Latest Updates of Companies in the RNA Interference Therapy Market
| Company | Headquarters | Latest Update |
| Alnylam Pharmaceuticals | United States | Alnylam has led the translation of the breakthrough discovery of RNA interference into an entirely novel class of medicines, which is disrupting disease. |
| Arrowhead Pharmaceuticals | California | Arrowhead Pharmaceuticals has recently transitioned into a commercial-stage company, marking a significant milestone in its development of RNA interference (RNAi) therapeutics. |
| Silence Therapeutics | United Kingdom | In September 2025, DIANT Pharma, Inc. and RNAV8 Bio announced a strategic partnership to provide biopharma organizations with an integrated path from mRNA sequence design via scalable lipid nanoparticle (LNP) manufacturing. |
| Sirnaomics | United States | Silence Therapeutics designs and develops siRNAs to address unmet patient requirements in a broad range of diseases with a genetic basis. |
| Sanofi Genzyme | United States | Sanofi announced the Phase 1-2 study of SAR446268, an investigational gene therapy intended to eliminate toxic RNA foci responsible for splicing defects in muscle tissue, directing myotonic dystrophy type 1 (DM1). |
Recent Developments in the RNA Interference Therapy Market
- In October 2025, Aerska, a biotechnology company aiming to redefine the treatment of neurological diseases, announced it had raised $21 million in seed financing, co-led by Age1, Backed VC, and Speedinvest, with participation from Blueyard, Lingotto, Norrsken VC, Kerna, PsyMed, Saras, and Ada Ventures.
- In September 2025, Alnylam Pharmaceuticals, Inc., the leading RNAi therapeutics company, announced that it had commenced a private offering of $500 million aggregate principal amount of convertible senior
Supply Chain Analysis
R&D:
- Research and development (R&D) in RNA interference therapy involves target identification, siRNA design and modification, delivery platform advancement, preclinical in vivo proof of concept, and clinical trials.
- Key Players: Novartis and Sanofi
Manufacturing Processes:
- Manufacturing processes for RNA interference therapy include chemical synthesis, purification, chemical modification, and advanced formulation to ensure stability and targeted delivery.
- Key Players: Alnylam Pharmaceuticals and Arrowhead Pharmaceuticals
Patient Services:
- Patient services in the drug delivery nanoparticles formulation involve comprehensive education, financial assistance, insurance navigation, and specialized nursing to help manage the administration of siRNA-based drugs.
- Key Players: Silence Therapeutics and Sirnaomics
Market Growth
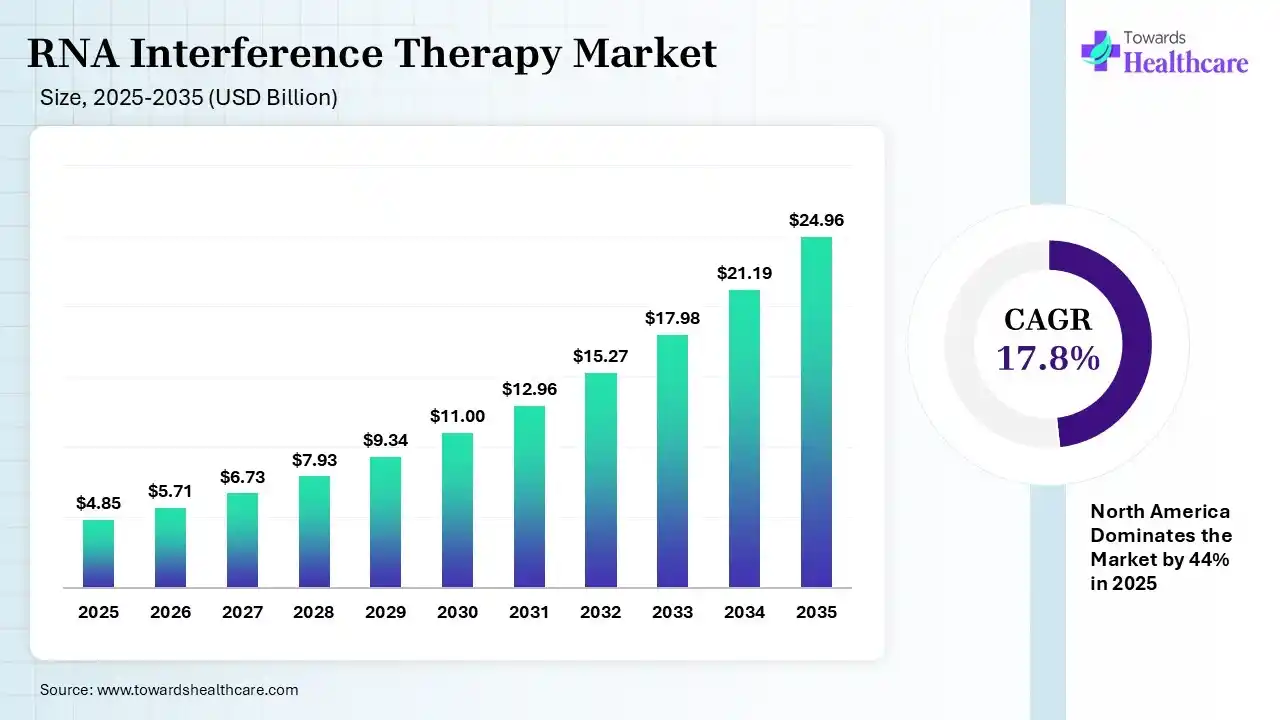
The global RNA interference therapy market size was estimated at USD 4.85 billion in 2025 and is predicted to increase from USD 5.71 billion in 2026 to approximately USD 24.96 billion by 2035, expanding at a CAGR of 17.8% from 2026 to 2035. The RNA interference therapy market is growing because RNAi has become more broadly applied in gene silencing and drug advancement, with its high level of specificity, reducing side effects, and ease of synthesis.
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
