Latest Updates of Key Players in the Small Molecule CMC Service Market with Recent Development 2026

Company Overview
| Company | Headquarters | Latest Update |
| Lonza | Switzerland | Lonza expects a healthy level of contract signings in technologies and sites within its CDMO business. |
| Thermo Fisher Scientific | United States | Thermo Fisher Scientific is retaining an enhanced platform technology and a new CHO K-1 cell line that can reduce timelines to Investigational New Drug (IND) filing. |
| B. Braun SE | Germany | In December 2025, B. Braun Medical Inc. announced the launch of its Supply Assurance Program, a first-of-its-kind initiative intended to reinforce supply chain resiliency and protect patient care from disruption in the healthcare tools and pharmaceutical field. |
| WuXi AppTec | China | In June 2025, WuXi supports more than 3,400 small-molecule CDMO projects, with an increasing share of Phase III and commercial-stage drugs. |
| Catalent | Switzerland | Catalent delivers end-to-end pharma solutions as a trusted CDMO, progressing drug development and manufacturing globally. |
| CordenPharma | Switzerland | CordenPharma is a leading CDMO for cGMP development & manufacturing of APIs, lipid excipients, medicinal products, injectables, peptides, LNPs, oligos, & osd. |
Market Forecast
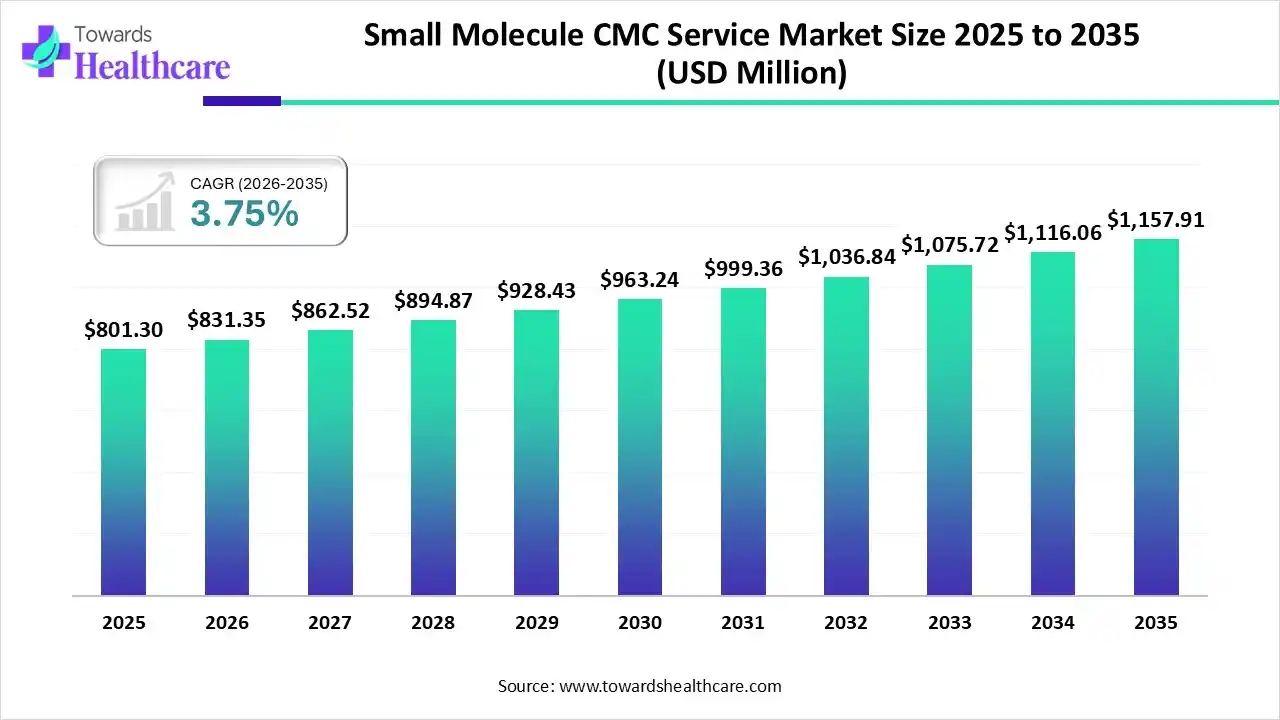
The global small molecule CMC service market size was estimated at USD 801.3 million in 2025 and is predicted to increase from USD 831.35 million in 2026 to approximately USD 1157.91 million by 2035, expanding at a CAGR of 3.75% from 2026 to 2035.
Recent Developments in the Small Molecule CMC Service Market
- In November 2025, BioDlink, a leading biologics CDMO, together with Lepu Biopharma, announced the successful market launch of MEIYOUHENG, the world's first EGFR-targeting antibody-drug conjugate (ADC). The launch also represents the first ADC fully manufactured by a CDMO to reach the market in China, underscoring a new level of maturity for the region's biopharmaceutical ecosystem.
- In January 2026, BioCryst Pharmaceuticals, Inc. announced that it had completed its acquisition of Astria Therapeutics, Inc., as initially announced. The transaction strengthens its position as a leader in hereditary angioedema (HAE) and enhances the company’s long-term growth trajectory.
- In July 2025, ESTEVE acquired Regis Technologies, a United States-based Contract Development and Manufacturing Organization (CDMO), headquartered in Chicago.
- In April 2025, PCI Pharma Services, a worldwide contract development and manufacturing organization (CDMO) focused on innovative biopharma therapies, is acquiring an entire equity stake in Ajinomoto Althea Inc., a U.S.-based sterile fill-finish CDMO and 100% subsidiary of Japan-based Ajinomoto Co. Inc.
Access full Small Molecule CMC Service Market trends, Stats and in-depth insights instantly: Checkout the Dashboard
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
