Latest Updates of Companies in the Poly-L-Lactic Acid Filler Market

| Company | Headquarters | Latest Update |
| Galderma | Switzerland | In February 2025, Galderma, the pure-play dermatology category leader, announced the launch of Restylane Defyne and Restylane Refyne, the first OBT‑based hyaluronic acid injectables ever authorized in Japan. |
| Sinclair Pharma | England | Sinclair continues to differentiate Lanluma with a focus on body contouring and facial rejuvenation. |
| Regen Biotech | South Korea | Regen is the global third and South Korea's first organization to obtain monitoring approval to sell polylactic acid skin filler products. |
| Dermapharm | Germany | Dermapharm Holding SE strives to be recognized as a significant player in the growing Poly-L-Lactic Acid filler. |
| Prollenium Medical Technologies | Ontario | In September 2025, PROLLENIUM is proudly manufactured in Canada under FDA-approved superiority standards and holds a seven-year safety record with its REVANESSE product line. |
Recent Developments in the Poly-L-Lactic Acid Filler Market
- In March 2026, Allergan Aesthetics, an AbbVie company, a worldwide leader in healthcare aesthetics treatments and manufacturer of the market-leading JUVÉDERM collection of fillers, is once again forging ahead and has announced the 'Undetectable† Era'.
- In April 2025, Galderma today announced the launch of Sculptra, the first proven regenerative biostimulator with a particular poly-L-lactic acid formulation, in China. Sculptra, the first proven regenerative biostimulator, offers novel options for patients in China who are looking for new treatments to manage mid-facial volume loss.
- In February 2025, Galderma, the pure-play dermatology category leader, announced the launch of Restylane Defyne and Restylane Refyne, the first OBT‑based hyaluronic acid injectables ever authorized in Japan.
Supply Chain Analysis
R&D:
- Research and development (R&D) in poly-l-lactic acid filler includes material synthesis and modification, microsphere engineering and formulation, biological activity and collagen stimulation studies, progressive delivery and application technologies, and quality control and government validation.
- Key Players: Sculptra Aesthetic
Manufacturing Processes:
- The manufacturing processes for poly-l-lactic acid filler involve high-purity polymer synthesis, microparticle fabrication, and sterile wrapping.
- Key Players: Rejeunesse
Patient Services:
- Patient services are comprehensive consultation, a series of injection sessions, particular post-procedure care protocols, and long-term evaluation to achieve natural, long-lasting volume restoration
- Key Players: AestheFill and Derma Veil
Market Growth
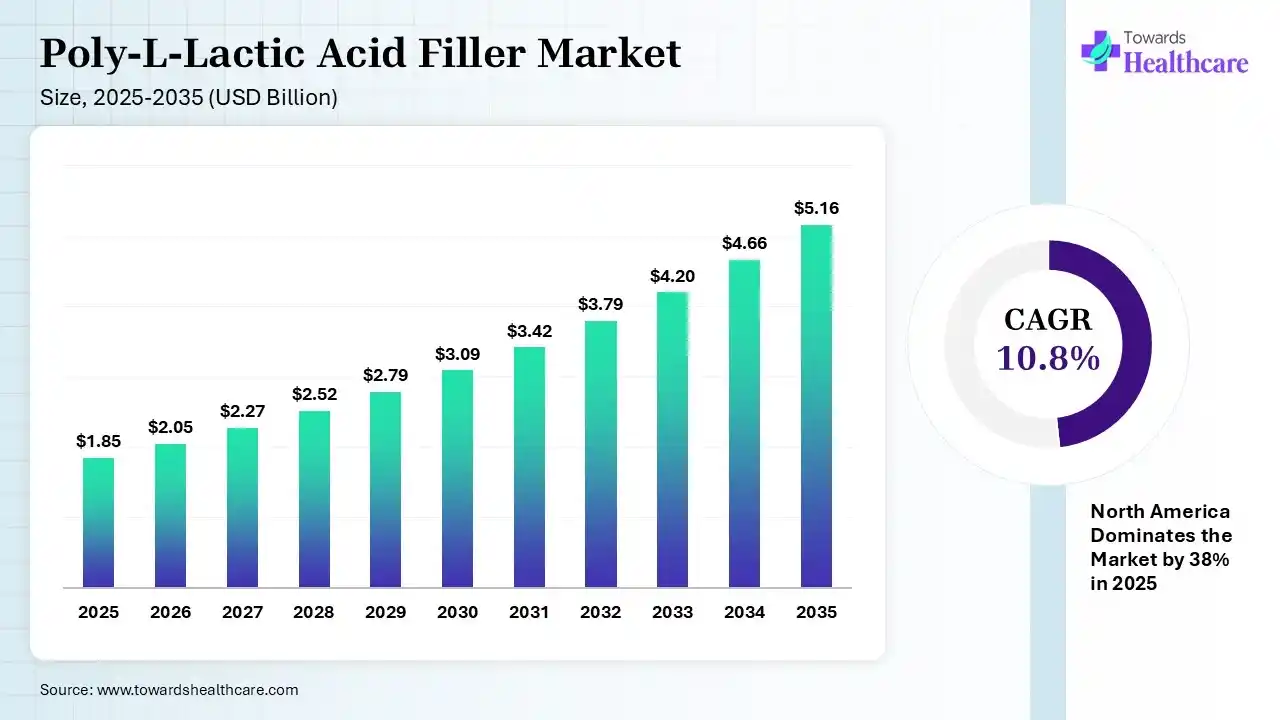
The global poly-l-lactic acid filler market size was estimated at USD 1.85 billion in 2025 and is predicted to increase from USD 2.05 billion in 2026 to approximately USD 5.16 billion by 2035, expanding at a CAGR of 10.8% from 2026 to 2035. The poly-l-lactic acid filler market is growing because it improves skin elasticity and firmness, lowering wrinkles, and encouraging tissue renewal and scar remodelling.
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
