April 2026

The global pre-clinical scientific research medical device market size was estimated at USD 25.04 billion in 2025 and is predicted to increase from USD 26.67 billion in 2026 to approximately USD 47 billion by 2035, expanding at a CAGR of 6.5% from 2026 to 2035.
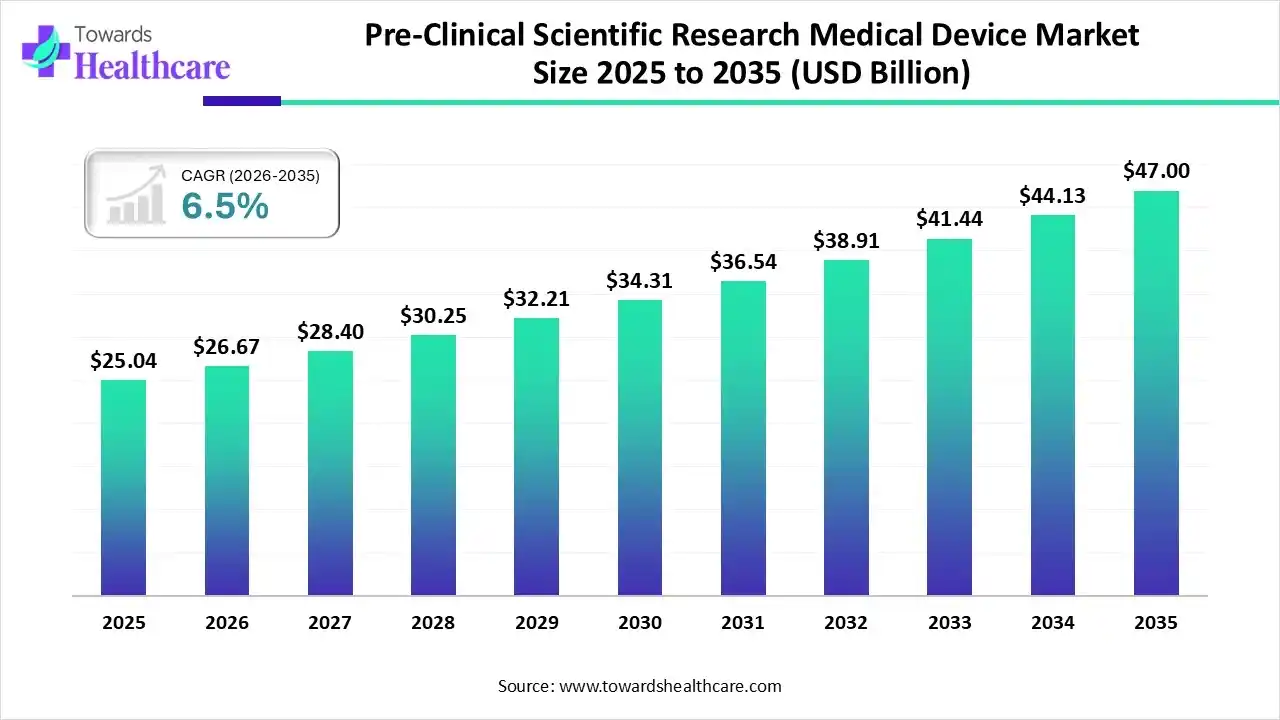
The growing chronic diseases and stringent regulations are increasing the pre-clinical scientific research on medical devices. Additionally, the growing AI integration, technological advancements, R&D activities, and launches are promoting the market growth.
The pre-clinical scientific research medical device market is driven by a surge in the innovative device pipeline. The pre-clinical scientific research medical device encompasses the non-human and laboratory testing of the device, which are conducted before its clinical trials in humans. They are further used for the safety assessment, performance evaluation, biocompatibility testing, design optimization, and risk assessments.
The impact of AI in the pre-clinical scientific research medical device market is increasing as it offers predictive modelling, enhancing the device safety and performance. It is also used in automated image evaluation and data management, and the development of digital models to identify the interactions between devices and organs. Its use in the design optimization, risk assessment, and decision-making is also increasing, promoting a shift from animal testing to virtual experiments.
The growing, stringent regulations are increasing the complexities associated with the development of medical devices, which are driving the demand for pre-clinical scientific research services.
The growing advancements, regulatory hurdles, and funding are increasing the shift towards the outsourcing of medical devices, leading to new collaborations to leverage various pre-clinical scientific research medical device services.
A rise in the disease burden is increasing the development of new medical devices, such as 3D-printed implants, smart and wearable medical devices, which are creating new opportunities, driving the demand for their preclinical testing services.
| Table | Scope |
| Market Size in 2026 | USD 25.04 Billion |
| Projected Market Size in 2035 | USD 26.67 Billion |
| CAGR (2026 - 2035) | 6.5% |
| Leading Region | North America |
| Historical Data | 2020 - 2023 |
| Base Year | 2025 |
| Forecast Period | 2026 - 2035 |
| Measurable Values | USD Millions/Units/Volume |
| Market Segmentation | By Product Type, By Service Type (Testing & Validation), By Application, By End-User, By Region |
| Top Key Players | Charles Rive Laboratories, Eurofins Scientific, WuXi AppTec, SGS SA, ICON plc, IQVIA, NAMSA, Intertek Group, Medpace, Veranex |
Which Product Type Dominated in the Market in 2025?
The pre-clinical in-vitro diagnostic (IVD) systems & reagents segment held the largest revenue share of the pre-clinical scientific research medical device market in 2025, due to its critical role in biomarker identification and safety evaluation. They were also used in disease detection, drug development, and personalized medicine development.
Animal Patient Monitoring & Life Support Systems
The animal patient monitoring & life support systems segment is expected to expand rapidly during the predicted time, due to the growing preclinical studies complexities. Moreover, expanding R&D activities, pharmacological studies, and technological innovations are also increasing their adoption rates.
How Biocompatibility Testing Segment Dominated the Market in 2025?
The biocompatibility testing segment contributed the biggest revenue share of the pre-clinical scientific research medical device market in 2025, due to mandatory safety regulations for all implantable devices. The growing development of surgical devices, new polymer combinations, and nanomaterials is also increasing their demand.
Chemistry & Toxicology Testing
The chemistry & toxicology testing segment is expected to grow at the fastest CAGR of approximate 9.7% during the predicted time, as chemical characterization is driving the replacement of the long-term animal studies. Additionally, the increasing development of biologics and advanced therapies is also increasing their demand for safety assessments.
Which Application Type Segment Held the Dominating Share of the Market in 2025?
The oncology segment held the dominating share of the pre-clinical scientific research medical device market in 2025, due to a growth in the R&D focused on cancer therapeutics and diagnostic imaging. The growth in the cancer incidence rates and government funding also promoted the development of advanced medical devices and biomarker preclinical research.
Neurology
The neurology segment is expected to show the highest growth during the upcoming period, which is fueled by research into neurodegenerative diseases and brain-computer interfaces. The growing technological innovations are also driving the development of new per clinical models, leading to new collaborations among the industries.
What Made Medical Device Manufacturers the Dominant Segment in the Market in 2025?
The medical device manufacturers segment led the pre-clinical scientific research medical device market in 2025, driven by the highest volume of internal and outsourced R&D. The growth in new product development also increased their pre-clinical scientific research of medical devices. Advancements in precision tools and testing services also contributed to their increased use.
Contract Research Organizations (CROs)
The contract research organizations (CROs) segment is expected to witness the fastest growth rate during the upcoming period, due to manufacturers' shift toward flexible, cost-effective outsourcing models. The expansion of the pharma and biotech startup are also increasing pre-clinical scientific research of medical devices, driving the development of new service models.
North America dominated the pre-clinical scientific research medical device market in 2025, due to the growth in the R&D investments, which increased the R&D activities focused on the medical device development, leading to a growth in the demand for pre-clinical services. The presence of stringent regulations also contributed to the market growth.
The growing R&D investments and presence of robust industries are increasing the preclinical scientific medical device R&D activities. The growing, stringent regulations and technological advancements are also increasing the development of new automated preclinical systems, increasing their adoption rates.
Asia Pacific is expected to host the fastest-growing pre-clinical scientific research medical device market during the forecast period, due to the rapid expansion of the pharmaceutical and biotechnology industries, which is driving the demand for medical device pre-clinical research capabilities. The growing CRO infrastructure is also enhancing the market growth.
China Market Trends
China is experiencing an expansion in the pharmaceutical and biotechnology R&D infrastructure, which is increasing the pre-clinical scientific research of medical devices, novel therapies, and advanced technologies. Moreover, growing government initiatives and outsourcing trends are also increasing the demand for preclinical services.
Europe is expected to grow significantly in the pre-clinical scientific research medical device market during the forecast period, due to the presence of stringent regulations, which are increasing the demand for medical devices' preclinical testing. The robust R&D infrastructure is also increasing the development of new preclinical systems, promoting market growth.
UK Market Trends
The UK consists of well-developed research institutes and industries, driving the development of medical devices. This, in turn, is increasing the demand for their preclinical testing services, where the outsourcing trends, healthcare investments, and stringent regulations are also encouraging their demand.

| Companies | Headquarters | Pre-clinical Scientific Research Medical Device Services |
| Charles Rive Laboratories | Massachusetts, U.S. | Preclinical research and safety assessments |
| Eurofins Scientific | Luxembourg, Europe | Advanced analytical testing |
| WuXi AppTec | Shanghai, China | Integrated R&D services |
| SGS SA | Geneva, Switzerland | Multidisciplinary validation and high-quality control |
| ICON plc | Dublin, Ireland | Comprehensive preclinical consulting and automated clinical research solutions |
| IQVIA | Durham, North Carolina | AI and advanced analytics |
| NAMSA | Northwood, Ohio | End-to-end medical device development services |
| Intertek Group | London, UK | Mechanical and electrical testing |
| Medpace | Cincinnati, Ohio | Integrated medical expertise and regulatory guidance |
| Veranex | Raleigh, North Carolina | Clinical and regulatory expertise |
By Product Type
By Service Type (Testing & Validation)
By Application
By End-User
By Region
April 2026
March 2026
March 2026
March 2026