Top Innovators of U.S. Bispecific Antibody Market
Introduction
Revolutionizing Targeted Therapies: The Rise of Bispecific Antibodies in the U.S.
The U.S. bispecific antibody market is emerging as one of the most transformative segments within the biopharmaceutical industry. Bispecific antibodies (bsAbs) are engineered to simultaneously bind to two different antigens or epitopes, offering a novel therapeutic mechanism for complex diseases such as cancer, autoimmune disorders, and infectious diseases. This dual-targeting capability enhances precision and efficacy compared to traditional monoclonal antibodies, positioning bsAbs as a cornerstone of next-generation biologics.
Driven by rapid advancements in antibody engineering, growing clinical success, and strong regulatory support, the U.S. market is experiencing accelerated growth. The increasing number of FDA approvals for drugs such as blinatumomab, emicizumab, and teclistamab reflects growing clinical confidence in bispecific technologies. Major pharmaceutical companies like Amgen, Roche, Johnson & Johnson, and Regeneron are expanding their research pipelines and collaborations, fueling innovation and commercialization. With a robust biotechnology ecosystem, advanced healthcare infrastructure, and a high demand for precision oncology treatments, the U.S. continues to lead the global bispecific antibody landscape.
Impact in Different U.S. Regions
1. Northeast (New York, Massachusetts, Pennsylvania)
-
Strongest impact due to the presence of major biotech hubs like Boston–Cambridge and Philadelphia.
-
Home to leading research institutions (Harvard, MIT, UPenn) and pharmaceutical giants supporting bsAb innovation.
-
Access to skilled R&D workforce and venture capital drives early-stage bsAb development.
2. Midwest (Illinois, Ohio, Indiana, Michigan)
-
Moderate impact with focus on clinical manufacturing and bioprocessing facilities.
-
States like Indiana and Illinois support biosimilar and antibody manufacturing infrastructure.
-
Growth driven by collaborations between academic centers and pharma manufacturing firms.
3. South (Texas, North Carolina, Georgia, Florida)
-
Emerging bsAb activity fueled by biotech clusters in Raleigh-Durham (Research Triangle Park) and Houston.
-
Increasing investments in biomanufacturing capacity and clinical research.
-
Favorable tax incentives and low operational costs attract biotech startups focused on antibody therapeutics.
4. West (California, Washington)
-
Highest concentration of bispecific antibody developers due to major biotech ecosystems in San Francisco Bay Area and San Diego.
-
Companies like Amgen, Genentech (Roche), and CytomX Therapeutics drive bsAb innovation.
-
Strong funding, robust clinical pipeline, and presence of leading CRO/CDMO services boost market growth.
U.S. Bispecific Antibody Market Growth
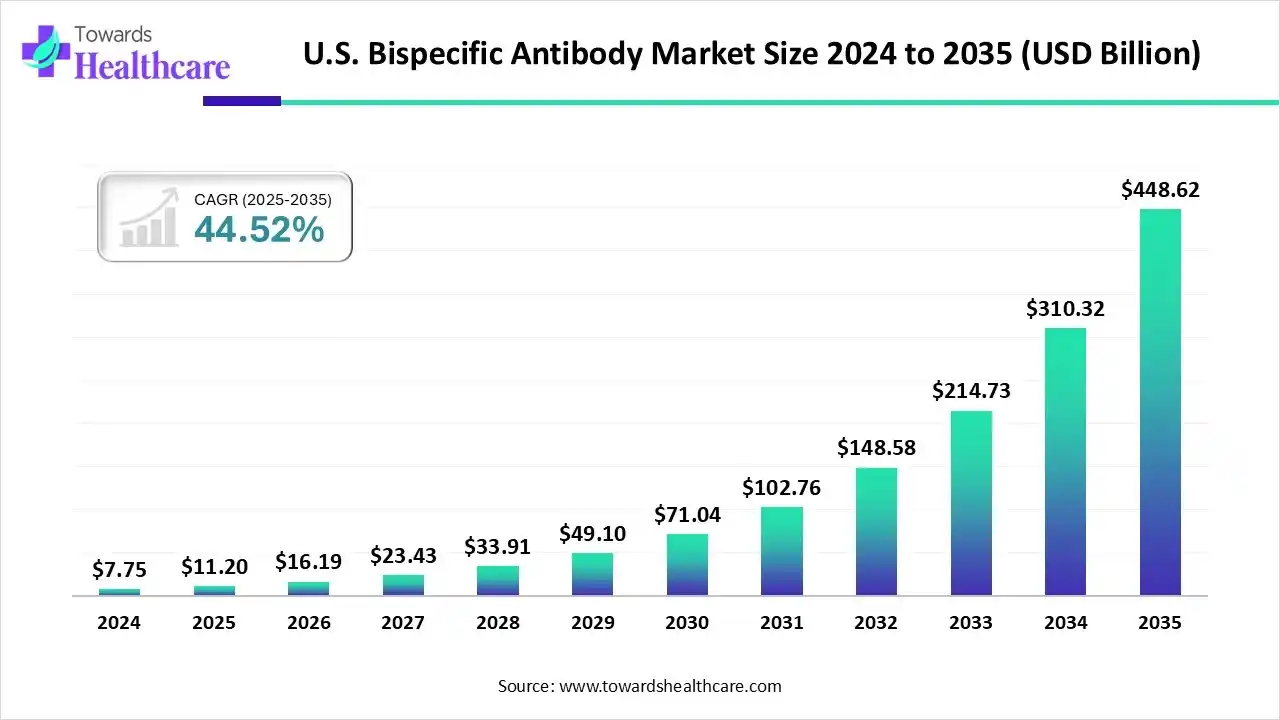
The U.S. bispecific antibody market size is estimated at US$ 11.2 billion in 2025, increased to US$ 16.19 billion in 2026, and is expected to reach around US$ 448.62 billion by 2035. The market is growing at a CAGR of 44.52% between 2025 and 2035.
Global Bispecific Antibody Market Growth
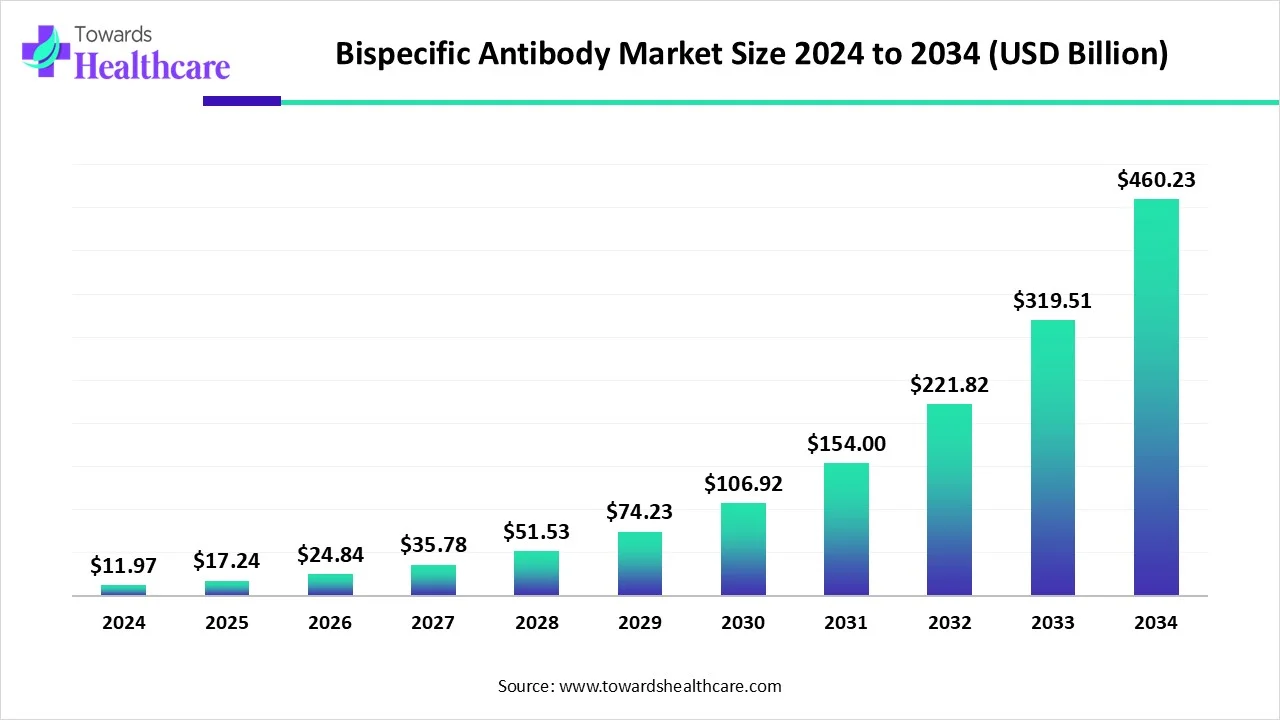
The global bispecific antibody market size is calculated at USD 11.97 in 2024, grew to USD 17.24 billion in 2025, and is projected to reach around USD 460.23 billion by 2034. The market is expanding at a CAGR of 44.04% between 2025 and 2034.
Leading Giants in the U.S. Bispecific Antibody Market
Amgen
Corporate Information
- Headquarters: One Amgen Center Drive, Thousand Oaks, California, U.S. | Year Founded: 1980 (originally as Applied Molecular Genetics)
Business Overview
Amgen is a leading U.S.-based biotechnology company focused on discovering, developing, manufacturing, and delivering innovative human therapeutics. The company’s strategic focus spans oncology, cardiovascular, inflammation, rare disease, and general medicine, and it increasingly invests in advanced biologics and next-generation modalities, including bispecific antibodies.
Business Segments / Divisions
While Amgen previously reported its operations simply as “Human Therapeutics”, it is increasingly organized around therapeutic areas and technology platforms:
- Oncology/Hematology (including bispecific antibody programs)
- Inflammation / Immunology
Rare Diseases
General Medicine / Cardiovascular / Bone Health
Additionally, Amgen emphasizes its biologics manufacturing, biosimilars, and advanced molecular engineering platforms.
Geographic Presence
Amgen has a global footprint, operating in approximately 100 countries and regions worldwide. Its manufacturing and R&D operations are located in the U.S. (California, Rhode Island, Massachusetts, North Carolina) and internationally (Ireland, Netherlands, Singapore), among others.
Key Offerings
- Amgen has a robust pipeline that includes bispecific Tcell engagers (BiTEs) and dualtargeting antibody formats, positioning it to capture growth in the bispecific antibody market.
- The company’s biologics manufacturing network supports production of complex engineered antibodies, enabling scale-up for commercialization.
- Amgen’s broad portfolio of therapeutics (including flagship biologics) provides a platform for technologyleveraging into next-generation modalities like bispecifics.
SWOT Analysis
- Strengths
- Large and diversified biologics portfolio with strong market recognition and revenue generation.
- Advanced R&D capabilities and robust innovation pipeline, particularly in biologics engineering.
- Global manufacturing and supplychain infrastructure, enabling scaling of complex biologics.
- Weaknesses
- Dependency on a few key drugs for a large portion of revenue, exposing the company to patent expiration and biosimilar competition risk.
- High R&D and capital expenditure burdens tied to biologics innovation and manufacturing operations.
- Opportunities
- Growth in the U.S. bispecific antibody market presents a significant expansion vector for Amgen’s biologics engineering capabilities. (Implicit from pipeline focus)
- Emerging markets and increasing global demand for advanced biologic therapies and bispecific formats.
- Technological advances, including AI/data science (Amgen India tech center), could accelerate discovery and development.
- Threats
- Pricing pressures and regulatory/reimbursement challenges in the U.S. biotech/biopharma environment.
- Competitive dynamics from other biotech firms developing bispecifics, and the potential for disruptive modalities (e.g., CART, cell therapies) to impact market share.
- Patent losses and biosimilar competition are reducing margins on established biologics.
Other Major Players
- Regeneron Pharmaceuticals: The company is listed among prominent U.S. companies in bispecific antibodies. It has approved and launched bispecific antibodies in the U.S., which are Lynozyfic and Odronextamab. Investigational bispecific antibodies in clinical trials include REGN7075, REGN5713, and REGN5715, and a Platinum-resistant ovarian cancer bispecific antibody.
- Genentech/Roche: Bispecific antibodies offered in the U.S. are Columvi, Lunsumio, Vabysmo, and Piasky.
- Bristol Myers Squibb/Celgene-era assets: BMS bispecific pipeline encompasses BNT327 (partnered with BioNTech) and iza-bren (BL-B01D1) (partnered with SystImmune).
- Gilead/Kite: Investigational bispecific antibody is Anitocabtagene autoleucel, and these companies focus on CAR-T cell therapies.
- Pfizer Inc.: It is active in licensing and developing next‑generation bispecific therapies.
- Merck & Co., Inc.: It is expanding its bispecific antibody portfolio in oncology & autoimmune disease.
- Xencor Inc.: It is a biotech firm with a focus on bispecific antibody platforms.
- AbbVie Inc.: The company is engaged in bispecific antibody development, especially in hematologic and immune disease spaces.
- Johnson & Johnson: It is part of the evolving bispecific antibody market via collaborations and internal programs.
U.S. Bispecific Antibody Market Value Chain Analysis
R&D
The R&D process for a new bispecific antibody candidate includes discovery and preclinical development, manufacturing and regulatory considerations, clinical development, and post-approval activities.
Key Players: Regeneron, Amgen, Genentech (Roche), Janssen (Johnson & Johnson), Pfizer, MacroGenics, and AbbVie.
Distribution to Hospitals, Pharmacies
Distribution of bispecific antibodies to pharmacies is driven through online and retail pharmacies by emphasizing increased outpatient use and expanded application.
Key Players: McKesson Corporation, Cencora, Inc., Cardinal Health, Inc., Amgen, Johnson & Johnson, Genentech, AbbVie, Pfizer, and Regeneron Pharmaceuticals.
Patient Support and Services
Patient support focuses on managing side effects, educating patients, expanding access to care, providing financial assistance, and helping navigate reimbursement.
Key Players: Roche/Genentech, Amgen, Johnson & Johnson, Genentech, AbbVie, Pfizer, and Regeneron Pharmaceuticals.
Recent Developments
- In June 2024, Amgen showcased its ongoing excellence in Bispecific T-cell Engager (BiTE®) technology to advance this groundbreaking innovation and evaluate its impact on people facing difficult-to-treat cancers.
- In June 2024, Regeneron Pharmaceuticals announced its clinical trial results for its multiple myeloma antibody, linvoseltamab, from the Phase I/II LINKER-MM1 (NCT03761108) study, which were presented at the European Hematology Association (EHA) Congress.
Exclusive Analysis
- The U.S. bispecific antibody market is at an inflection point where therapeutic modality innovation converges with scalable commercialization. Fueled by a dense pipeline of dual-target and multispecific designs, the U.S. ecosystem is rapidly shifting from proofofconcept toward late-stage development and regulatory filing readiness. Operators who can effectively navigate the challenges of bispecific engineering, such as manufacturability, immunogenicity, and target selection, are set to gain an outsized competitive advantage.
- Moreover, the horizon for bispecifics extends well beyond oncology into immunology, infectious disease, and other non-malignant indications, thereby unlocking multiple high-growth vectors. Partnerships and licensing deals worth multiple billions underscore the strategic significance of this modality and signal a readiness of capital markets and biopharma stakeholders to invest in bispecific platforms. With manufacturing capacity scaling and biologicsengineering platforms maturing, the structural supplychain constraints are loosening, creating operational levers for early adopters.
- Finally, the U.S. regulatory, payer, and commercial infrastructure is uniquely supportive of next-generation biologics, providing first-mover territories for bispecifics with validated differentiation. For developers that can demonstrate clear clinical differentiation, patientselection strategies, and scalable manufacturing, the commercial prize is substantial. In short, the U.S. bispecific antibody market is not simply expanding; it is transforming, and those that align technological capability, regulatory acumen, and commercial execution are set to define the next wave of biologic leadership.
Partner with our experts to explore the U.S. bispecific antibody market at sales@towardshealthcare.com
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
