Explore Respiratory Disease Vaccine Market Companies with SWOT Analysis
- Sanofi Pasteur
- Seqirus (CSL Group)
- Bharat Biotech
- Serum Institute of India
- Novavax
- Valneva
- Sinovac Biotech
- Sinopharm
- Bavarian Nordic
- Medicago, Inc.
- SK Bioscience
- Anhui Zhifei Longcom
Respiratory Disease Vaccine: Immunity for Breathing
The respiratory disease vaccine market is experiencing robust growth, driven by public-health vaccination programs, seasonal immunization campaigns, pandemic preparedness, technological innovations, and demographic aging. It comprises vaccines and vaccine delivery technologies developed to prevent or reduce the severity of infectious respiratory diseases, notably influenza, pneumococcal disease, respiratory syncytial virus (RSV), SARS-CoV-2 (COVID-19), pertussis (whooping cough), and other viral/bacterial respiratory pathogens. Includes vaccine development (platforms and formulations), manufacturing (bulk antigen, adjuvants), distribution & immunization programs, and related services (cold chain, surveillance).
Market Forecast
The global respiratory disease vaccine market size is calculated at USD 65.72 billion in 2025, grew to USD 68.43 billion in 2026, and is projected to reach around USD 98.41 billion by 2035. The market is expanding at a CAGR of 4.12% between 2026 and 2035.
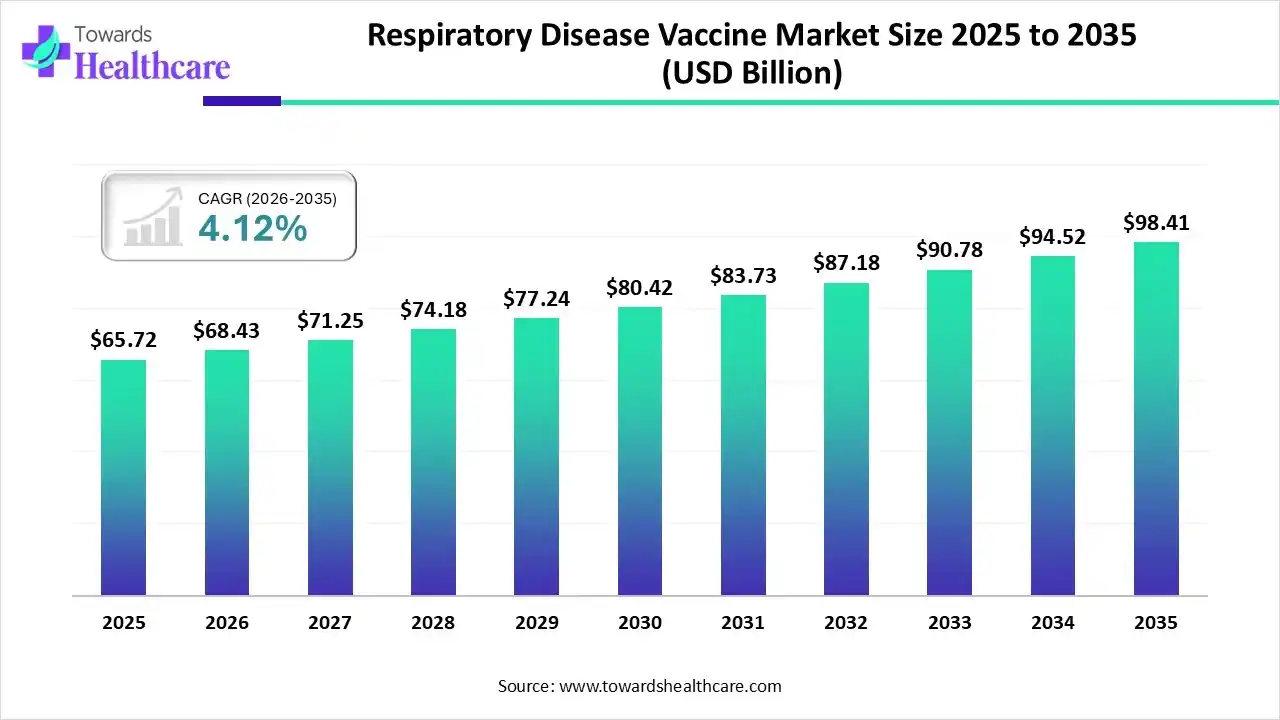
Respiratory Disease Vaccine Market Outlook
- Industry Growth Overview: The market is expected to grow rapidly in emerging markets, driven by the local manufacturing and tiered pricing influencing uptake. Advances in vaccine manufacturing, such as mRNA and recombinant protein, also boost market growth.
- Sustainability Trends: Key players are adopting sustainable practices in vaccine development, manufacturing, packaging, and supply chain. For instance, GSK aims to achieve a 90% reduction in carbon emissions by 2045 and to contribute to a more natural, positive world.
- Major Investors: Numerous government and private organizations provide funding for expanding vaccine manufacturing infrastructure and support the design of novel vaccines. TCGX, Goldman Sachs Alternatives, and Avoro Ventures are some private investors.
Company Landscape
Pfizer Inc.
Company Overview:
- A leading American multinational pharmaceutical and biotechnology corporation.
- Focuses on the discovery, development, manufacture, and commercialization of healthcare products, including medicines and vaccines, for humans.
- The company is globally recognized for its contribution to the COVID-19 vaccine response.
Corporate Information:
- Headquarters: New York City, New York, USA
- Year Founded: 1849
- Ownership Type: Public (NYSE: PFE)
History and Background:
- Founded by two German cousins, Charles Pfizer and Charles F. Erhart, as a chemical company.
- Initially produced fine chemicals, famously providing mass quantities of penicillin during World War II.
- Transformed into a research-driven pharmaceutical company over the 20th century through organic growth and major acquisitions.
Key Milestones/Timeline:
- 1849: Founded as Charles Pfizer & Company.
- 1950s: Expanded globally, establishing offices across the world.
- 2000s: Major acquisitions include Warner-Lambert, Pharmacia, and Wyeth.
- 2020: Joint development with BioNTech of the first mRNA-based COVID-19 vaccine authorized for emergency use in the US and EU.
- 2023: US FDA approval for ABRYSVO™ (RSV vaccine) for older adults and for pregnant individuals to prevent RSV in infants.
Business Overview:
- Operates across a wide range of therapeutic areas, including Vaccines, Oncology, Inflammation & Immunology, Rare Diseases, Internal Medicine, and Sterile Injectable Pharmaceuticals.
- The Vaccines segment is a key growth driver, heavily weighted toward pneumococcal and COVID-19/RSV vaccines.
Business Segments/Divisions:
Pfizer Biopharmaceuticals Group (Biopharma): Divided into six therapeutic areas/categories: Internal Medicine, Inflammation & Immunology, Oncology, Rare Disease, Hospital (Sterile Injectable Pharmaceuticals and Anti-Infectives), and Vaccines.
Geographic Presence:
- Sells products in more than 180 countries and territories globally.
- Major revenue sources include North America and Europe, with a growing presence in emerging markets.
Key Offerings (Respiratory Vaccines):
- Comirnaty (in partnership with BioNTech): mRNA COVID-19 vaccine.
- Prevnar Family (e.g., Prevnar 13, Prevnar 20/Vaxneuvance): Pneumococcal Conjugate Vaccines (PCV) for infants, children, and adults.
- ABRYSVO™: Respiratory Syncytial Virus (RSV) vaccine for older adults and maternal immunization.
End-Use Industries Served:
- Hospitals and Clinics
- Retail and Hospital Pharmacies
- Government Agencies/Public Health Programs
- Pediatric and Adult Patient Populations
Key Developments and Strategic Initiatives:
- Mergers & Acquisitions:
2022: Completed the acquisition of ReViral, a clinical-stage biopharmaceutical company focused on antiviral therapeutics for Respiratory Syncytial Virus (RSV).
- Partnerships & Collaborations:
Continues its collaboration with BioNTech for the development and commercialization of mRNA-based vaccines, including updated COVID-19 and combination vaccine candidates.
- Product Launches/Innovations:
Development of next-generation mRNA technology platforms to target multiple respiratory pathogens simultaneously (e.g., flu/COVID-19 combination vaccine candidates).
- Capacity Expansions/Investments:
Continues to invest heavily in its manufacturing network to support the global supply of its pneumococcal, COVID-19, and RSV vaccines.
- Regulatory Approvals:
April 2025: Announced the amendment in the marketing authorization for ABRYSVO (RSV vaccine) to include the extension of indication for the prevention of Lower Respiratory Tract Disease (LRTD) caused by RSV in new populations.
- Distribution Channel Strategy:
Multi-channel approach: Utilizes Hospital Pharmacies and Retail Pharmacies for adult and pediatric vaccinations, and Government Suppliers for national immunization programs globally.
- Technological Capabilities/R&D Focus:
- Core Technologies/Patents: mRNA technology, Conjugate Vaccine technology (for pneumococcal), and Prefusion F protein technology (for RSV).
- Research & Development Infrastructure: Global R&D centers in the US, Europe, and Asia. Focus on highly innovative, breakthrough therapies and vaccines.
- Innovation Focus Areas: Combination respiratory vaccines (e.g., Flu/COVID-19), next-generation pneumococcal vaccines, and expanded indications for RSV prevention.
Competitive Positioning:
- Strengths & Differentiators:
- Market leadership in the high-value Pneumococcal and COVID-19 vaccine segments.
- Early mover advantage in the new RSV adult and maternal vaccine market.
- Robust and flexible mRNA platform technology for rapid response to new pathogens.
- Market presence & ecosystem role: Global pharmaceutical powerhouse with established relationships with governments and healthcare providers worldwide, acting as a critical supplier for pandemic-response and routine immunization.
- SWOT Analysis:
- Strengths: Strong brand recognition; leading position in PCV, COVID-19, and new RSV markets; diversified pipeline; extensive global manufacturing and distribution.
- Weaknesses: High dependence on a few blockbuster products; recent revenue fluctuations post-COVID-19 vaccine peak.
- Opportunities: Expansion of RSV vaccine into new demographics; development of combination respiratory vaccines (e.g., Flu/COVID-19); geographic expansion in emerging markets.
- Threats: Patent expiry for key drugs; intense competition in the vaccine landscape (especially from GSK and Moderna); regulatory scrutiny.
Recent News and Updates:
Press Releases:
- August 2024: Reported positive top-line results from a Phase 3 trial evaluating the mRNA vaccine candidate against influenza and COVID-19.
- April 2025: Received an extended indication approval for ABRYSVO for certain populations.
Industry Recognitions/Awards:
Consistently recognized for leadership in vaccine development and manufacturing innovation.
GlaxoSmithKline (GSK) plc
Company Overview:
- A British multinational pharmaceutical and biotechnology company.
- One of the largest vaccine manufacturers globally, with a dedicated focus on the prevention and treatment of infectious and respiratory diseases.
- GSK's Vaccines division is a core part of its biopharma strategy.
Corporate Information:
- Headquarters: Brentford, London, United Kingdom
- Year Founded: 2000 (Formed by the merger of Glaxo Wellcome and SmithKline Beecham, tracing origins back to the 18th century)
- Ownership Type: Public (LSE: GSK, NYSE: GSK)
History and Background:
- A long history in the pharmaceutical industry through its predecessor companies.
- The merger in 2000 created one of the world's largest pharmaceutical companies.
- Has focused heavily on building a dominant Vaccines portfolio, which has become a key driver of the company's growth.
Key Milestones/Timeline:
- 2000: Formation of GSK through merger.
- 2015: Acquired Novartis’s vaccine business (excluding influenza).
- 2017: Launched Shingrix (non-respiratory, but a major vaccine success).
- 2022: Spun off its Consumer Healthcare division (Haleon) to focus purely on biopharma and vaccines.
- 2023: US FDA approval for Arexvy (RSV vaccine) for adults 60 years and older.
Business Overview:
- Focused biopharma company with R&D in four core therapeutic areas: Infectious Diseases, HIV, Oncology, and Immunology/Respiratory.
- Vaccines are managed under the Infectious Diseases and Respiratory/Immunology focus areas, contributing significantly to group revenue.
Business Segments/Divisions:
- Pharmaceuticals: Focuses on respiratory, HIV, oncology, and specialty medicines.
- Vaccines: Develops, manufactures, and distributes vaccines to prevent a wide range of infectious diseases, including major respiratory pathogens.
Geographic Presence:
- Operates in over 100 countries, with vaccines distributed to more than 170 countries.
- Significant presence in North America, Europe, and a long-standing commitment to lower-income countries through global health initiatives.
Key Offerings (Respiratory Vaccines):
- Arexvy: Respiratory Syncytial Virus (RSV) vaccine for adults 60 years and older.
- Fluarix/FluLaval/FluMist: Portfolio of Influenza vaccines (Quadrivalent/Nasal Spray).
- Bexsero: Meningococcal Group B vaccine (a major respiratory-linked disease).
- Synflorix: Pneumococcal Conjugate Vaccine (PCV) for infants and children (competes with Pfizer's Prevnar).
End-Use Industries Served:
- Public Health Systems and Government Immunization Programs (especially for pediatric and seasonal vaccines)
- Hospitals and Clinics
- Retail Pharmacies and Private Healthcare Providers
Key Developments and Strategic Initiatives:
- Mergers & Acquisitions:
2022: Acquired Affinivax Inc. to enhance its pneumococcal vaccine pipeline, including a next-generation 24-valent pneumococcal vaccine candidate (AFX3772).
- Partnerships & Collaborations:
June 2023: Announced funding agreement with the Bill & Melinda Gates Foundation and Wellcome to accelerate late-stage development of its tuberculosis vaccine candidate, M72 (TB is a major chronic respiratory disease).
- Product Launches/Innovations:
- Successful launch of Arexvy in the adult RSV market in 2023, a significant respiratory disease vaccine innovation.
- Focusing on developing next-generation pneumococcal and meningococcal vaccines.
Capacity Expansions/Investments:
Continuously investing in its global manufacturing network to meet high demand for its new and established vaccine products.
Regulatory Approvals:
April 2025: Announced that the CDC's Advisory Committee on Immunization Practices (ACIP) voted to endorse the inclusion of Penmenvy (Meningococcal Groups A, B, C, W, and Y Vaccine) in the adolescent meningococcal vaccination schedule.
Distribution Channel Strategy:
- Leverages strong government relationships for national and global immunization programs.
- Extensive network with hospitals and retail pharmacies for widespread administration of seasonal and adult vaccines (e.g., flu, RSV).
Technological Capabilities/R&D Focus:
- Core Technologies/Patents: Adjuvants (e.g., AS01E used in Arexvy and Shingrix), Conjugate Vaccine technology, and Recombinant protein technology.
- Research & Development Infrastructure: Focused R&D centers globally, with a strong emphasis on Infectious Diseases and the use of genetic data.
- Innovation Focus Areas: Next-generation bacterial vaccines (e.g., pneumococcal), RSV prevention, and new vaccine platforms for infectious diseases.
Competitive Positioning:
- Strengths & Differentiators:
- World-class adjuvant technology (AS01E) demonstrated in Shingrix and Arexvy.
- Diversified vaccine portfolio spanning pediatric to adult immunization.
- Deep expertise and long-standing presence in the respiratory and infectious disease areas.
- Market presence & ecosystem role: A core provider to global health organizations (like Gavi) and a dominant player in the global adult and pediatric vaccine market.
- SWOT Analysis:
- Strengths: Leading global vaccine manufacturer; strong late-stage pipeline (PCV, TB); successful entry into the high-value RSV market; robust adjuvant platform.
- Weaknesses: Lagging in the COVID-19 mRNA vaccine space compared to Pfizer/Moderna; historical legal and regulatory challenges.
- Opportunities: Maximizing the growth potential of Arexvy in the RSV market; advancing next-generation pneumococcal vaccines (e.g., AFX3772); expanding global access programs.
- Threats: Intense competition in the RSV market from Pfizer and Moderna; pricing pressure in government tenders; manufacturing complexity of its broad portfolio.
Recent News and Updates:
- Press Releases:
-
- October 2023: Signed a partnership deal with Zhi Fei Biological to co-promote Shingrix (shingles vaccine) in China, leveraging its distribution for other vaccines.
- April 2025: CDC ACIP endorsement for its MenACYW vaccine, Penmenvy, strengthening its meningococcal portfolio.
Industry Recognitions/Awards:
Frequently recognized for contributions to global immunization and public health, particularly for its innovative adjuvant system.
Value Chain Analysis - Respiratory Disease Vaccine Market
R&D
The latest research activities involve developing novel vaccines to deal with viral variants and developing novel drug delivery systems for vaccines.
Key Players: AstraZeneca, Pfizer, and Dr. Reddy’s Laboratories.
Clinical Trials & Regulatory Approval
Clinical trials are conducted to assess the safety and efficacy of vaccines and study their immune responses in preventing respiratory diseases.
Key Players: GlaxoSmithKline, Medicago, Inc., and Janssen Vaccines & Prevention B.V.
Distribution to Hospitals, Pharmacies
Vaccines are delivered to hospitals and retail pharmacies, either directly through manufacturers or through wholesalers or distributors.
Key Players: FFF Enterprises, Inc., Aark Pharmaceuticals
Patient Support & Services
Patient support & services refer to the focus on education, accessibility, and financial assistance to patients.
Recent Developments in the Respiratory Disease Vaccine Market
- In July 2025, Emory University and Micron Biomedical announced the initiation of their first clinical trial for a novel rotavirus vaccine, CC24. The trial evaluates the safety and effectiveness of a vaccine administered using dissolvable microarray technology. CC24 aims to save the lives of children from rotavirus illness and death.
- In May 2025, Yale researchers found that nasal vaccine boosters can trigger strong immune defenses in the respiratory tract even without the help of adjuvants. These findings may offer critical insights into developing safer, more effective nasal vaccines in the future.
Collaborate with our experts to explore the Respiratory Disease Vaccine Market at sales@towardshealthcare.com
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
