Latest Updates of Companies in the Rare Disease-Focused Cell & Gene Therapy CDMO Market

| Company | Headquarters | Latest Update |
| AGC Biologics | United States | This company focuses on both cell and viral vector therapies, with noteworthy experience in accelerating therapies. |
| WuXi Advanced Therapies | United States | This leading player provides specialized services for CAR-T, gene-edited therapies, and viral vector stages. |
| Thermo Fisher Scientific Inc. | United States | In April 2026, Thermo Fisher Scientific Inc., the world leader in serving science, announced that it had signed a definitive agreement to sell its microbiology business to Astorg, a leading pan-European private equity organization. |
| FUJIFILM Diosynth Biotechnologies | United States | FUJIFILM Diosynth Biotechnologies is a CDMO Partner for Life, providing end-to-end advancement and manufacturing services. |
| Catalent, Inc. | United States | Catalent Cell & Gene Therapy is an industry-leading Contract Development and Manufacturing Organization (CDMO) for better therapeutics. |
Recent Developments in the Rare Disease-Focused Cell & Gene Therapy CDMO Market
- In April 2026, Andelyn Biosciences, a leading and patient-focused cell and gene therapy Contract Development and Manufacturing Organization, and ENCell Co., Ltd., a Korea-driven advanced biopharmaceutical CDMO, announced the signing of a Collaboration Agreement to establish a strategic manufacturing bridge between the United States and Asia-Pacific (APAC) regions.
- In January 2026, RoslinCT and BOOST Pharma announced a strategic manufacturing collaboration to support the advancement of BOOST Pharma’s cell therapy, BT-101, for the treatment of infants with Osteogenesis Imperfecta (OI), also known as brittle bone disease.
Supply Chain Analysis
R&D:
- Research and development (R&D) in RNA interference therapy includes process development and optimization, analytical development and characterization, preclinical and IND-enabling studies, regulatory and strategy alignment, and transition to clinical manufacturing
- Key Players: WuXi Advanced Therapies and AGC Biologics
Manufacturing Processes:
- Manufacturing processes in rare disease-focused cell & gene therapy CDMOs are dedicated, agile workflows tailored to small batch sizes, surrounding cell isolation, expansion, and genetic modification.
- Key Players: FUJIFILM Diosynth Biotechnologies and Thermo Fisher Scientific Inc.
Patient Services:
- Patient services majorly focus on the specialized, often autologous (patient-driven) logistics needed for small patient populations.
- Key Players: Lonza and Catalent, Inc.
Market Growth
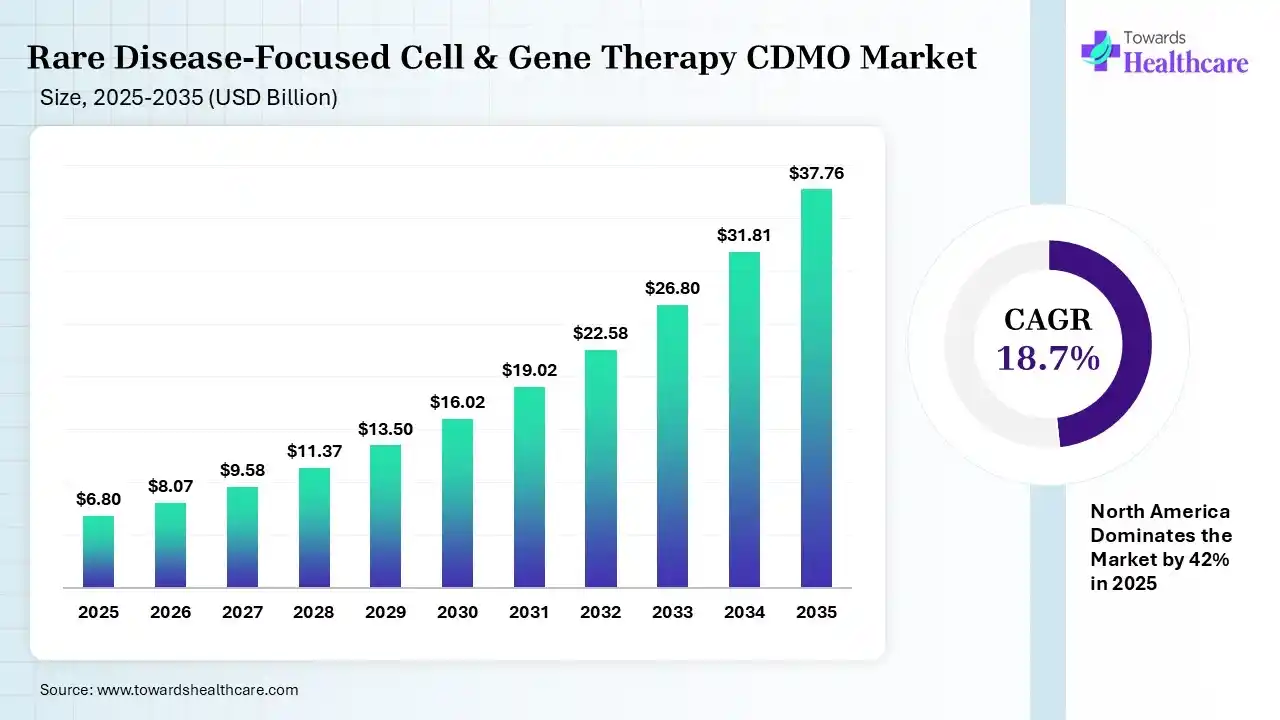
The global rare disease-focused cell & gene therapy CDMO market size was estimated at USD 6.8 billion in 2025 and is predicted to increase from USD 8.07 billion in 2026 to approximately USD 37.76 billion by 2035, expanding at a CAGR of 18.7% from 2026 to 2035.
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
