Top Vendors in the U.S. Respiratory Durable Medical Equipment (DME) Market & Their Offerings

Company Profile
| Companies | Headquarters | Offerings |
| Arjo AB | Sweden | Patient mobility solutions, hospital beds, hygiene systems, and therapeutic support surfaces are used in long-term and home care settings. |
| Baxter International Inc. | USA | Infusion systems, renal care products, and critical care equipment supporting chronic disease management. |
| Becton, Dickinson and Co. | New Jersey, USA | Medical supplies, diagnostic devices, and patient monitoring tools are used alongside DME in home and clinical care. |
| Cardinal Health Inc. | Ohio, USA | Distribution of medical supplies, respiratory equipment, and home healthcare products across DME networks. |
| Compass Health Brands | Ohio, USA | Mobility aids, respiratory products, bathroom safety equipment, and home care solutions. |
| DRIVE MEDICAL GMBH and CO. KG | Germany | Wheelchairs, oxygen therapy equipment, beds, and daily aids for homecare patients. |
| GF Healthcare Technologies | Illinois, USA | Imaging systems, monitoring devices, and digital health solutions supporting respiratory and critical care. |
| GF Health Products, Inc. | Georgia, USA | Patient beds, respiratory equipment, mobility products, and rehabilitation aids for home and institutional use. |
Everything you need to monitor the mRNA CDMO market, organized and accessible; Access the Dashboard
Supply Chain Analysis
R&D
- Research and development in the U.S. respiratory durable medical equipment (DME) market is increasingly centered on creating smaller, smarter, and more connected devices to better manage chronic conditions like COPD and sleep apnea. Companies are focusing on improving portability, integrating digital monitoring, and using AI to enhance treatment efficiency and patient outcomes.
- Key Players: ResMed, Philips Healthcare, Invacare Corporation, Drive DeVilbiss Healthcare, Fisher & Paykel Healthcare.
Regulatory Approvals
- In the United States, respiratory durable medical equipment (DME) must pass a strict, step-by-step approval process that emphasizes safety, performance, and adherence to federal regulations before reaching the market. This ensures devices are reliable and effective for patient use.
- Key Players: Medtronic, GE HealthCare, Vyaire Medical, Hill-Rom Holdings, and 3M Health Care.
Patient Support and Services
- Patient support focuses on end-to-end home care, including device installation, continuous technical assistance, and monitoring of patient usage to ensure proper therapy adherence. These services help improve treatment effectiveness and patient comfort outside clinical settings.
- Key Players: Apria Healthcare, Lincare Holdings, AdaptHealth, Rotech Healthcare, and Owens & Minor.
Market Growth
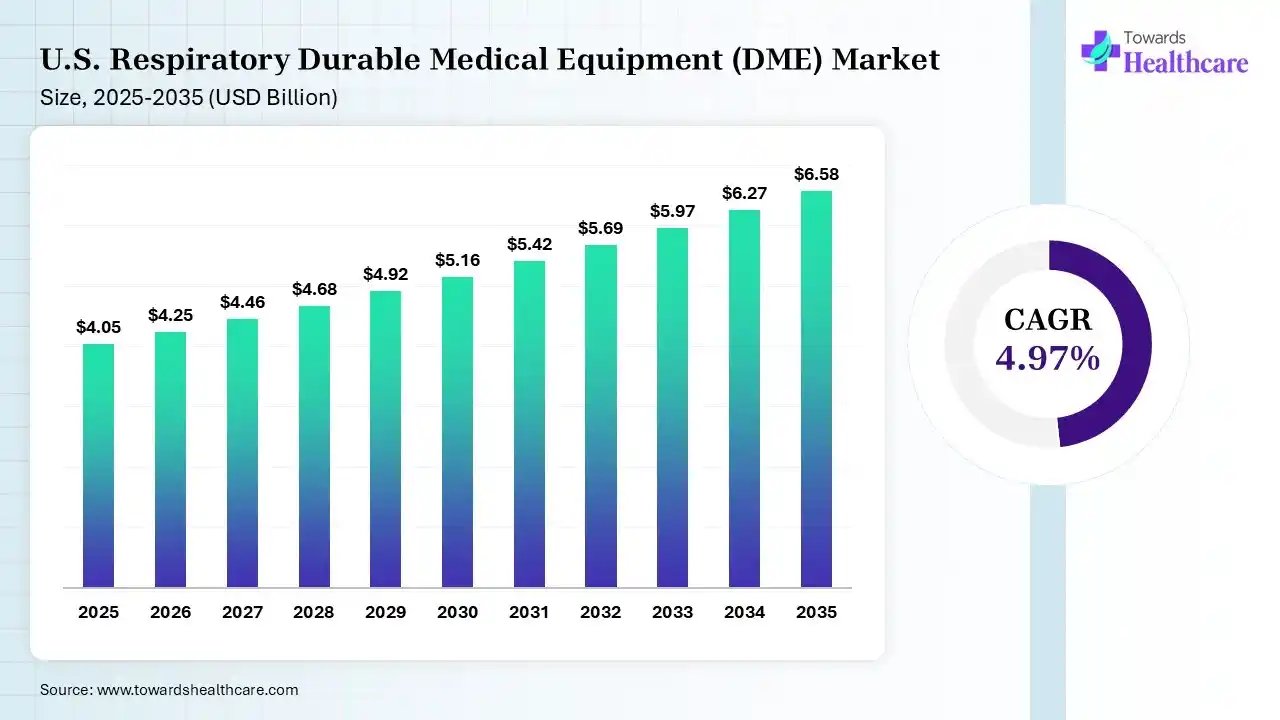
The U.S. respiratory durable medical equipment (DME) market size was estimated at USD 4.05 billion in 2025 and is predicted to increase from USD 4.25 billion in 2026 to approximately USD 6.58 billion by 2035, expanding at a CAGR of 4.97% from 2026 to 2035.
What are the Recent Developments in the U.S. Respiratory Durable Medical Equipment (DME) Market?
- In August 2025, Baxter International Inc. introduced an upgraded range of therapeutic beds aimed at lowering the risk of pressure injuries in healthcare environments, helping enhance patient care and overall treatment outcomes.
- In May 2025, Stryker Corp. launched a new range of advanced patient support surfaces equipped with wireless connectivity, enabling smoother data exchange between care systems and hospital networks to improve monitoring and efficiency.
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
