Latest Updates of Companies in the U.S. Sex Reassignment Hormone Therapy Market

From trends to key players, keep your entire U.S. Sex Reassignment Hormone Therapy Market view under one roof - Access the dashboard.
| Company | Headquarters | Latest Update |
| Viatris Inc. | United States | Viatris Inc. is aggressively increasing its portfolio of technically challenging and hormonal products. |
| AbbVie Inc. | United States | AbbVie’s AndroGel 1.62% remains a noteworthy product in its testosterone portfolio. |
| Pfizer Inc. | United States | Pfizer believes the coverage is based on a misleading and discerning reading of both the science and history of hormone therapy (HT) medicines. |
| ASCEND Therapeutics US, LLC. | United States | ASCEND Therapeutics US, LLC, is a field pharmaceutical organization focused on hormone therapies and a subsidiary of Besins Healthcare. |
| Novartis AG | United States | Novartis AG is focusing its portfolio on cardiovascular, neuroscience, immunology, and oncology, with significant developments in radioligand therapies rather than direct advancement. |
Supply Chain Analysis
R&D:
- Research and development (R&D) for U.S. sex reassignment hormone therapy involves new drug delivery systems, pharmacokinetic and pharmacodynamic modelling, new formulations and therapy optimization, clinical research and long-term safety research, and specialized diagnostic solutions.
- Key Players: Novartis AG and Eli Lilly
Manufacturing Processes:
- Manufacturing processes, including the manufacturing of feminizing hormones, production of masculinizing hormones, production of GnRH agonists, and quality control and supervisory standards.
- Key Players: ASCEND Therapeutics and Pfizer Inc.
Patient Services:
- Patient services in the U.S. sex reassignment hormone therapy include inclusive, multidisciplinary care, including mental health assessments, initial healthcare evaluations, and tailored hormone prescriptions.
- Key Players: Viatris Inc. and AbbVie Inc.
Recent Developments in the U.S. Sex Reassignment Hormone Therapy Market
- In February 2026, the U.S. Food and Drug Administration approved drug labeling changes to six menopausal hormone therapy products, also called hormone replacement therapy (HRT), to clarify the challenges and considerations for these drugs.
- In December 2025, the U.S. Department of Health and Human Services (HHS) announced a series of proposed government actions to carry out President Trump’s Executive Order directing HHS to end the practice of sex-rejecting procedures on children that expose young people to irreparable challenges.
Market Trends
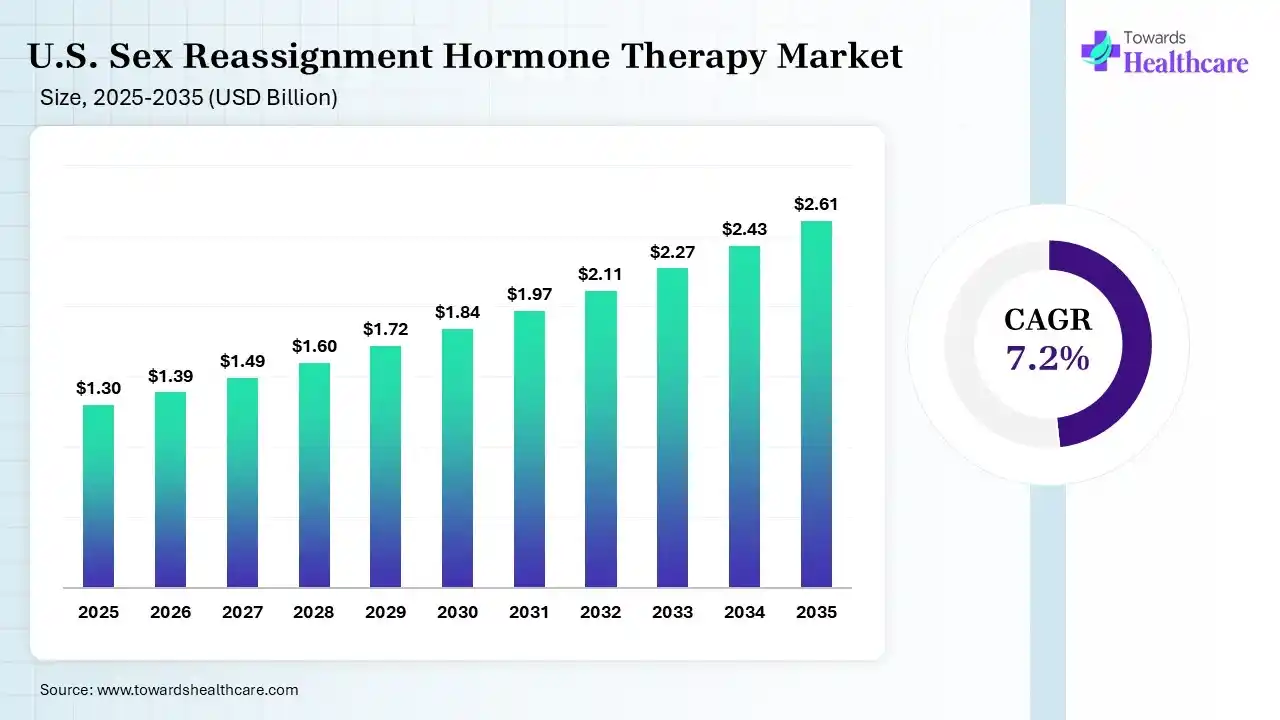
The U.S. sex reassignment hormone therapy market size was estimated at USD 1.3 billion in 2025 and is predicted to increase from USD 1.39 billion in 2026 to approximately USD 2.61 billion by 2035, expanding at a CAGR of 7.2% from 2026 to 2035. The U.S. sex reassignment hormone therapy market is growing because this therapy improves both gender incongruence and dysphoria by permitting individuals to express their gender identity and supportive changes in primary and secondary sex physiognomies
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
