April 2026

The global small molecule innovator API CDMO market size was estimated at USD 27.6 billion in 2025 and is predicted to increase from USD 28.46 billion in 2026 to approximately USD 50.54 billion by 2035, expanding at a CAGR of 6.59% from 2026 to 2035.
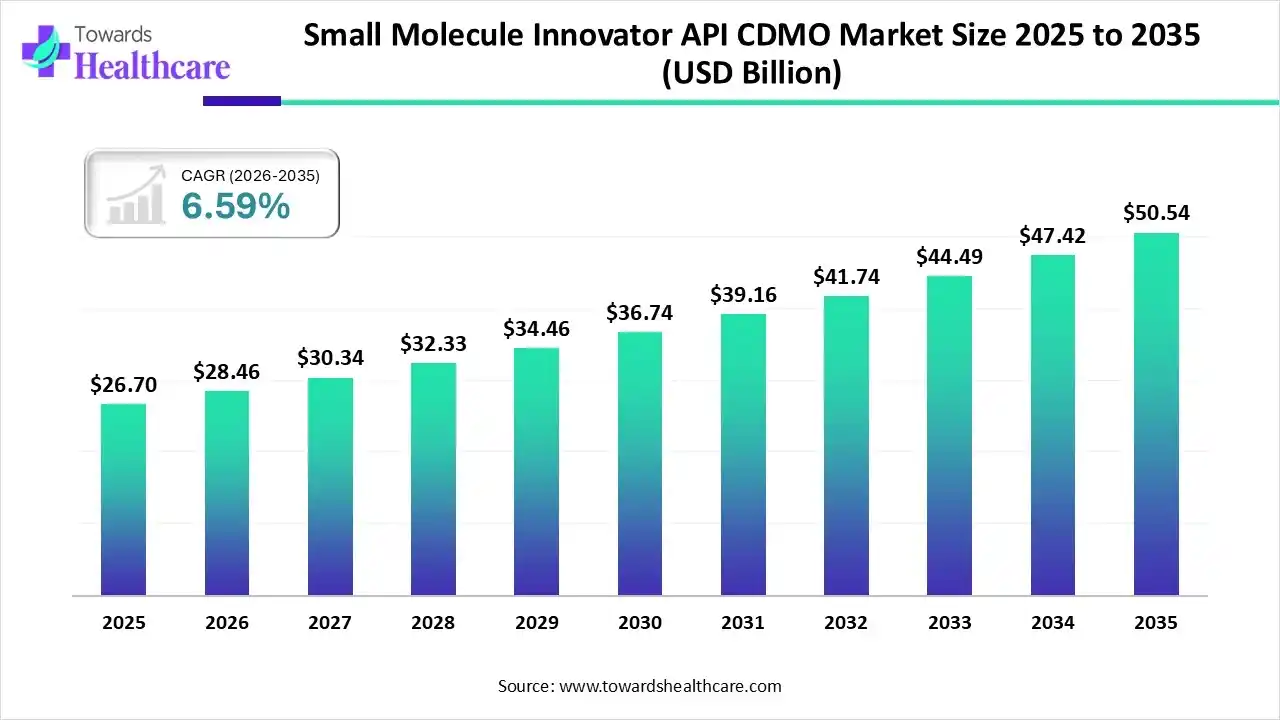
The market is growing as drug development increasingly outsources specialized synthesis and manufacturing to reduce costs, improve scalability, and focus on core research, driven by rising demand for complex APIs and global clinical expansion.
A small molecule innovator API CDMO is a contract development and manufacturing organization that specializes in producing active pharmaceutical ingredients (APIs) for novel small-molecule drugs, offering expertise in synthesis scale-up and regulatory-compliant manufacturing. The small molecule innovator API CDMO market is growing as pharmaceutical companies increasingly outsource API development and manufacturing to reduce costs, shorten drug development timelines, and access specialized expertise. Rising global demand for novel therapies, complex small-molecule drugs, and efficient scale-up capabilities are driving CDMOs adoption. Regulatory support and the need for flexible, high-quality production further accelerate market expansion worldwide.
AI is set to revolutionize the market by optimizing drug design, predicting reaction outcomes, and accelerating process development. It enables faster synthesis route selection, improves yield and quality, and reduces development costs. By integrating AI-driven analytics, CDMOs can enhance efficiency, minimize risks, and deliver APIs more rapidly, strengthening their role in supporting innovative pharmaceutical pipelines.
| Table | Scope |
| Market Size in 2026 | USD 28.46 Billion |
| Projected Market Size in 2035 | USD 50.54 Billion |
| CAGR (2026 - 2035) | 6.59% |
| Leading Region | Asia Pacific by 43% |
| Historical Data | 2020 - 2023 |
| Base Year | 2025 |
| Forecast Period | 2026 - 2035 |
| Measurable Values | USD Millions/Units/Volume |
| Market Segmentation | By Stage Type, By Customer Type, By Therapeutic Area, By Region |
| Top Key Players | Lonza Group Ltd., Catalent, Inc. (Novo Holdings), Thermo Fisher Scientific, Inc., Siegfried Holding AG, Recipharm AB, CordenPharma International |
Why Did the Clinical Segment Dominate in the Small Molecule Innovator API CDMO Market in 2025?
The clinical segment dominated the market in 2025 as pharmaceutical companies increasingly outsourced early-stage API development for preclinical and clinical trials. CDMOs provided specialized expertise, flexible production, and rapid scale-up, ensuring efficient testing, regulatory compliance, and faster scale-up for novel small-molecule drugs, driving higher demand in clinical-stage manufacturing services.
Commercial
The commercial segment is expected to grow at the fastest CAGR during the forecast period as more small-molecule drugs successfully progress from clinical to commercial stages. Rising drug approvals, increasing demand for large-scale API manufacturing, and long-term outsourcing partnerships drive growth. CDMOs offer cost-efficient production, regulatory compliance, and scalable capacity, making them preferred partners for sustained commercial manufacturing.
What Made the Pharmaceutical Segment Dominant in the Small Molecule Innovator API CDMO Market in 2025?
The pharmaceutical segment dominated the market as large and mid-sized pharma companies increasingly relied on CDMOs to support their expanding small-molecule innovation pipelines. High R&D activity, strong financial resources, and the need to accelerate development timelines drove outsourcing. Additionally, pharma companies preferred CDMOs for regulatory expertise, relied on quality standards, and scalable manufacturing across clinical and commercial stages.
Biotechnology
The biotechnology segment is expected to grow at the fastest CAGR as biotech companies increasingly focus on innovative small-molecule therapies while lacking in-house manufacturing infrastructure. Heavily relance in CDMOs for end-to-end API development, cost efficiency, and faster scale-up supports growth. Rising venture funding, expanding biotech pipelines, and increased clinical activity accelerate outsourcing demand throughout the forecast period.
How did the Oncology Segment Dominate the Small Molecule Innovator API CDMO Market in 2025?
The oncology segment dominated the market due to the strong pipeline of cancer therapies requiring complex and high-potency APIs. Rising cancer prevalence, increased R&D investment, and accelerated regulatory approvals drive demand. CDMOs offered specialized capabilities in potent compound synthesis, scale-up, and compliance, making them essential partners for oncology drug development and manufacturing.
| Year | Oncology Approval (Drug/API) |
| 2024 | Osimertinib with chemotherapy (Tagrisso) |
| Lifileucel (Amtagvi) | |
| Tepotinib (Tepmetko) | |
| Irinotecan liposome (Onivyde combo) | |
| Erdafitinib (Balversa) | |
| Nivolumab + hyaluronidasenvhy (Opdivo Qvantig) | |
| Encorafenib + cetuximab + mFOLFOX6 | |
| 2025 | Acalabrutinib + bendamustine + rituximab (Calquence combo) |
| Brentuximab vedotin + lenalidomide & rituximab (Adcetris combo) | |
| Vimseltinib (Romvimza) | |
| Pembrolizumab + chemo (Keytruda combos) |
Infectious Diseases
The infectious diseases segment is expected to grow at the fastest CAGR during the forecast period due to rising global infection rates and the continued threat of emerging and drug-resistant pathogens. Increased investment in antiviral and antibacterial drug development, along with faster regulatory pathways, drives demand for rapid, scalable API manufacturing. CDMOs play a critical role by supporting accelerated development timelines and flexible production needs.
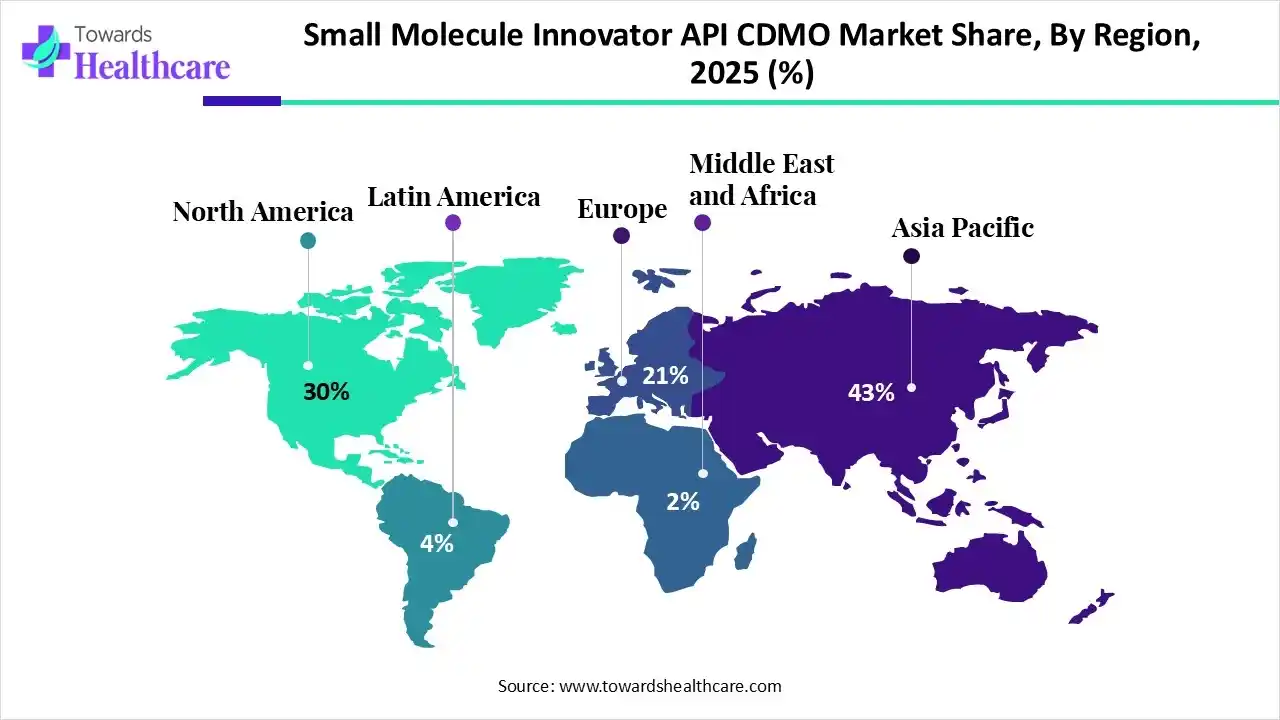
Asia Pacific dominated the market in 2025 due to its robust manufacturing ecosystem, cost advantages, and availability of skilled chemists. Strong presence of established CDMOs, expanding pharmaceutical R&D activities, supportive government initiatives, and increasing outsourcing from global pharma companies, particularly to India and China, further reinforced the region’s market leadership and production scalability.
India Rise as a Global API CDMO Revenue Leader
India led the market in2025 by capturing the largest revenue share due to its strong expertise in small-molecule synthesis, cost-competitive manufacturing, and a large, skilled workforce. A robust network of established CDMIs, regulatory-compliant facilities, and growing trust from global pharmaceutical innovators accelerates outsourcing, long-term contracts, and high-volume API production, strengthening India’s revenue dominance.
North America is expected to grow at the fastest CAGR during the forecast period due to strong pharmaceutical and biotechnology innovation, increasing clinical activity, and rising demand for complex small-molecule APIs. Growing outsourcing to specialized CDMOs, early adoption of advanced manufacturing technologies, and supportive regulatory pathways further accelerate development and commercialization, positioning the region for sustained high growth momentum.
U.S. Accelerates as a High-Growth Hub for API CDMOs
The U.S. is anticipated to grow at a rapid CAGR during the forecast period due to strong innovation in small-molecule drug development and a robust biotech ecosystem. Rising clinical trial activity, increasing demand for complex and high-potency APIs, and growing reliance on outsourcing drive growth. Advanced manufacturing technologies, favorable regulatory support, and faster commercialization timelines further strengthened the U.S. market outlook.
Europe is anticipated to grow at a notable CAGR during the forecast period due to increasing investments in pharmaceutical innovation and expanding clinical pipelines. Strong regulatory standards, growing demand for complex and high-value APIs, and rising outsourcing by pharma and biotech companies support growth. Additionally, focusing on sustainable manufacturing practices and advanced process technologies enhances Europe’s competitiveness and long-term market expansion.
UK Emerges as a Fast-Growing Hub in the Small Molecule Innovator API CDMO Market
The UK is anticipated to grow at a rapid CAGR during the forecast period due to strong pharmaceutical and biotechnology research, expanding clinical trials activity, and rising demand for innovative small-molecule drugs. Increased outsourcing to specialized CDMOs, supportive regulatory frameworks, and government initiatives promoting life science investment further accelerate market growth and strengthen the UK’s position in advanced API development and manufacturing.
| Companies | Headquarters | Offerings |
| Lonza Group Ltd. | Basel, Switzerland | End-to-end small-molecule API services including route scouting, process development, scale-up, HPAPI manufacturing, and commercial GMP production. |
| Catalent, Inc. (Novo Holdings) | New Jersey, USA | Integrated API development, clinical and commercial manufacturing, formulation support, and supply solutions for innovator small-molecule programs. |
| Thermo Fisher Scientific, Inc. | Massachusetts, USA | Small-molecule API process development, cGMP manufacturing, analytical services, and global clinical-to-commercial supply through its CDMO network |
| Siegfried Holding AG | Zofingen, Switzerland | Custom API synthesis, process optimization, scale-up, and commercial manufacturing for innovative small-molecule drugs. |
| Recipharm AB | Stockholm, Sweden | Small-molecule API development, clinical manufacturing, oral solid dosage production, and integrated drug development services. |
| CordenPharma International | Basel, Switzerland | Specialized API development, high-potency compound manufacturing, process scale-up, and commercial GMP production. |
| Samsung Biologics | Incheon, South Korea | Primarily biologics-focused, with limited small-molecule and integrated biopharma development support for innovator pipelines. |
| Labcorp | North Carolina, USA | Early-stage API development support, analytical services, formulation, and clinical trial supply enabling accelerated small-molecule development. |
By Stage Type
By Customer Type
By Therapeutic Area
By Region
April 2026
April 2026
February 2026
February 2026