Latest Updates of Key Players in the U.S. Ureteral Access Sheath Market

Centralize Your U.S. Ureteral Access Sheath Market Intelligence and Stay Ahead - Download For Free
| Company | Headquarters | Latest Update |
| Applied Medical Resources Corporation | United States | Applied Medical specializes in advanced access, retrieval, and closure tools for minimally invasive surgery. |
| BD | United States | In August 2025, BD announced that its Urology and Critical Care (UCC) and Surgery business units are the first in the medical technology sector to achieve a diamond-level rating for all seven supply chain resilience domains of the Healthcare Industry Resilience Collaborative’s (HIRC) Resiliency Badging program |
| Merit Medical Systems, Inc. | United States | In August 2025, Merit Medical Systems, Inc. announced the United States commercial release of the Prelude Wave hydrophilic sheath introducer with SnapFix securement technology. |
| Boston Scientific Corporation | United States | In March 2025, Boston Scientific Corporation announced it had entered into a definitive agreement to acquire SoniVie Ltd., a privately held healthcare tools company that has developed the TIVUS Intravascular Ultrasound System. |
| Rocamed | Monaco | Rocamed's first transformative build-up was the acquisition of Delmont Imaging, a renowned specialist in gynecological medical devices. |
| Well Lead Medical | United States | Well Lead Medical Co., Ltd. remains a dominant player in the global urological consumables market, specifically known for its ClearPetra suction-evacuation system. |
Recent Developments in the U.S. Ureteral Access Sheath Market
- In October 2025, Dornier MedTech America (DMTA) launched in the United States its Dornier Hoover Flexible and Navigable Suction Ureteral Access Sheath (FANS) and Dornier Axis II Slim single-use ureteroscope with a 5.6Fr. distal tip. Both products were cleared by the U.S. Food and Drug Administration.
- In September 2025, Dornier MedTech America (DMTA) announced the full U.S. commercial launch of its Dornier Hoover Flexible and Navigable Suction Ureteral Access Sheath (FANS) and Dornier Axis II Slim single-use ureteroscope with a 5.6Fr. distal tip.
- In September 2025, Calyxo, Inc., a healthcare tools organization advancing innovative solutions for kidney stone treatment, today announced the launch of Reverse Deflection on its second-generation CVAC System, an improvement that empowers endourologists to perform the SURE technology using their preferred steering mode.
U.S. Ureteral Access Sheath Market Trends for 2026
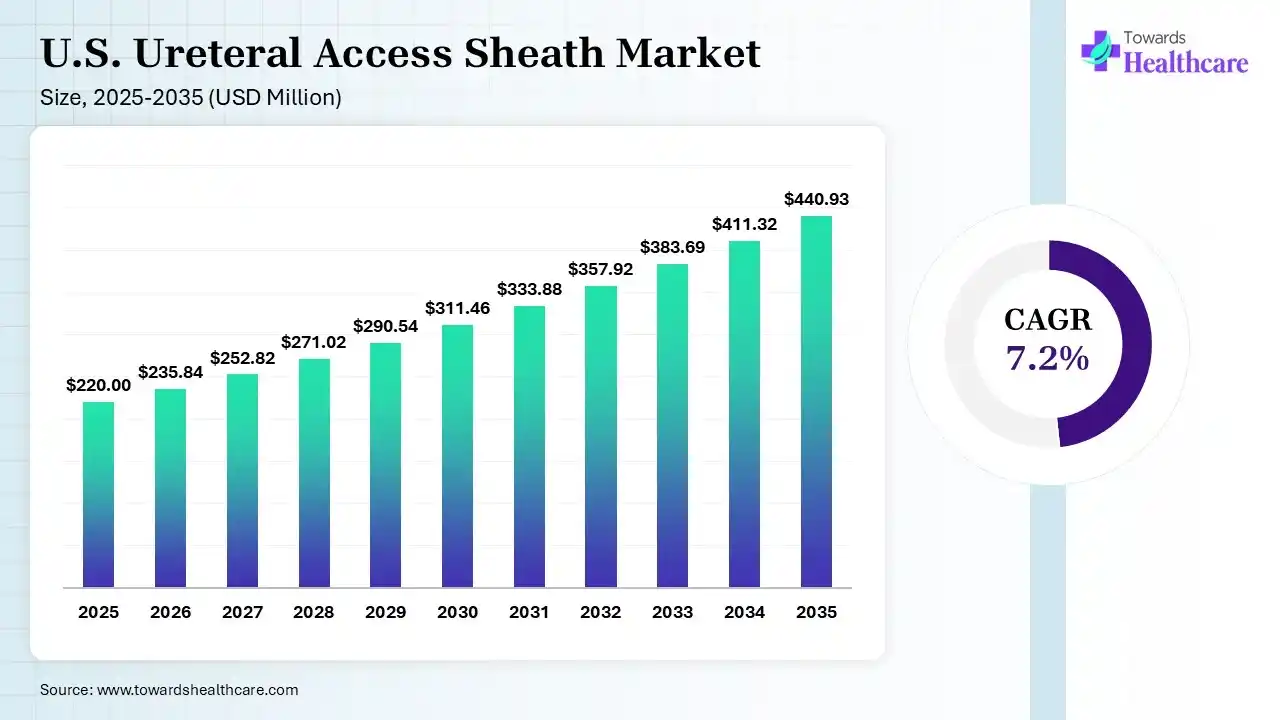
The U.S. ureteral access sheath market size was estimated at USD 220 million in 2025 and is predicted to increase from USD 235.84 million in 2026 to approximately USD 440.93 million by 2035, expanding at a CAGR of 7.2% from 2026 to 2035.
Supply Chain Analysis
R&D:
- Research and development (R&D) for U.S. ureteral access sheath involves design and product development, testing and mechanical benchmarking, clinical and regulatory services, and specialized engineering support.
- Key Players: Boston Scientific Corporation and Cook Medical
Manufacturing Processes:
- Manufacturing processes in the U.S. ureteral access sheath involve tube extrusion, reinforcement, tip and hub assembly, surface coating, sterilization, and packaging.
- Key Players: Coloplast Corp and Olympus Corporation
Patient Services:
- Patient services of U.S. ureteral access sheath, including improving safety, lowering operative time, and reducing challenges during retrograde intrarenal surgery (RIRS) and ureteroscopy, are characteristic of kidney stone management.
- Key Players: Teleflex Incorporated and Karl Storz Endoscopy GmbH
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
