMO landscape by improving process development.
| Table | Scope |
| Market Size in 2026 | USD 46.72 Billion |
| Projected Market Size in 2035 | USD 89.72 Billion |
| CAGR (2026 - 2035) | 7.52% |
| Leading Region | North America by 38% |
| Historical Data | 2020 - 2023 |
| Base Year | 2025 |
| Forecast Period | 2026 - 2035 |
| Measurable Values | USD Millions/Units/Volume |
| Market Segmentation | By Product Type, By Service Type, By End Use, By Region |
| Top Key Players | Lonza Group, Thermo Fisher Scientific, Catalent, WuXi Biologics/AppTec, Samsung Biologics, CordenPharma |
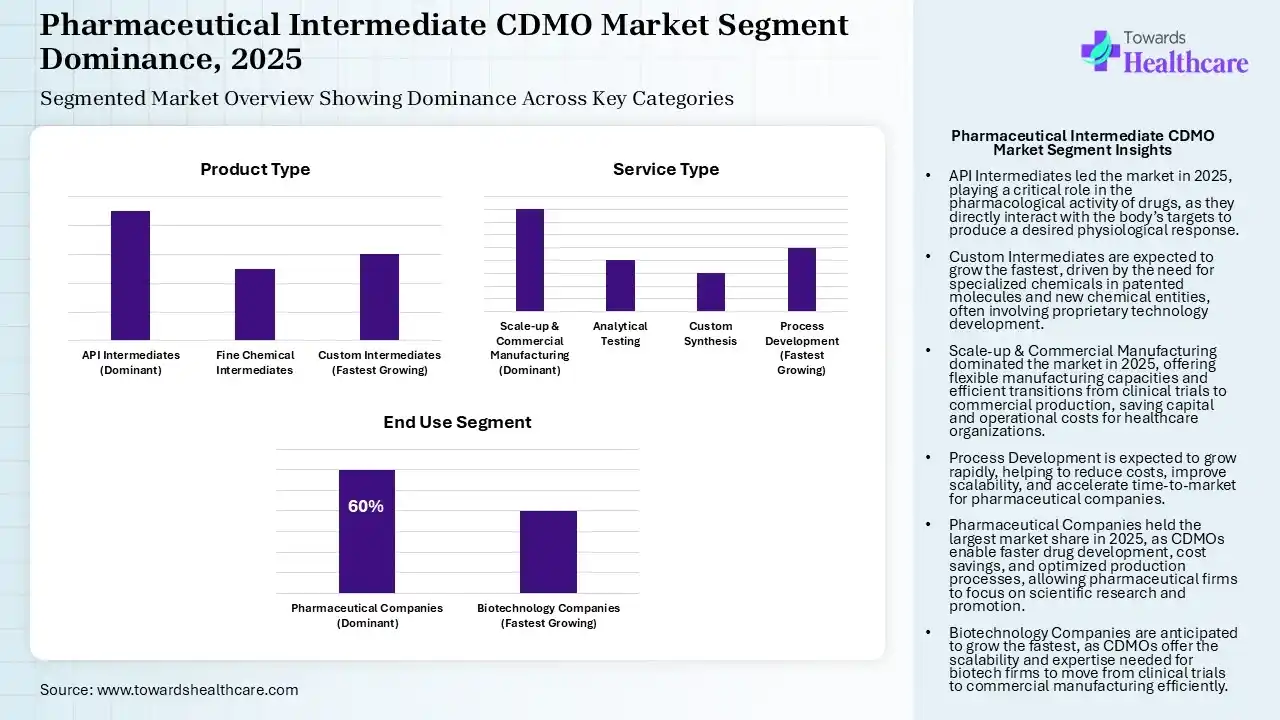
The API Intermediates Segment Led the Pharmaceutical Intermediate CDMO Market in 2025
| Segment | Share 2025 (%) |
| API Intermediates (Dominant) | 45% |
| Fine Chemical Intermediates | 25% |
| Custom Intermediates (Fastest Growing) | 30% |
Explanation
The API intermediates segment dominated the market by 45% in 2025, as this intermediate is a chemically active substance that is accountable for the designed pharmacological activity of a drug. These ingredients are generally synthesized via chemical processes or derived from natural sources. The major role of API intermediates in pharmaceutical products is significant. They are responsible for the healing effect of the drugs, meaning they directly interact with the target in the body to produce a desired physiological response.
Whereas the custom intermediates segment is estimated to be the fastest-growing in the market, as these intermediates are satisfactory chemicals that are intermediate compounds produced during the manufacturing of active pharmaceutical ingredients (APIs). Custom intermediates are industrialized precisely for a client’s proprietary medication molecule. These needs require confidential technology development, route optimization, impurity control, and long-term exclusivity arrangements. Custom synthesis is broadly applicable for patented molecules, new chemical entities, and specialised medication pipelines.
The Scale-up & Commercial Manufacturing Segment Led the Pharmaceutical Intermediate CDMO Market in 2025
| Segment | Share 2025 (%) |
| Scale-up & Commercial Manufacturing (Dominant) | 40% |
| Analytical Testing | 20% |
| Custom Synthesis | 15% |
| Process Development (Fastest Growing) | 25% |
Explanation
The scale-up & commercial manufacturing segment dominated the market by 40% in 2025, as CDMOs offer adaptable manufacturing capacities, which are significant during scale-up phases and when responding to marketplace demands, confirming effective transitions from clinical trials to commercial production. Healthcare organizations significantly lower capital and operational expenditures, as CDMOs manage the entirety of the production process, from raw material procurement to end product. CDMOs streamline the production and quality control processes.
Whereas the process development segment is the fastest-growing in the market, as contract development and manufacturing administrations increase yield, save expenses, and support scalability in healthcare and biotech. Efficient process advancement plays a significant role in reducing challenges, accelerating time-to-market, and significantly lowering development expenses for pharmaceutical organizations.
The Pharmaceutical Companies Segment Led the Pharmaceutical Intermediate CDMO Market in 2025
| Segment | Share 2025 (%) |
| Pharmaceutical Companies (Dominant) | 60% |
| Biotechnology Companies (Fastest Growing) | 40% |
Explanation
The pharmaceutical companies segment dominated the market by 60% in 2025, as CDMOs speed up the drug advancement timeline, supporting healthcare organizations in bringing new drugs to market more rapidly. An efficient partnership with a CDMO leads to significant expense savings via optimized processes and economies of scale. A CDMO partner is a strategic decision that has a profound impact on the success of drug advancement and production. It allows them to focus on their core competencies, like scientific research and promotion.
Whereas the biotechnology companies segment is the fastest-growing in the market, as CDMO services provide scalability, expertise, cost savings, and speed, CDMOs allow biotech organizations to focus on novel strategies while trusting the production process to specialists. CDMOs streamline advancement timelines, enabling biotechs to transition significantly from clinical trials to commercial manufacturing.
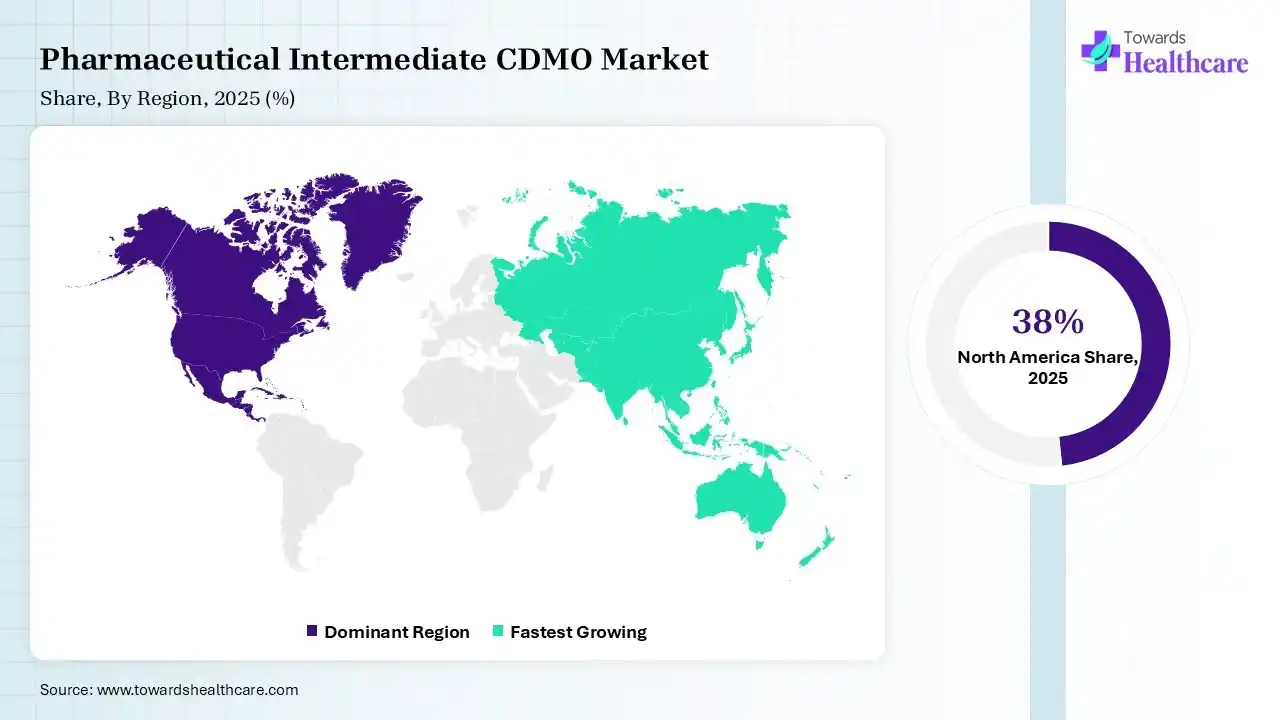
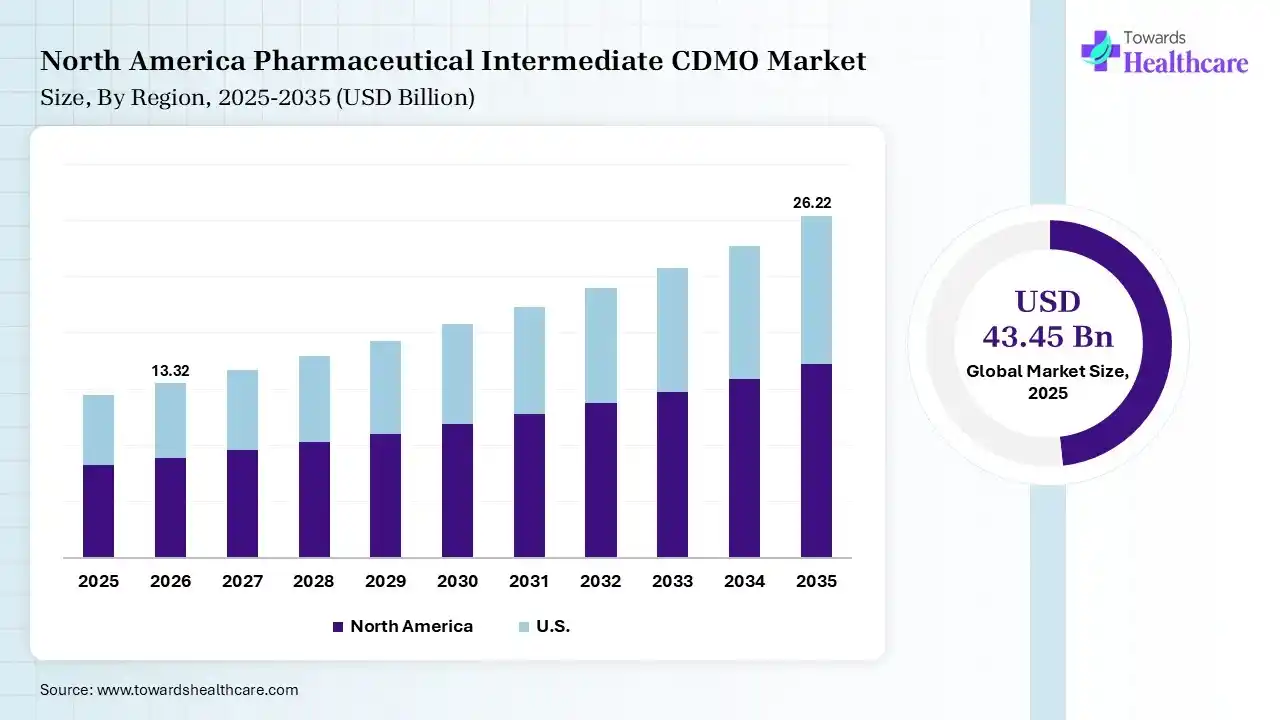
In 2025, North America dominated the pharmaceutical intermediate CDMO market by 38% because growing demand for care and changes in the mix and intensity of solutions explicate much of the recent speedup in investing, specifically for hospital care, surgeon services, and prescription drugs. The growing prevalence of chronic diseases and rising R&D spending in pharmaceuticals and biotechnology which drives the growth of the market.
U.S. Market Trends
U.S.-driven CDMO is well-positioned to prioritize a domestic supply chain and ensure reliable access to vital raw materials, specialty chemicals, and solvents. Increasing spending in vertical integration for significant raw materials like regulatory starting materials (RSMs) and intermediates further improves supply chain security.
Asia Pacific is expected to see rapid growth in the pharmaceutical intermediate CDMO, as with rapidly increasing aging populations and lessening workforces in APAC, the growth in healthcare expenses is inevitable. This region evolves as a competitive biologics production hub. These outsourcing activities are increasing across the discovery continuum, from chemical synthesis to target identification and validation to biological testing, which drives the growth of the market.
India Market Trends
India provides pharmaceutical contract manufacturing solutions at costs nearly 20% lower than China, while upholding worldwide regulatory standards. India is the largest global supplier of generic medicines, accounting for around 20 % of the worldwide supply, producing about 60,000 generic brands in 60 therapeutic categories. India provides healthcare services at reasonably low costs, attracting international patients looking for quality treatment at cost-effective prices.
R&D:
Manufacturing Processes:
Patient Services:

| Company | Headquarters | Latest Update |
| Lonza Group | Switzerland | Lonza Group completed its strategic transformation into a pure-play CDMO by initiating the divestment of its Capsules & Health Ingredients (CHI) business. |
| Thermo Fisher Scientific | United States | Thermo Fisher Scientific has increased its bioprocess design center network in Asia, adding a novel facility in Hyderabad, India, and expanding existing centers in Incheon, Korea, and Singapore. |
| Catalent | United States | In October 2025, Catalent, a leading worldwide contract development and manufacturing organization (CDMO), stated that it would address recent inaccuracies in media coverage regarding the ownership of one of its former sites located in Bloomington, Indiana. |
| WuXi Biologics/AppTec | United States | The company continuously drives green technology inventions to provide advanced end-to-end Green CRDMO services for its worldwide partners |
| Samsung Biologics | South Korea | Samsung Biologics continued to increase its global CDMO business through long-term partnerships with pharmaceutical companies worldwide. |
| CordenPharma | Switzerland | CordenPharma is a CDMO partner supporting biotech and pharma innovators of multifaceted modalities in the advancement of their drug development. |
Strengths
Weaknesses
Opportunities
Threats
By Product Type
By Service Type
By End Use
By Region
March 2026
March 2026
March 2026
March 2026
