Top Vendors in the Small Molecule Innovator API CDMO Market & Their Offering
| Companies | Headquarters | Offerings |
| Lonza Group Ltd. | Basel, Switzerland | End-to-end small-molecule API services including route scouting, process development, scale-up, HPAPI manufacturing, and commercial GMP production. |
| Catalent, Inc. (Novo Holdings) | New Jersey, USA | Integrated API development, clinical and commercial manufacturing, formulation support, and supply solutions for innovator small-molecule programs. |
| Thermo Fisher Scientific, Inc. | Massachusetts, USA | Small-molecule API process development, cGMP manufacturing, analytical services, and global clinical-to-commercial supply through its CDMO network |
| Siegfried Holding AG | Zofingen, Switzerland | Custom API synthesis, process optimization, scale-up, and commercial manufacturing for innovative small-molecule drugs. |
| Recipharm AB | Stockholm, Sweden | Small-molecule API development, clinical manufacturing, oral solid dosage production, and integrated drug development services. |
| CordenPharma International | Basel, Switzerland | Specialized API development, high-potency compound manufacturing, process scale-up, and commercial GMP production. |
| Samsung Biologics | Incheon, South Korea | Primarily biologics-focused, with limited small-molecule and integrated biopharma development support for innovator pipelines. |
| Labcorp | North Carolina, USA | Early-stage API development support, analytical services, formulation, and clinical trial supply enabling accelerated small-molecule development. |
Value Chain Analysis
R&D
- Small molecule API CDMO R&D services offer comprehensive support across the drug development lifecycle, from early preclinical stages to commercial manufacturing. These services focus on efficient process development, scalability, safety, and strict regulatory compliance to ensure high-quality drug substance production.
- Key players: Lonza, Catalent, WuXi AppTec, and Cambrex.
Clinical Trials
- Small molecule API CDMOs play a vital role in supporting Phase I–III clinical trials by handling API development, scale-up, and cGMP-compliant manufacturing. They ensure regulatory adherence, improve process efficiency, and manage reliable supply chains to speed progression from early development to commercialization.
- Key players: Piramal Pharma Solutions, Divi’s Laboratories, Thermo Fisher Scientific, Asymchem.
Regulatory Approvals
- Regulatory approvals for small molecule API CDMOs depend on rigorous compliance with cGMP requirements and global quality standards, including alignment with FDA and EMA guidelines. Maintaining validated processes, robust quality systems, and inspection readiness is critical for approval and long-term operations.
- Key players: Dr. Reddy’s Laboratories, Aurobindo Pharma, Hovione, and Recipharm.
Market Growth
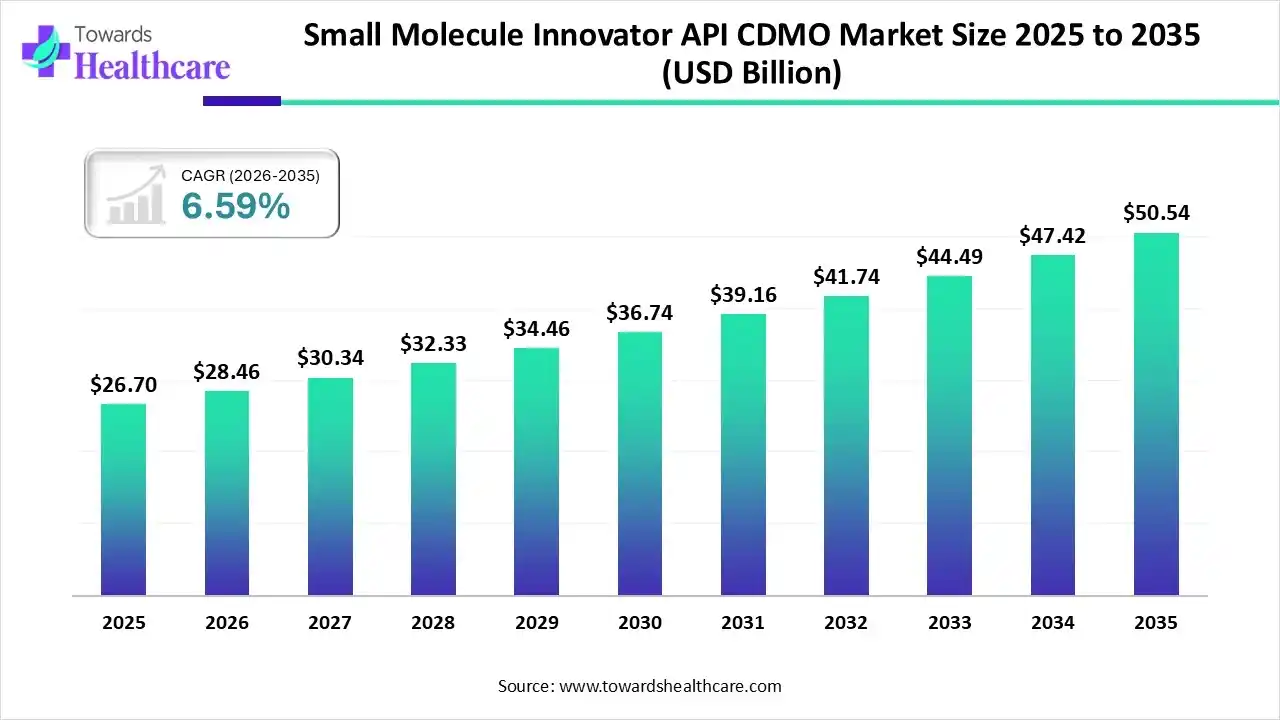
The global small molecule innovator API CDMO market size was estimated at USD 27.6 billion in 2025 and is predicted to increase from USD 28.46 billion in 2026 to approximately USD 50.54 billion by 2035, expanding at a CAGR of 6.59% from 2026 to 2035.
What are the Recent Developments in the Small Molecule Innovator API CDMO Market?
- In October 2025, Cambrex committed USD 120 million to expand its Charles City, Iowa, facility by 40%, enhancing API and peptide manufacturing capacity while reinforcing domestic pharmaceutical supply chain resilience.
- In September 2025, Thermo Fisher Scientific, Inc. finalized the acquisition of a sterile fill-finish and packaging facility from Sanofi in Ridgefield, New Jersey, strengthening its U.S. manufacturing footprint and expanding support capabilities for pharmaceutical and biotechnology clients.
Grab a custom sample packed with relevant data for smarter decisions: Download Now
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
