April 2026

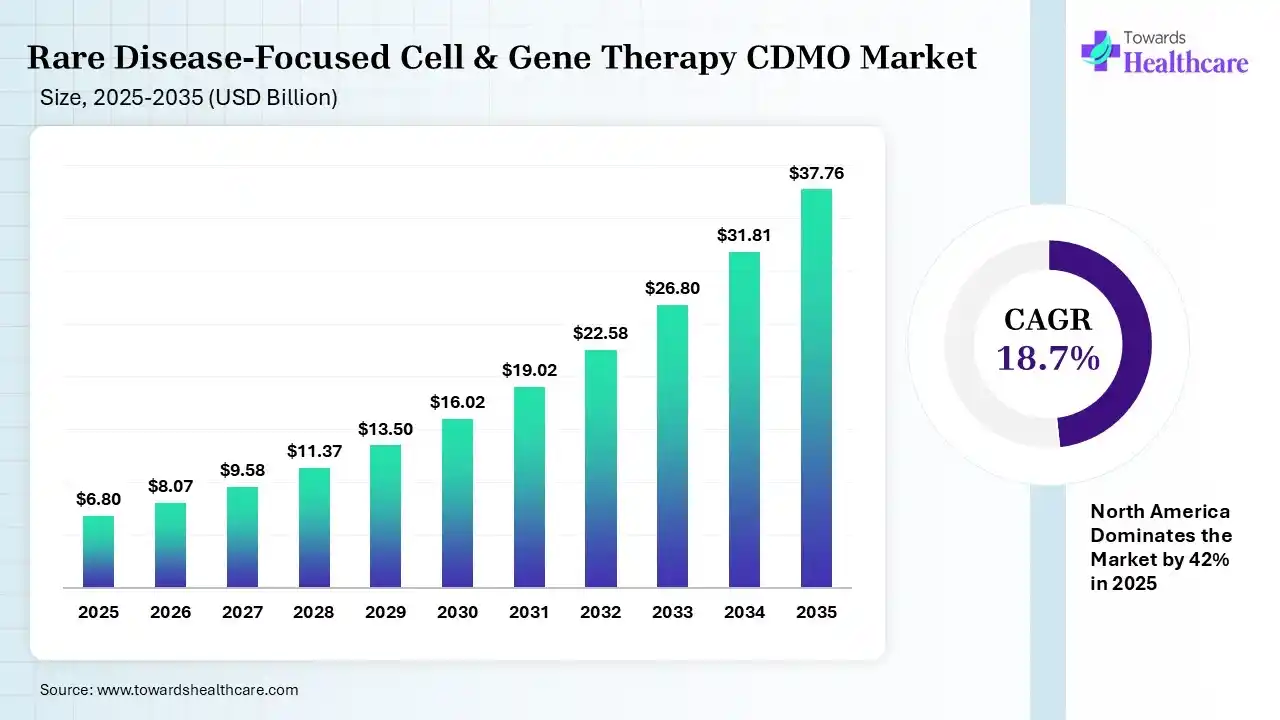
The global rare disease-focused cell & gene therapy CDMO market size was estimated at USD 6.8 billion in 2025 and is predicted to increase from USD 8.07 billion in 2026 to approximately USD 37.76 billion by 2035, expanding at a CAGR of 18.7% from 2026 to 2035. The rare disease-focused cell & gene therapy CDMO market is growing because CDMOs provide an affordable service by offering access to specialized expertise and infrastructure without requiring companies to make significant capital spending.
Cell and gene therapy CDMOs work as a specialised delay of a biopharma’s team, giving their knowledge, amenities, and tools to handle significant parts of C> advancement and manufacturing. CDMO efficiently manages materials, labor, operations, and quality, output, and cost control for drug developers. Cell and gene therapy CDMO technologies are progressively recognized as vital services for biopharmaceutical companies, providing precise viral vector production abilities, government compliance assurance, and inclusive manufacturing characteristics. Cell and gene therapies (CAGTs) hold promise for transforming the treatment of major diseases.
Artificial intelligence and automation technology have recently transformed cell therapy manufacturing via their integration, which allows better advancement and distribution of targeted treatments. AI-based insights, automation, and advanced analytics are reshaping this landscape to speed up discovery, optimizing clinical trials, and streamlining production. AI-based technology creates virtual, high-fidelity replicas of bioreactors to simulate cell growth and optimize parameters in earlier physical manufacturing. AI-based augments data extraction and offer unique insights into the disease mechanisms, like new symptom clusters or subtle features that deviate from population norms. AI-based technology supports genomicists in interrogating differences in data quality and bioinformatic strategy.
| Table | Scope |
| Market Size in 2026 | USD 8.07 Billion |
| Projected Market Size in 2035 | USD 37.76 Billion |
| CAGR (2026 - 2035) | 18.7% |
| Leading Region | North America by 42% |
| Historical Data | 2020 - 2023 |
| Base Year | 2025 |
| Forecast Period | 2026 - 2035 |
| Measurable Values | USD Millions/Units/Volume |
| Market Segmentation | By Service Type, By Therapy Type, By Indication, By End User, By Scale of Operation, By Region |
| Top Key Players | AGC Biologics, WuXi Advanced Therapies, Thermo Fisher Scientific Inc., FUJIFILM Diosynth Biotechnologies, Catalent, Inc. |
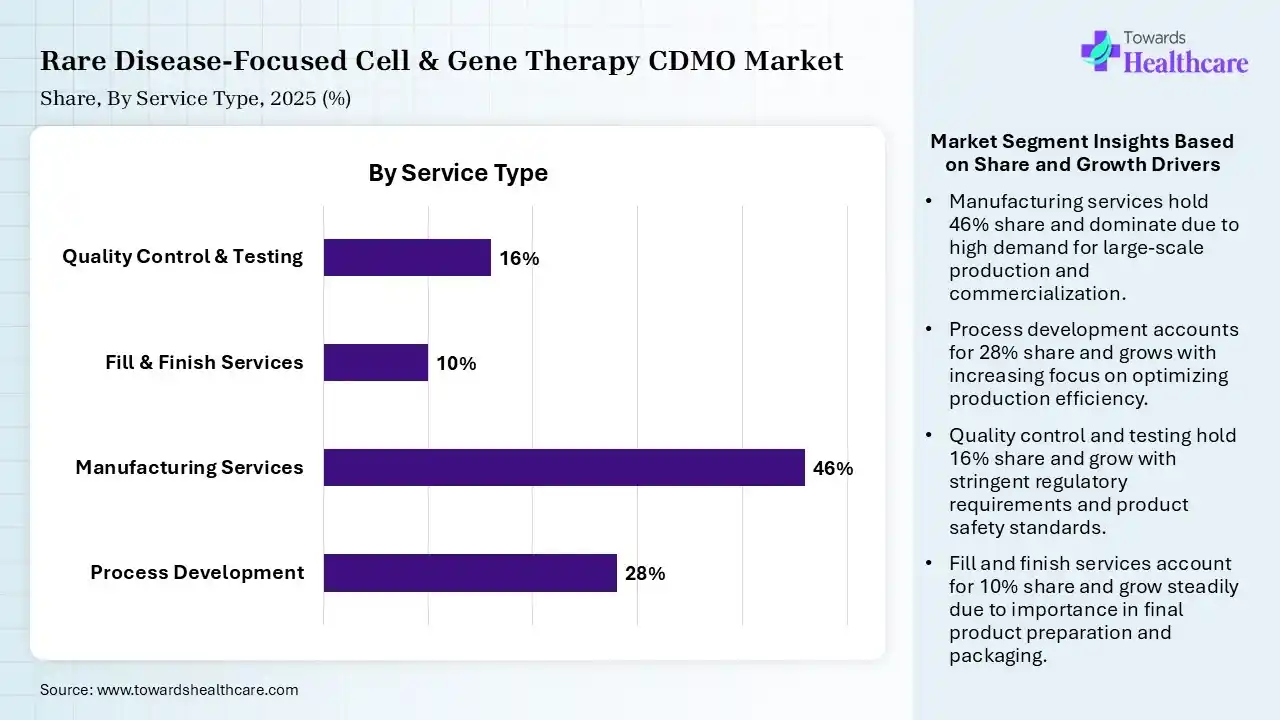
| Segment | Share 2025 (%) |
| Process Development | 28% |
| Manufacturing Services | 46% |
| Fill & Finish Services | 10% |
| Quality Control & Testing | 16% |
The Manufacturing Services Segment Led the Rare Disease-Focused Cell & Gene Therapy CDMO Market in 2025
The manufacturing services segment contributed the largest market share of 46% in 2025, as manufacturing facilities provided by CDMOs offer collaboration, flexibility, and innovation solutions that speed up drug development and reduce costs. CDMOs offer end-to-end support for pharmaceutical organizations, involving both research and manufacturing technology. Significant organization seeking flexibility, consistency, and expertise in the manufacturing process.
The process development segment held a significant share of 28% in the market, as a CDMO helps multifaceted activities involving active pharmaceutical ingredient (API) advancement, formulation development, government compliance, clinical trial management, process advancement, upscaling, and commercial manufacturing. The significance of CDMOs in healthcare is further highlighted by their momentous participation in both the creation and production of medical products.
The quality control & testing segment held a significant share of 16% in the rare disease-focused cell & gene therapy CDMO market, expected to grow at the fastest CAGR during the forecast period as CDMOs streamline the manufacturing and quality control technology, ensuring that products not only meet government compliance standards rapidly. CDMOs lessen production and supply chain challenges by employing their deep expertise and robust quality control systems.
The fill & finish services segment held a significant share of 10% in the market, as fill and finish services support in expediting the production timeline. Their streamlined processes, inclusive quality control measures, and experienced team allow a faster turnaround time, lowering inclusive project timelines.
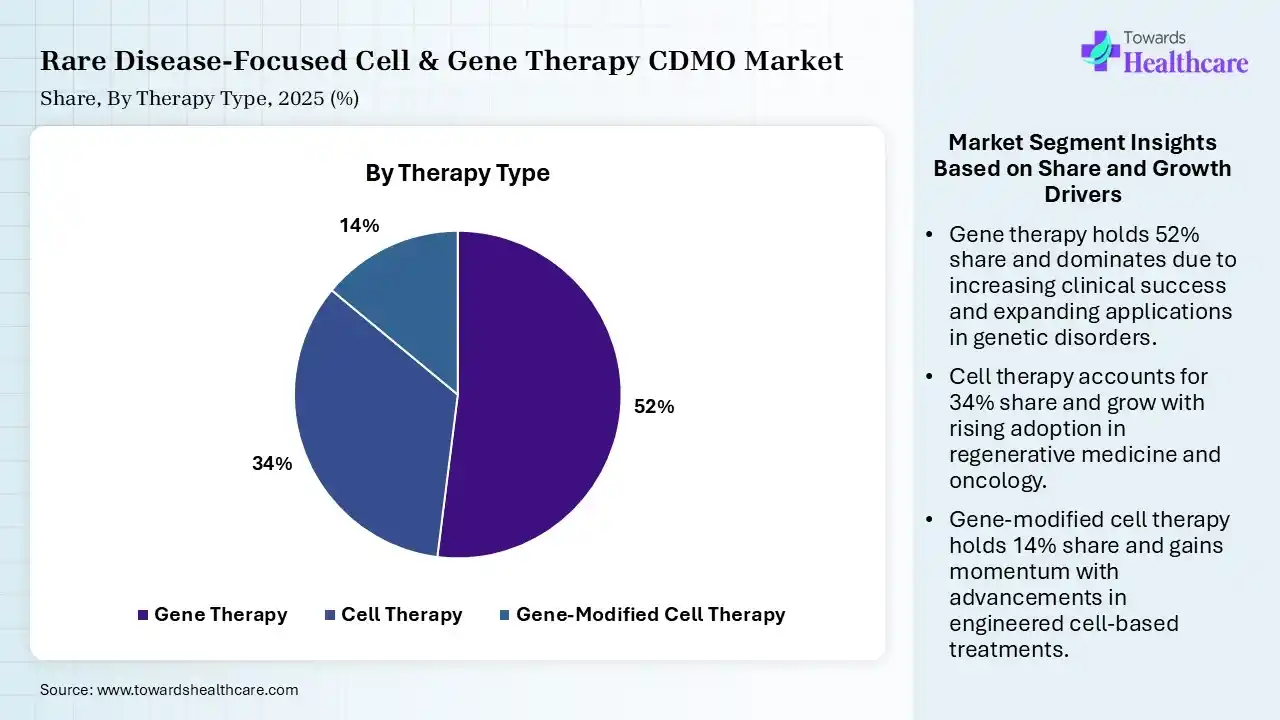
| Segment | Share 2025 (%) |
| Gene Therapy | 52% |
| Cell Therapy | 34% |
| Gene-Modified Cell Therapy | 14% |
Gene Therapy Segment Led the Rare Disease-Focused Cell & Gene Therapy CDMO Market in 2025
The gene therapy segment contributed the largest market share of 52%, as gene therapy is specifically promising because major rare diseases are caused by a single genetic mutation. Gene therapy is life-saving for some people with particular healthcare conditions, but it's expensive and causes adverse effects. Cell and gene therapies hold significant promise for managing rare diseases by targeting a disease's genetic roots.
The cell therapy segment held a significant share of 34% of the market, and is expected to grow at the fastest CAGR during the forecast period, which promotes the repair response in diseased, dysfunctional, or injured tissue using stem cells or their derivatives. Cell-driven therapy, particularly stem cells, offers novel hope for patients suffering from incurable diseases where the management strategy focuses on managing the disease, not treating it.
The gene-modified cell therapy segment held a significant share of 14% of the market, expected to grow at the fastest CAGR during the forecast period. It holds potential as a treatment for a broad range of diseases, like cystic fibrosis, cancer, heart disease, diabetes, hemophilia, and AIDS. Gene therapy is which continuing gene expression allows for a cure following a single treatment instead of ongoing administration of a drug with a comparatively short half-life.
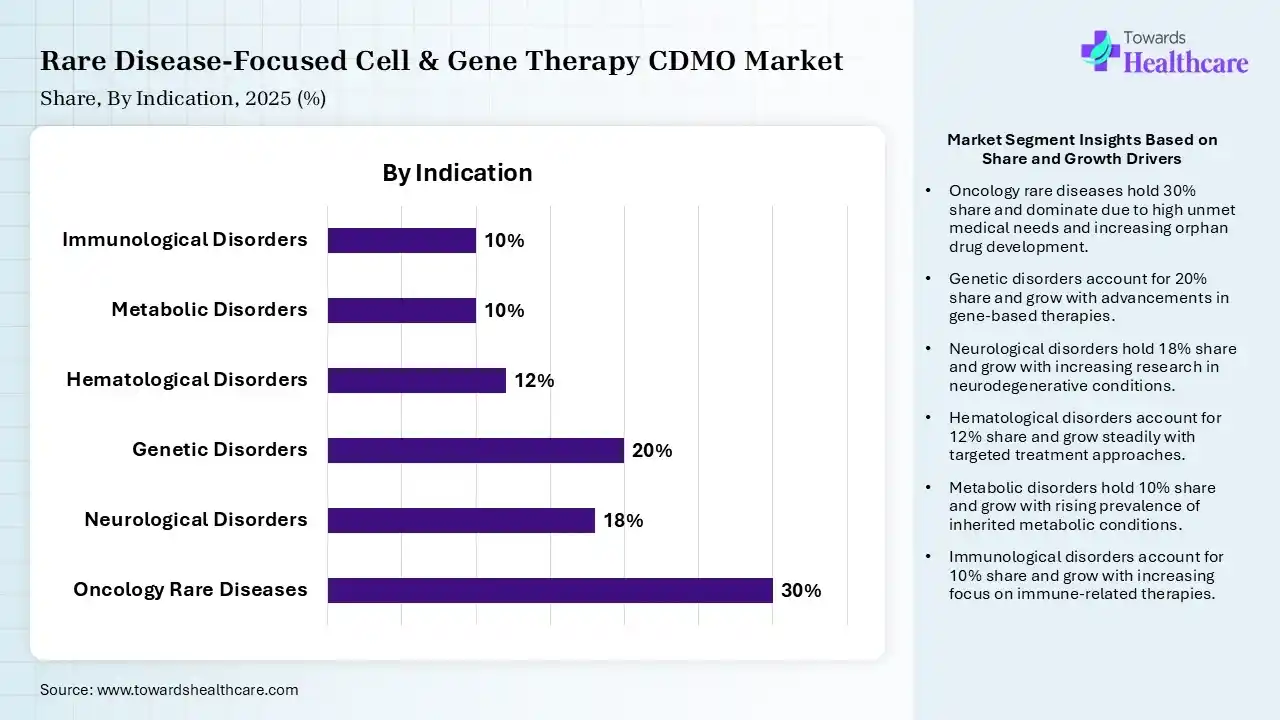
| Segment | Share 2025 (%) |
| Oncology Rare Diseases | 30% |
| Neurological Disorders | 18% |
| Genetic Disorders | 20% |
| Hematological Disorders | 12% |
| Metabolic Disorders | 10% |
| Immunological Disorders | 10% |
Oncology Rare Diseases Segment Led the Rare Disease-Focused Cell & Gene Therapy CDMO Market in 2025
The oncology rare diseases segment contributed the largest market share of 30%, as oncology drug advancement with an experienced oncology CDMO providing integrated manufacturing, in-house testing, and proven regulatory success. Oncology CDMOs speed up cancer drug development with advanced manufacturing, targeted therapies, and worldwide compliance proficiency.
The genetic disorders segment held a significant share of 20% in the market, as expected to grow at the fastest CAGR during the forecast period. Partnering with a CDMO offers access to scientific expertise, industrial facility capacity, navigation via regulatory requirements, and earlier speed. CDMOs often offer advanced analytical technology for characterizing multifaceted genetic products.
The neurological disorders segment held a significant share of 18% in the market, as gene therapy and stem cell therapy, biotechnology provides other novel services to address the challenges of neurological disorders. Gene therapy introduces the potential to correct or modulate disease-causing genetic alterations.
The hematological disorders segment held a significant share of 12% in the market, as cell and gene therapy is a promising treatment for various inherited and acquired hematologic disorders. Gene therapy includes the introduction of a functional gene to replace a mutated gene or a therapeutic gene to offer an absent or defective protein to the organism.
The metabolic disorders segment held a significant share of 10% in the rare disease-focused cell & gene therapy CDMO market, and is expected to grow at the fastest CAGR during the forecast period, as gene therapy has presently shown great promise as an effective treatment for several metabolic diseases caused by genetic faults. A CDMO efficiently manages labor, materials, operations, and quality, resulting in better expense control for drug developers.
The immunological disorders segment held a significant share of 10% in the market, as gene- and cell-driven immunotherapies intended to promote immune tolerance or induce a desired response against cancer or vaccine antigens. Cell therapies, despite their transformative strength, face significant challenges that hinder their advancement and commercialization. A CDMO efficiently manages labor, materials, operations, and quality, resulting in better cost control for the drug producer.
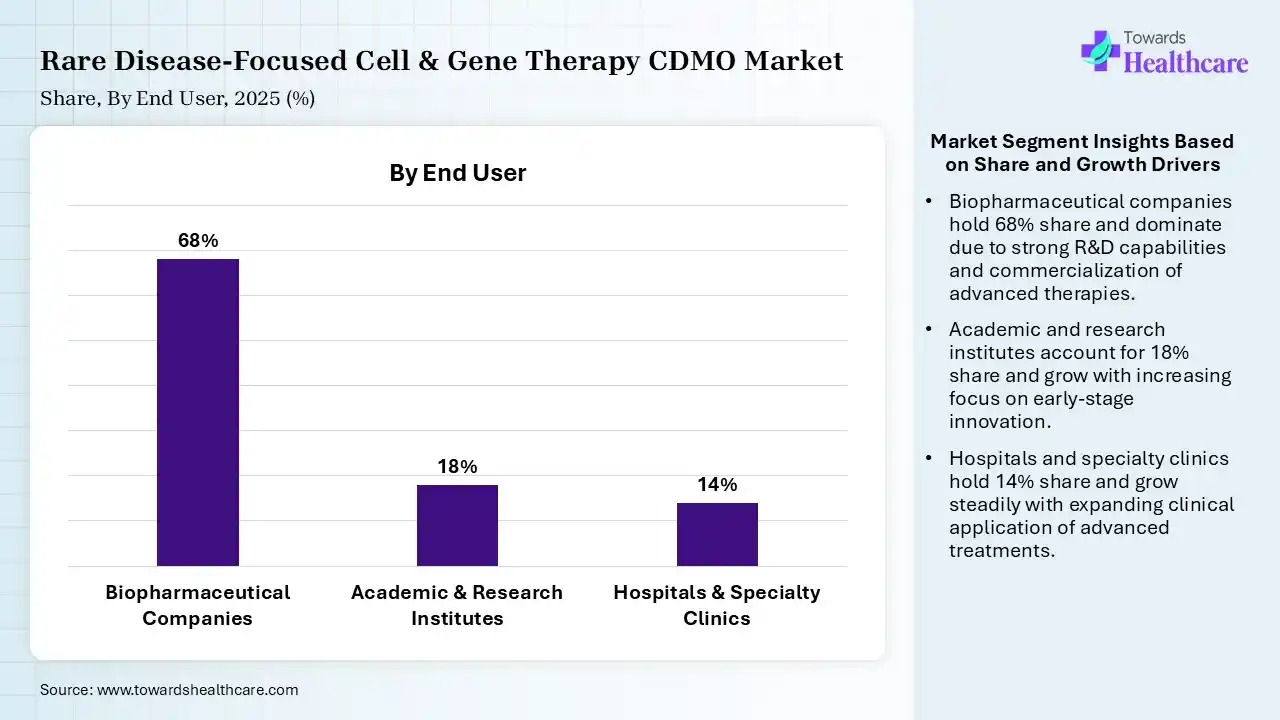
| Segment | Share 2025 (%) |
| Biopharmaceutical Companies | 68% |
| Academic & Research Institutes | 18% |
| Hospitals & Specialty Clinics | 14% |
Biopharmaceutical Companies Segment Led the Rare Disease-Focused Cell & Gene Therapy CDMO Market in 2025
The biopharmaceutical companies segment contributed the largest market share of 68%, as biopharma organizations use genetic material to create genomic, gene, cell, and gene editing management, which repair genetic mutations or present functioning genes to take the place of or inhibit the changed gene. CDMOs provide platform-based production, simplified tech transmissions, AI-based automation, and robust analytical solutions.
The academic & research institutes segment held a significant share of 18% in the market, as these CDMOs offer specialised knowledge in multifaceted technology like cell culturing, the generation of gene editing, and viral vectors. Cell Therapy CDMO services play a significant role in pharmaceutical organizations and research institutes by offering expertise in the development, optimization, and production of cell-driven therapies.
The hospitals & specialty clinics segment held a significant share of 14% in the rare disease-focused cell & gene therapy CDMO market, and is expected to grow at the fastest CAGR during the forecast period, as a CDMO efficiently manages labor, operations, materials, and quality, output in advance cost control for drug developers. Physicians have direct control over the production process, enabling treatments to be tailored particularly to the patient’s instant profile.
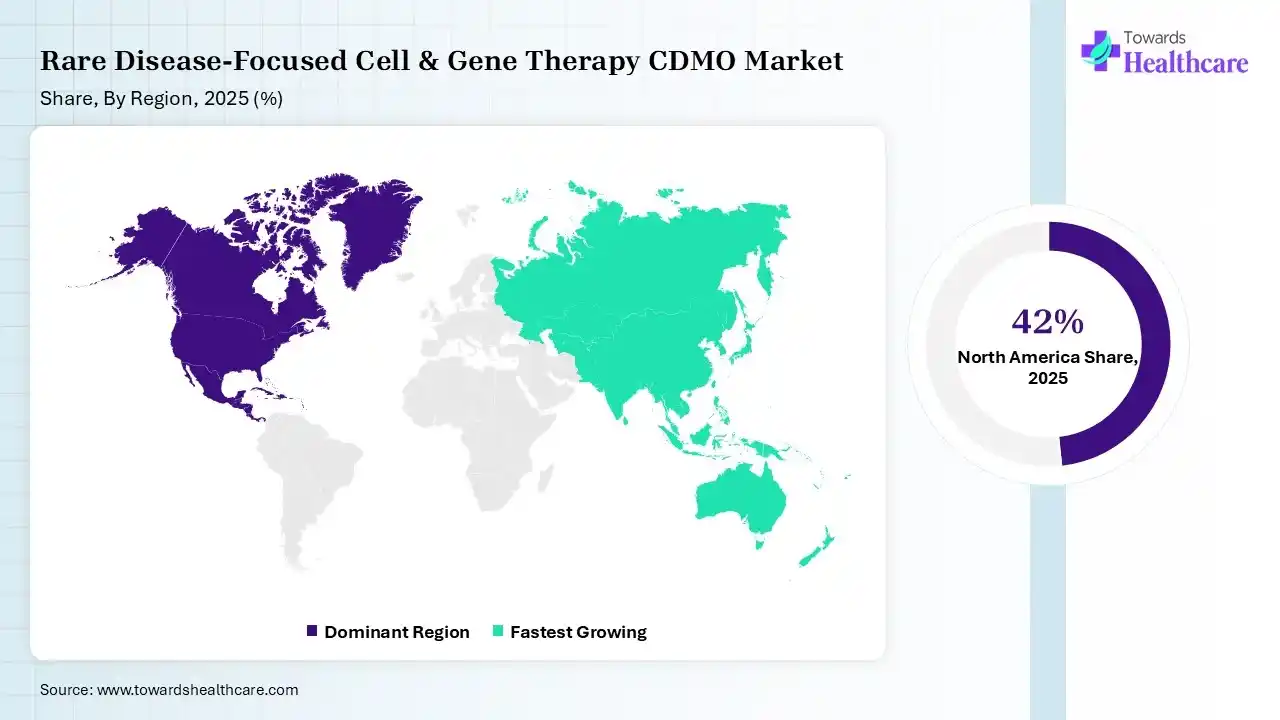
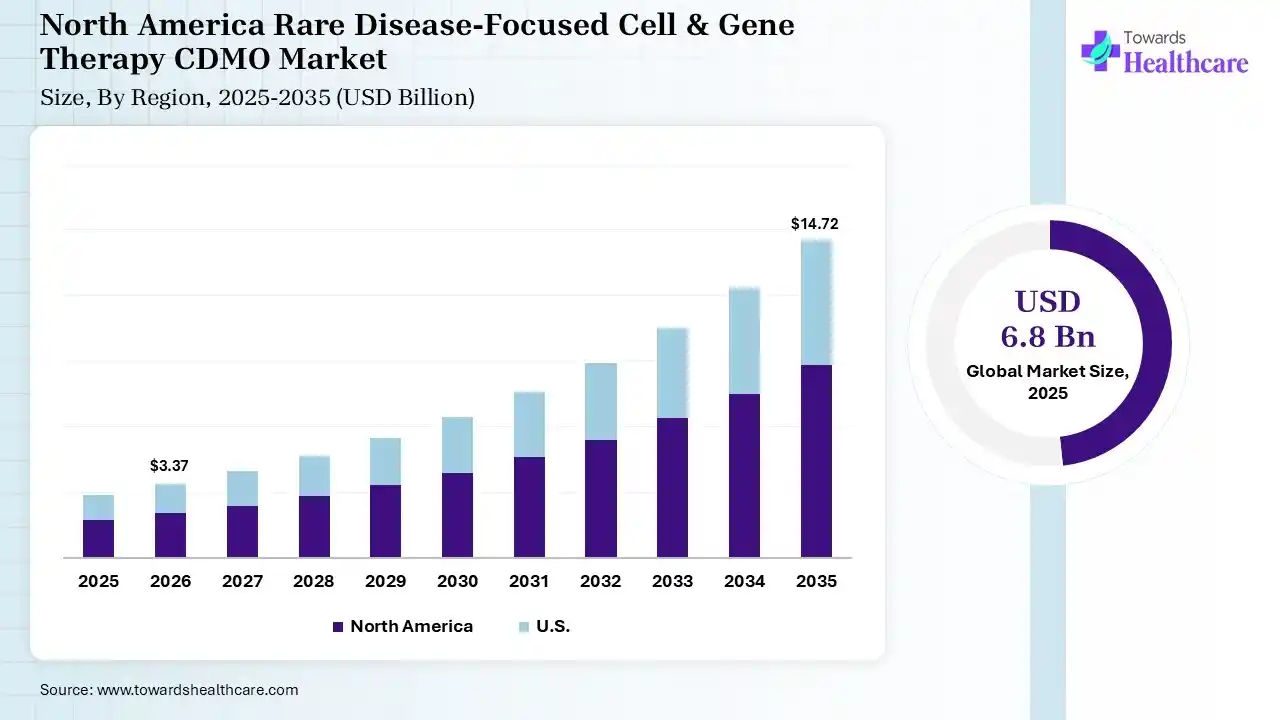
In 2025, North America dominated the rare disease-focused cell & gene therapy CDMO market with a share of 42% in 2025, due to the regenerative medicine advanced therapy designation (RMAT), which shows a specialized FDA pathway intended particularly for advanced regenerative medicine therapies. Private equity (PE) and venture capital (VC) organizations play a significant role in driving research and development (R&D) efforts in different industries. Gene therapy has transitioned from a long-awaited promise to a clinical reality, providing transformative treatments for rare congenital diseases and various cancers, which contributes to the growth of the market.
For Instance,
U.S. Market Trends
In the U.S., the FDA's approval of the first Wiskott-Aldrich gene therapy signals a shift in production and government standards for rare-disease CGTs. Manufacturers speed up innovation and enhance rare and ultra-rare disease therapy access by leveraging novel and established regulatory pathways. The U.S. dominates biotech spending, capturing nearly 55% of the entire worldwide spending.
Asia Pacific held 20% share of the rare disease-focused cell & gene therapy CDMO market, and is expected to have the fastest CAGR during the forecast period, due to the adoption of progressive technologies such as AI, machine learning, and automation, which enhance the CDMO solution, lowering drug development time and enhancing productivity. The Asia-Pacific area, home to 60% of the worldwide population, has the maximum number of Persons Living with a rare disease (PLWRD), which drives the growth of the market.
India Market Trends
Increasing India's CRDMO area from cost arbitrage to a revolution leader. An Indian pharma organization has strengthened its operations infrastructure to help its growth. India has a disproportionate load of genetic diseases, with over 70 million persons suffering from rare diseases. The Indian Council of Medical Research (ICMR) and the Department of Biotechnology (DBT) drive the growth of the market.
R&D:
Manufacturing Processes:
Patient Services:

| Company | Headquarters | Latest Update |
| AGC Biologics | United States | This company focuses on both cell and viral vector therapies, with noteworthy experience in accelerating therapies. |
| WuXi Advanced Therapies | United States | This leading player provides specialized services for CAR-T, gene-edited therapies, and viral vector stages. |
| Thermo Fisher Scientific Inc. | United States | In April 2026, Thermo Fisher Scientific Inc., the world leader in serving science, announced that it had signed a definitive agreement to sell its microbiology business to Astorg, a leading pan-European private equity organization. |
| FUJIFILM Diosynth Biotechnologies | United States | FUJIFILM Diosynth Biotechnologies is a CDMO Partner for Life, providing end-to-end advancement and manufacturing services. |
| Catalent, Inc. | United States | Catalent Cell & Gene Therapy is an industry-leading Contract Development and Manufacturing Organization (CDMO) for better therapeutics. |
Strengths
Weakness
Opportunities
Threat
By Service Type
By Therapy Type
By Indication
By End User
By Scale of Operation
By Region
April 2026
April 2026
April 2026
April 2026