April 2026

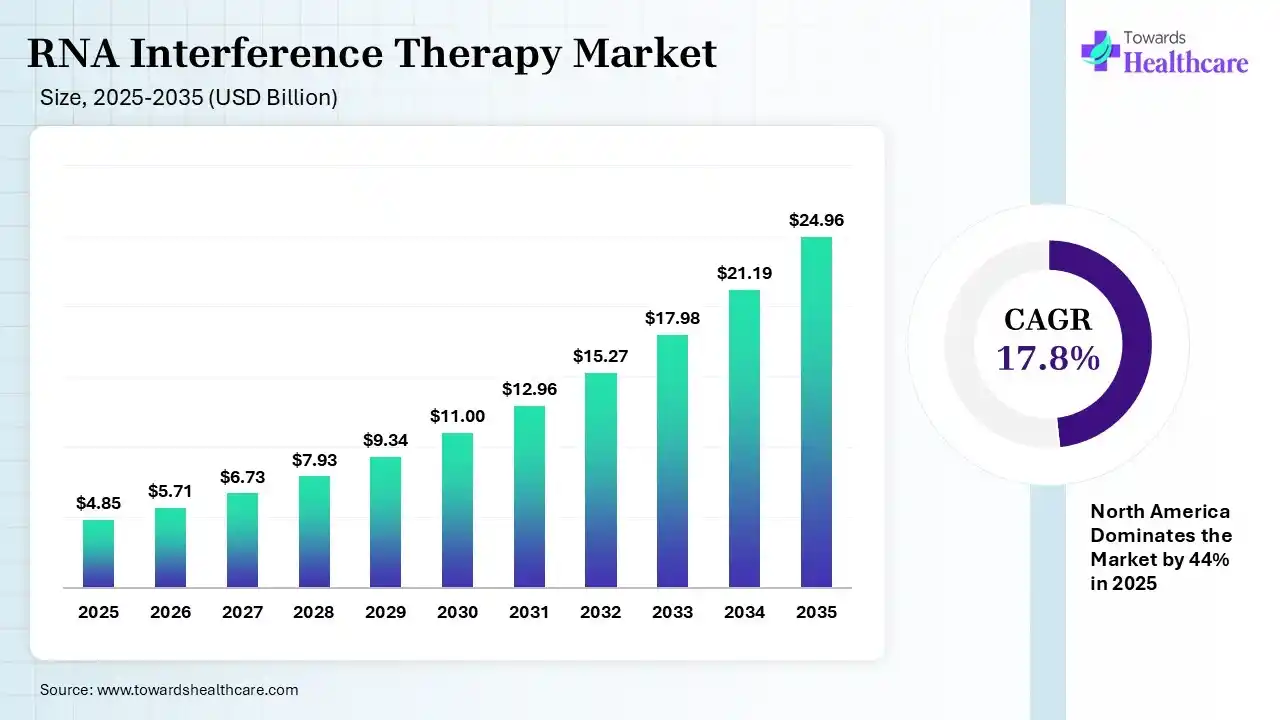
The global RNA interference therapy market size was estimated at USD 4.85 billion in 2025 and is predicted to increase from USD 5.71 billion in 2026 to approximately USD 24.96 billion by 2035, expanding at a CAGR of 17.8% from 2026 to 2035. The RNA interference therapy market is growing because RNAi has become more broadly applied in gene silencing and drug advancement, with its high level of specificity, reducing side effects, and ease of synthesis.
The RNA interference therapy market is growing as RNAi has recently been used in major research fields and management because of its target site specificity and therapeutic strength. RNAi is proposed as a potential cancer treatment by allowing targeted silencing of genes, which disrupts the expression of oncogenes and restores the function of cancer suppressor genes. RNA-driven therapies represent a promising class of treatments that harness the cellular machinery to address different diseases at the genetic level.
AI-driven technology has emerged as a significant innovation in RNAi therapeutics. AI-ready data for RNAi discovery efforts, explore solutions. AI-based technology improves the design of RNA treatments, involving small interfering RNAs (siRNAs) and antisense oligonucleotides (ASOs). AI-based automation lowers labour-intensive tasks, allowing faster and more precise RNA-target identification, resulting in cost savings and expedited testing of RNA therapies. The integration of AI into RNA drug advancement holds the strength to transform the future of therapeutics. AI-based technology improves specificity by assessing thermodynamic stability and complementarity, permitting scientists to design siRNAs that selectively target only the intended mRNA, therefore enhancing the safety profile of the treatment.
| Table | Scope |
| Market Size in 2026 | USD 5.71 Billion |
| Projected Market Size in 2035 | USD 24.96 Billion |
| CAGR (2026 - 2035) | 17.8% |
| Leading Region | North America by 44% |
| Historical Data | 2020 - 2023 |
| Base Year | 2025 |
| Forecast Period | 2026 - 2035 |
| Measurable Values | USD Millions/Units/Volume |
| Market Segmentation | By Molecule Type, By Delivery Method, By Application, By Route of Administration, By End User, By Region |
| Top Key Players | Alnylam Pharmaceuticals, Arrowhead Pharmaceuticals, Silence Therapeutics, Sirnaomics, Sanofi Genzyme |
| Segment | Share 2025 (%) |
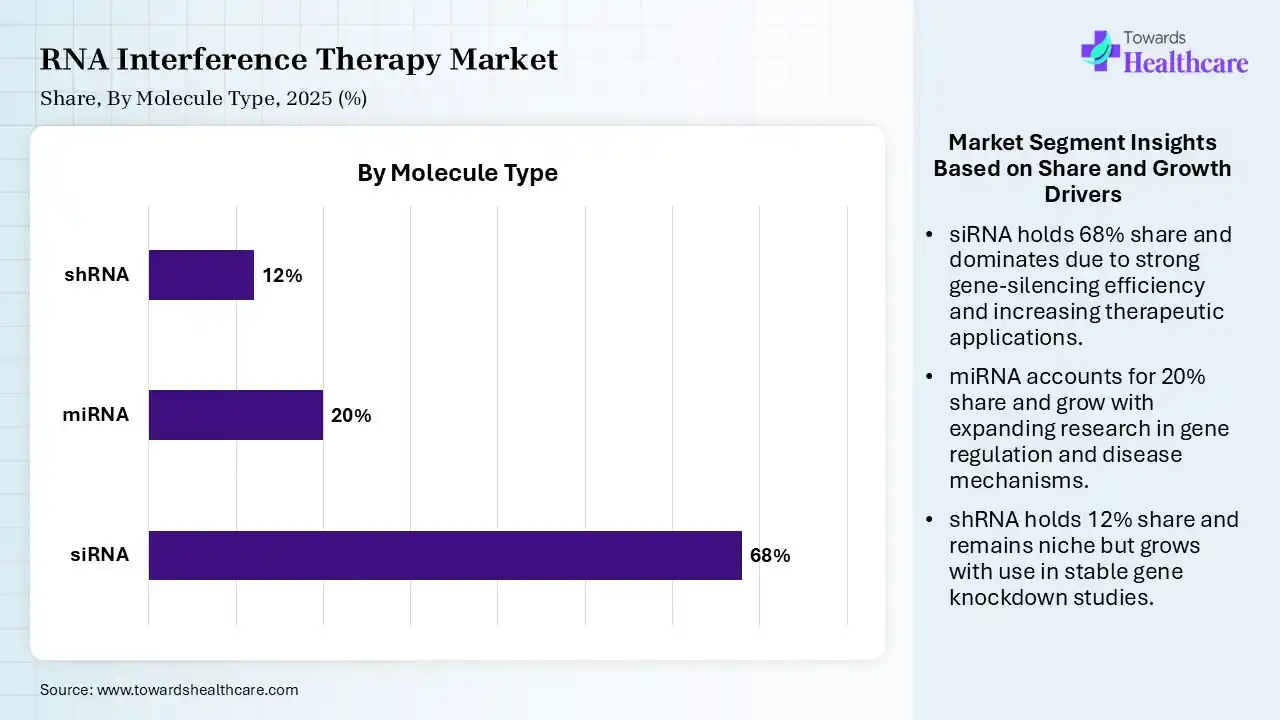
| siRNA | 68% |
| miRNA | 20% |
| shRNA | 12% |
The siRNA Segment Led the RNA Interference Therapy Market in 2025
The siRNA segment contributed the largest market share of 68% in 2025, as siRNAs are used as tools to study single gene function both in vivo and in vitro, and are an attractive novel class of therapeutics, particularly against undruggable targets for the treatment of cancer and other diseases. The siRNA delivery schemes are categorized as non-viral and viral delivery systems. siRNA for usage in clinical trials as drugs, different chemical alterations are being investigated to enhance qualities like low immune stimulation.
The miRNA segment held a significant share of 20% in the market, and is expected to be the fastest-growing during the forecast period, as using miRNA-based therapy includes the capability to target several genes in a given pathway and the facility to rapidly develop novel therapies. miRNAs exactly regulate gene expression to ensure proper organismal development. miRNA therapy is that one single miRNA can target multiple genes, which makes miRNA a significant regulator of the transcriptome.
The shRNA segment held a significant share of 12% in the market, as the advantage of shRNA over siRNA is the capability to use the viral vectors for delivery to overcome the challenges of transfecting certain cell types. Application to generate stable knockdown cell lines, thereby removing the requirement for multiple rounds of transfection and greatly increasing the reproducibility of the output. shRNA in therapeutic claims is that these macromolecules have a longer-standing effect on cellular function than siRNAs.
| Segment | Share 2025 (%) |
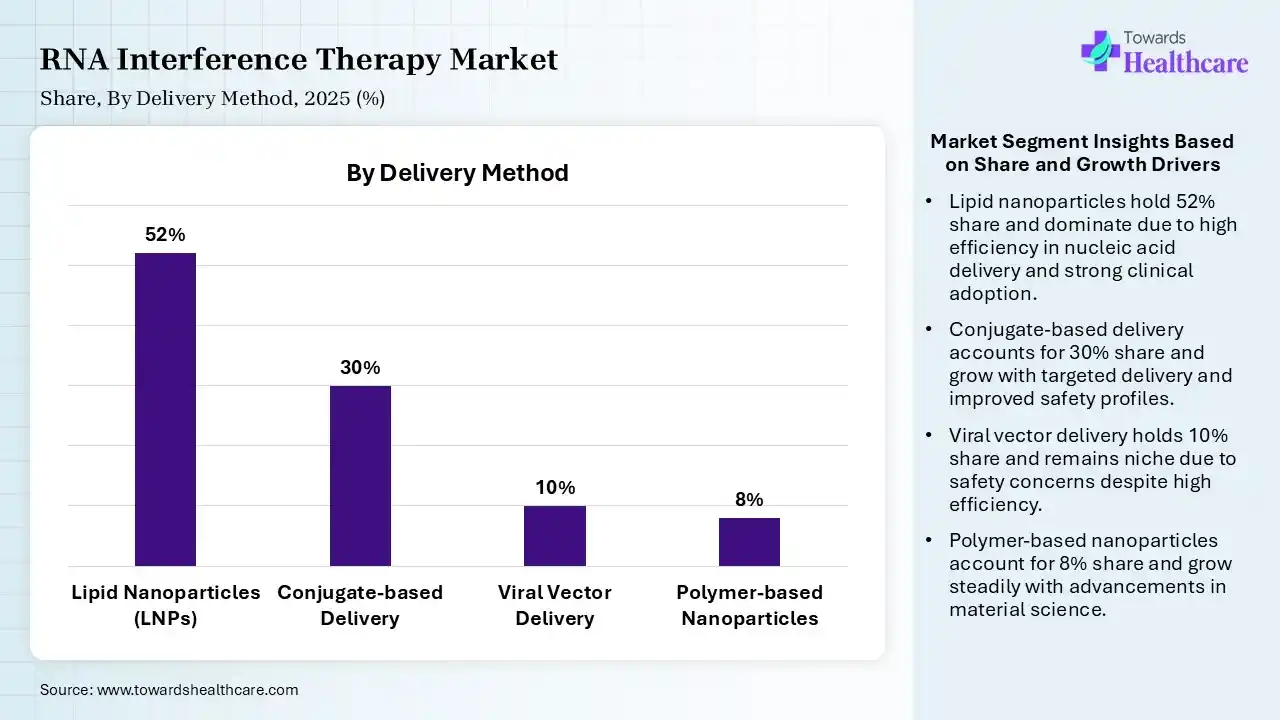
| Lipid Nanoparticles (LNPs) | 52% |
| Conjugate-based Delivery | 30% |
| Viral Vector Delivery | 10% |
| Polymer-based Nanoparticles | 8% |
Lipid Nanoparticles (LNPs) Segment Led the RNA Interference Therapy Market in 2025
The lipid nanoparticles (LNPs) segment contributed the largest market share of 52%, as lipid nanoparticles revolutionized the treatment of various types of oncology by improving the anti-cancer activity of chemotherapeutic agents. The use of LNPs allows for effective delivery of drugs, resulting in improved targeted delivery to particular tissues or cells. Advantages of LNPs for vaccine delivery are the capability to adapt their surface with targeting ligands, allowing specific delivery to immune cells or antigen-presenting cells, therefore improving the immune response.
The conjugate-based delivery segment held a significant share of 30% the market, and is expected to grow at the fastest CAGR during the forecast period. Conjugate drug delivery systems enhance patient health by maintaining the structural stability of drugs in molecular form, delivering therapeutics or diagnostic material to the target site, minimising off-target accumulation, and promoting patient compliance. A conjugate drug delivery system enhances patient health by maintaining the structural stability of medicines in molecular form, delivering therapeutics and diagnostic material to the specific site.
The viral vector delivery held a significant share of 10% of the RNA interference therapy market, as Viral vectors are currently the most effective way to deliver genetic material into cells. They offer a ground-breaking avenue for addressing both inherited and acquired disorders at their root cause, deep in the human genome. Viral vector vaccines use a harmless version of a carrier virus, or vector, to deliver specific genetic instructions to human cells.
The polymer-based nanoparticles segment held a significant share of 8% in 2025, as Polymer-based nanoparticles have emerged as powerful multifunctional platforms in cancer theranostics, offering the ability to integrate diagnostic imaging and targeted therapy within a single system. Polymer-based nanoparticles effectively carry drugs, proteins, and DNA to target cells and organs.
| Segment | Share 2025 (%) |
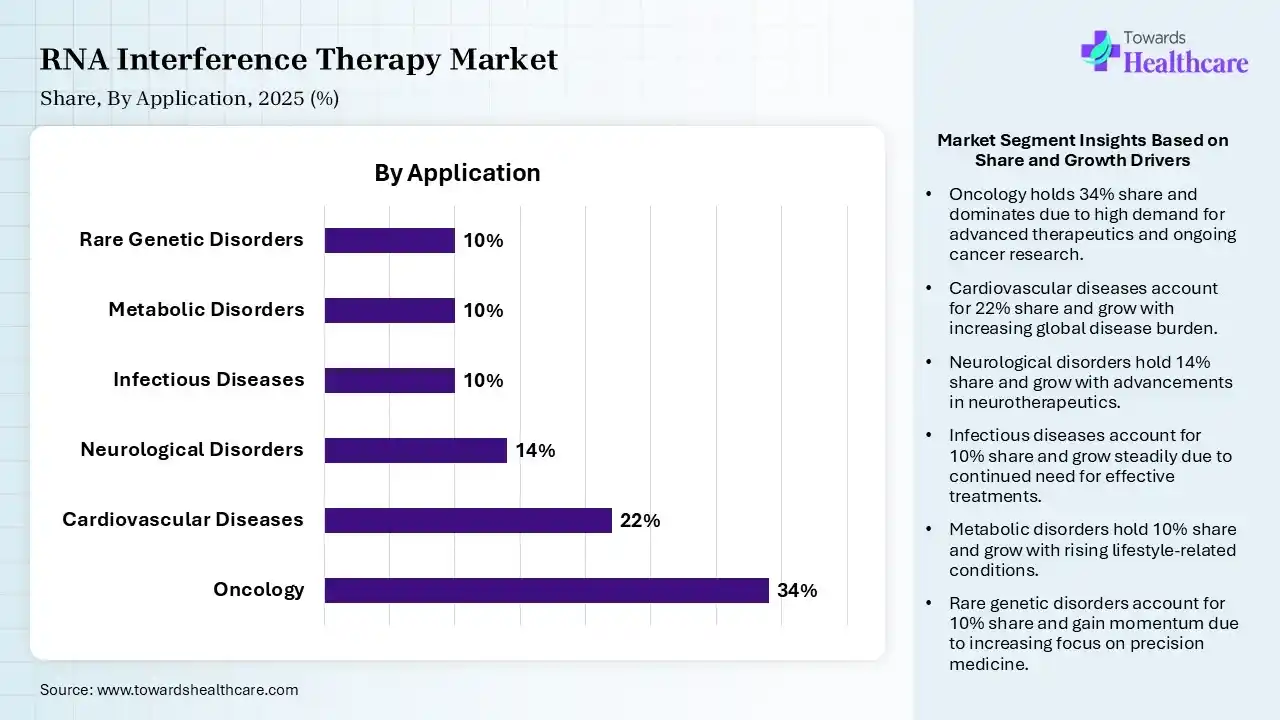
| Oncology | 34% |
| Cardiovascular Diseases | 22% |
| Neurological Disorders | 14% |
| Infectious Diseases | 10% |
| Metabolic Disorders | 10% |
| Rare Genetic Disorders | 10% |
Oncology Segment Led the RNA Interference Therapy Market in 2025
The oncology segment contributed the largest market share of 34%, as small-interfering RNA (siRNA) is a significant component of RNAi and has shown promise in silencing oncogenes and inhibiting cancer progression. RNA interference (RNAi) is a significant gene-silencing process that holds great promise in the field of cancer therapy. Therapies involving powerful antiproliferative and proapoptotic effects in cancer cells hold great promise as a new class of cancer treatments.
The cardiovascular diseases segment held a significant share of 22% in the market, as RNA therapeutics hold significant promise in the treatment of cardiovascular diseases. RNAs are biologically varied and functionally particular and can be used for gain-of-function or loss-of-function purposes. RNAi therapeutics are a new treatment choice for improving cardiac function because of their high target specificity and capability to target genes that conventional drugs struggle with.
The neurological disorders segment held a significant share of 14% in the market, and is expected to grow at the fastest CAGR during the forecast period, as allele-specific strategies are now used to silence dominant toxic genes implicated in several neurological disorders. RNA interference (RNAi) is a biological process in which RNA molecules are involved in the sequence-specific suppression of gene expression by double-stranded RNA.
The infectious diseases segment held a significant share of 10% in the RNA interference therapy market, as the therapeutic use of RNAi seems to be promising when directed against viruses that cause localized acute infections in accessible target cells. RNAi is used therapeutically in vitro and in vivo to target numerous respiratory viruses, involving SARS-CoV and possibly SARS-CoV-2, in managing and preventing infection.
The rare genetic disorders segment held a significant share of 10% in the market, as RNAi therapies efficiently silence the expression of faulty proteins, modifying the underlying molecular fault. RNAi therapies efficiently silence the expression of defective proteins, correcting the underlying molecular defect. RNAi uses siRNA to bind and degrade specific messenger RNA (mRNA), stopping the synthesis of toxic, mutant, or misfolded proteins.
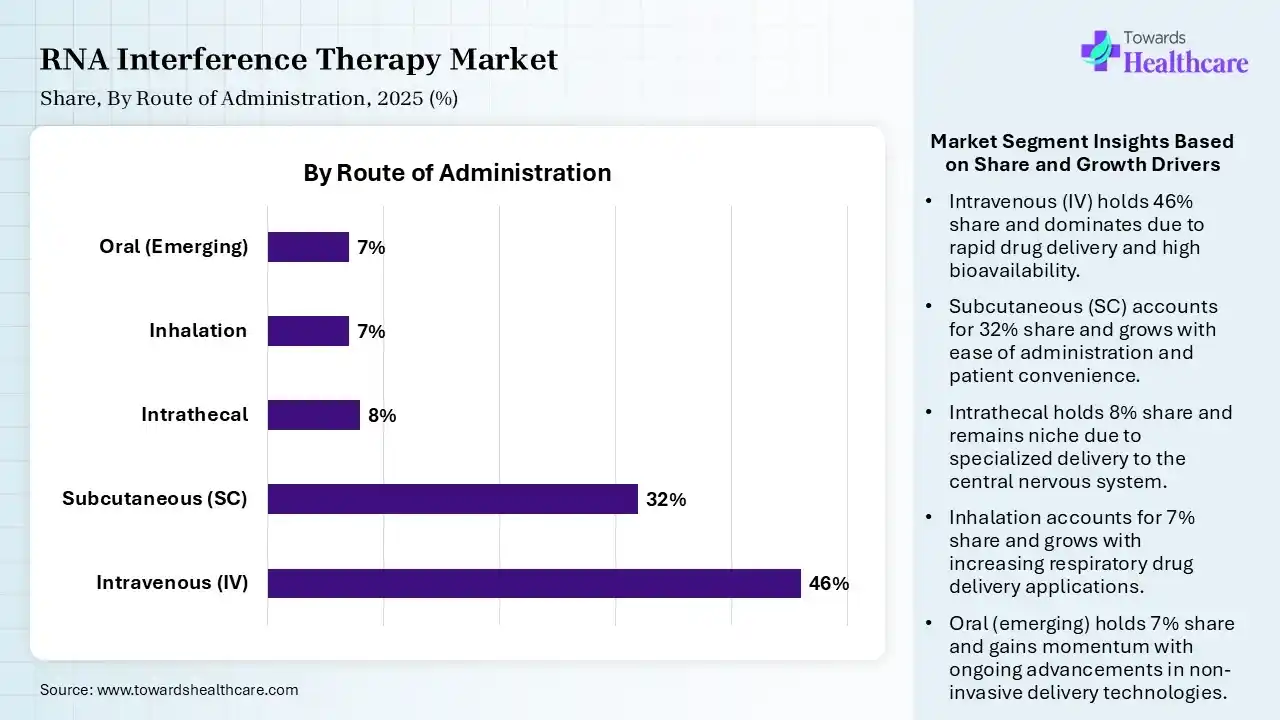
| Segment | Share 2025 (%) |
| Intravenous (IV) | 46% |
| Subcutaneous (SC) | 32% |
| Intrathecal | 8% |
| Inhalation | 7% |
| Oral (Emerging) | 7% |
Intravenous Segment Led the RNA Interference Therapy market in 2025
The intravenous segment contributed the largest market share of 46%, as this medication is directly administered to the systemic circulation. Major drugs have formulations that permit them to be given intravenously. It is an efficient and rapid process to achieve adequate analgesic concentrations in the systemic circulation. Drugs administered intravenously also have 100% bioavailability as they bypass most of the metabolic and absorptive barriers encountered via the enteral route.
The subcutaneous (SC) segment held a significant share of 32% in the market, and is expected to grow at the fastest CAGR during the forecast period, as subcutaneous administration is the administration of medications beneath the skin either by injection or infusion. The subcutaneous administration route is broadly used to administer various types of drugs, given its high bioavailability and quick onset of action.
The intrathecal segment held a significant share of 8% in the RNA interference therapy market, as intrathecal therapy permits a reduction in overall dosages and lowers the likelihood of systemic adverse events because of minimal interaction with systemic receptors. An intrathecal drug delivery system consists of a pump and an intrathecal catheter. Placing an intrathecal drug delivery system is used for cancer-related pain, severe spasticity, and noncancer pain conditions.
The inhalation segment held a significant share of 7% in the market, as its potential enhancement in aerosol delivery involves better standardization of function and patient use, greater reliability, and reduction of drug loss. Inhalation drugs generally act on target organs, reducing drug exposure to the rest of the body and so reducing systemic adverse effects. It provides rapid absorption and a rapid onset of action.
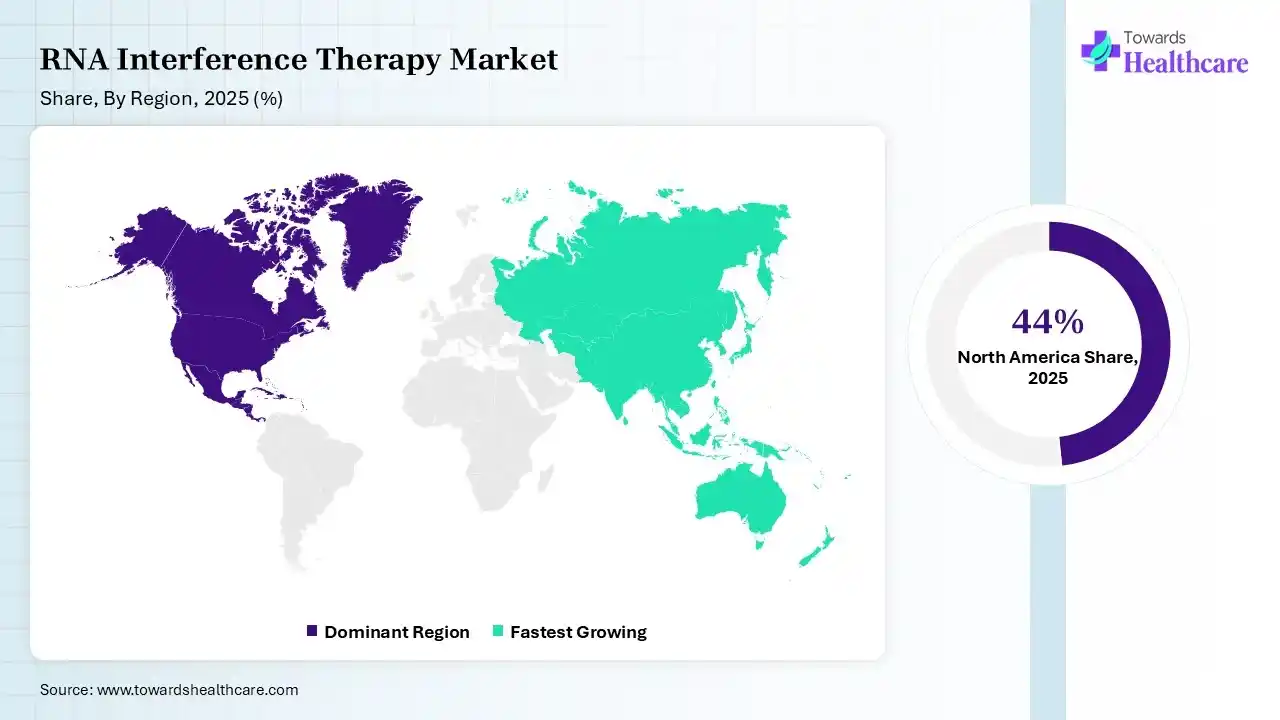
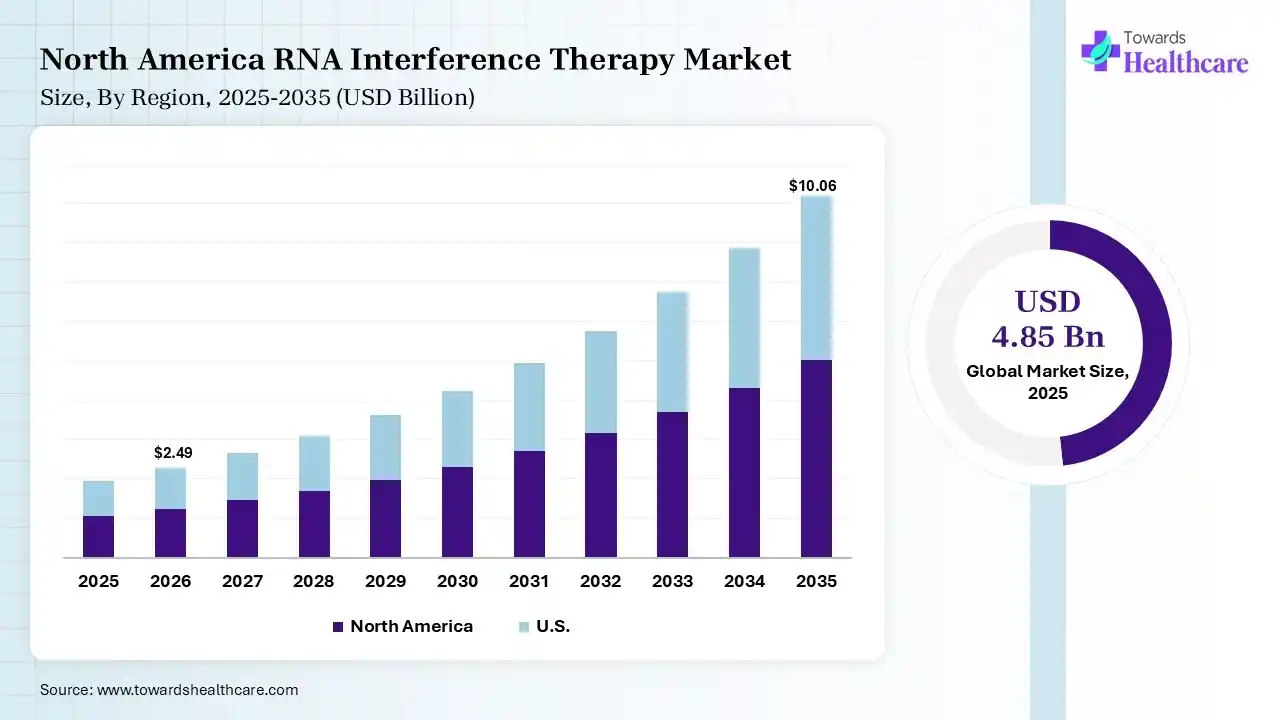
In 2025, North America dominated the RNA interference therapy market with a share of 44% in 2025, due to its advanced infrastructure and strong presence of significant players. The healthcare technology sector is quickly developing, driven by rising demand for effective data management, interoperability, and improved patient care. Regulatory reforms and financing innovations speed up approvals and support advancement, which drive the growth of the market.
For Instance,
U.S. Market Trends
In the U.S., federal research spending has contributed to medical science and enhanced public health. The biopharmaceutical company utilizes its technical and industrial expertise to build on and advance basic science research in the safe and effective treatments and vaccines that are made available to patients. Developing a novel medicine begins with understanding the disease or condition as methodically as possible.
Asia Pacific held 20% share of the RNA interference therapy market, expected to have the fastest growth during the forecast period, as the region is evolving as a biotechnology manufacturing hub. Favourable government reforms and increased spending in countries like China and Japan are quickening clinical trial approvals. A large patient population, specifically in infectious and chronic diseases, is increasing demand for advanced, localized therapeutic options. Advancing RNA innovation through collaboration and novel clinical strategies, which drives the growth of the market.
India Market Trends
In India, advancements in biotechnology, the growing prevalence of genetic and chronic diseases, and the success of mRNA vaccines. An increasing surge in strategic collaborations, joint ventures, and technology partnerships across healthcare sectors. Government initiatives to promote domestic production and enhance healthcare access are accelerating the development.
R&D:
Manufacturing Processes:
Patient Services:
| Company | Headquarters | Latest Update |
| Alnylam Pharmaceuticals | United States | Alnylam has led the translation of the breakthrough discovery of RNA interference into an entirely novel class of medicines, which is disrupting disease. |
| Arrowhead Pharmaceuticals | California | Arrowhead Pharmaceuticals has recently transitioned into a commercial-stage company, marking a significant milestone in its development of RNA interference (RNAi) therapeutics. |
| Silence Therapeutics | United Kingdom | In September 2025, DIANT Pharma, Inc. and RNAV8 Bio announced a strategic partnership to provide biopharma organizations with an integrated path from mRNA sequence design via scalable lipid nanoparticle (LNP) manufacturing. |
| Sirnaomics | United States | Silence Therapeutics designs and develops siRNAs to address unmet patient requirements in a broad range of diseases with a genetic basis. |
| Sanofi Genzyme | United States | Sanofi announced the Phase 1-2 study of SAR446268, an investigational gene therapy intended to eliminate toxic RNA foci responsible for splicing defects in muscle tissue, directing myotonic dystrophy type 1 (DM1). |
Strengths
Weaknesses
Opportunities
Threat
By Molecule Type
By Delivery Method
By Application
By Route of Administration
By End User
By Region
April 2026
April 2026
April 2026
April 2026