Top Vendors and Their Offerings in the Pre-Clinical Scientific Research Medical Device Market

| Companies | Headquarters | Pre-clinical Scientific Research Medical Device Services |
| Charles Rive Laboratories | Massachusetts, U.S. | Preclinical research and safety assessments |
| Eurofins Scientific | Luxembourg, Europe | Advanced analytical testing |
| WuXi AppTec | Shanghai, China | Integrated R&D services |
| SGS SA | Geneva, Switzerland | Multidisciplinary validation and high-quality control |
| ICON plc | Dublin, Ireland | Comprehensive preclinical consulting and automated clinical research solutions |
| IQVIA | Durham, North Carolina | AI and advanced analytics |
| NAMSA | Northwood, Ohio | End-to-end medical device development services |
| Intertek Group | London, UK | Mechanical and electrical testing |
| Medpace | Cincinnati, Ohio | Integrated medical expertise and regulatory guidance |
| Veranex | Raleigh, North Carolina | Clinical and regulatory expertise |
Market Value Chain Analysis
R&D
- The R&D of the pre-clinical scientific research medical device focuses on the integration of AI and machine learning and the development of advanced biological models.
- Key players: Charles Rive Laboratories, Eurofins Scientific, SGS SA.
Clinical Trials and Regulatory Approvals
- The biological performance, safety, and effectiveness are evaluated in the clinical trials and regulatory approvals of the pre-clinical scientific research medical device.
- Key players: SGS, NAMSA, Eurofins Scientific.
Patient Support and Services
- Home-based clinical support, digital engagement platforms, and travel and lodging concierge services are provided in the patient support and services of the pre-clinical scientific research medical device.
- Key players: IQVIA, ProPharma, ICON plc.
Market Growth
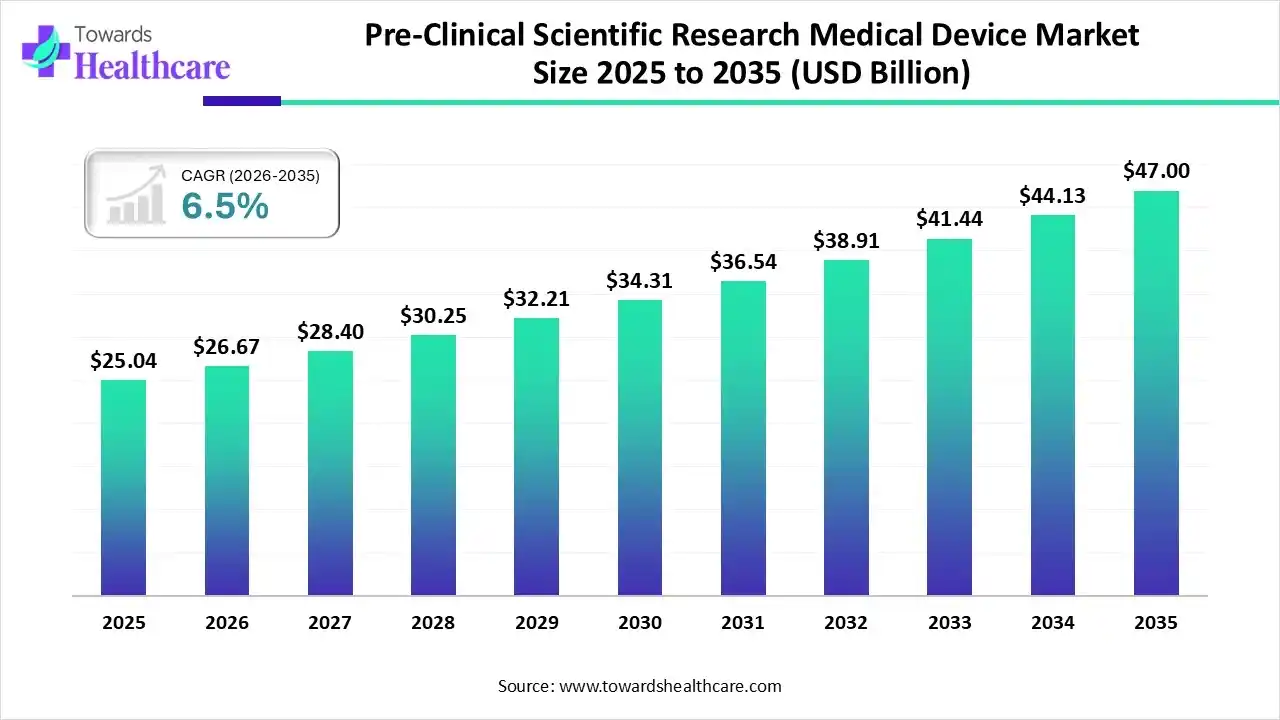
The global pre-clinical scientific research medical device market size was estimated at USD 25.04 billion in 2025 and is predicted to increase from USD 26.67 billion in 2026 to approximately USD 47 billion by 2035, expanding at a CAGR of 6.5% from 2026 to 2035.
What are the Recent Developments in the Market?
- In January 2026, a successful acquisition of Labcrop, offering innovative and comprehensive laboratory services, by NAMSA, which us leader in medical device testing, preclinical and clinical services, and regulatory consulting, was announced, where the Early Development medical device testing business of Labcrop will support the core areas of preclinical drug development and chemical testing of NAMSA.
- In July 2025, with a strategic supply and licensing agreement between MH3D and the Science and Technology Facilities Council (STFC) of the UK, the first MH3D systems were launched. This collaboration enables the integration of the next-generation imaging platforms of MH3D with the hyperspectral imaging technology of STFC to offer pre-clinical research applications.
Your complete Pre-Clinical Scientific Research Medical Device Market control center is just one click away - Access the Dashboard
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
