The widespread adoption and development of diverse, but robust, reusable, doctor-ordered medical devices for repeated home use, which support different issues & injuries, is termed as the durable medical equipment (DME). Moreover, this sophisticated equipment possesses a long-term lifespan, i.e., basically 3+ years & also favourable for remote use, like wheelchairs, walkers, ventilators, and oxygen concentrators.
In brief, the complete U.S. durable medical equipment industry is driven by the faster growth of the geriatric population, increasing respective chronic disease cases & an immersive step towards remote care.s
Thorough Look at the Significant Drivers of the Durable Medical Equipment in the U.S.
Apart from the eventual expansion of the ageing population, the respective industry has been experiencing several catalysts for the prospective progressions and breakthroughs, such as
Across the U.S. area, a huge population, including patients & providers, strongly prefers various advanced home care instead of hospital stays, which ultimately constrains spending and raises comfort among them. Also, this further escalates the need for home-use oxygen concentrators, hospital beds, and mobility solutions.
Another key driver is the increased healthcare expenditure, connected with broader insurance coverage and Medicare reimbursement, which enhances access to and leverage expensive, advanced DME.
However, numerous leading firms are emphasizing major technological breakthroughs, such as integrating AI & IoT into devices, like smart wheelchairs and connected monitoring tools, enabling expanded patient care, predictive maintenance, & data-driven insights.
Ongoing Diversity in Technologies with Extensive Focus on Patient Care
More specifically, this modern era is increasingly seeking more sophisticated mobility aids, such as the latest power wheelchairs, which incorporate modular platforms unveiled by Invacare. This rollout accelerates manoeuvrability, whereas advanced prosthetic devices use sensors to excellently mimic natural limb movement.
Besides this, a prominent effort includes an emphasis on transforming connected respiratory care, such as Portable Oxygen Concentrators (POCs) & modern nebulizers, which enables customised oxygen therapy, and also capturing patient breathing patterns & transferring usage data.
The U.S. is considered a substantial industrial country in the healthcare sector, so it has been taking efforts to develop smart hospital beds and mattresses from developers, such as Baxter International & Stryker Corp. They further use wireless communication to track patient data, lower pressure injuries, and aim at integration with hospital landscapes.
On the other hand, the worldwide expanding activities in the progression of remote monitoring are also involved in the U.S., which spurs various novel telehealth-enabled DME, like connected blood pressure monitors and scales, to enable real-time monitoring of chronic illnesses in home-based settings.
Spotlighting Home-Based DME Solutions in the U.S. & Their Indications
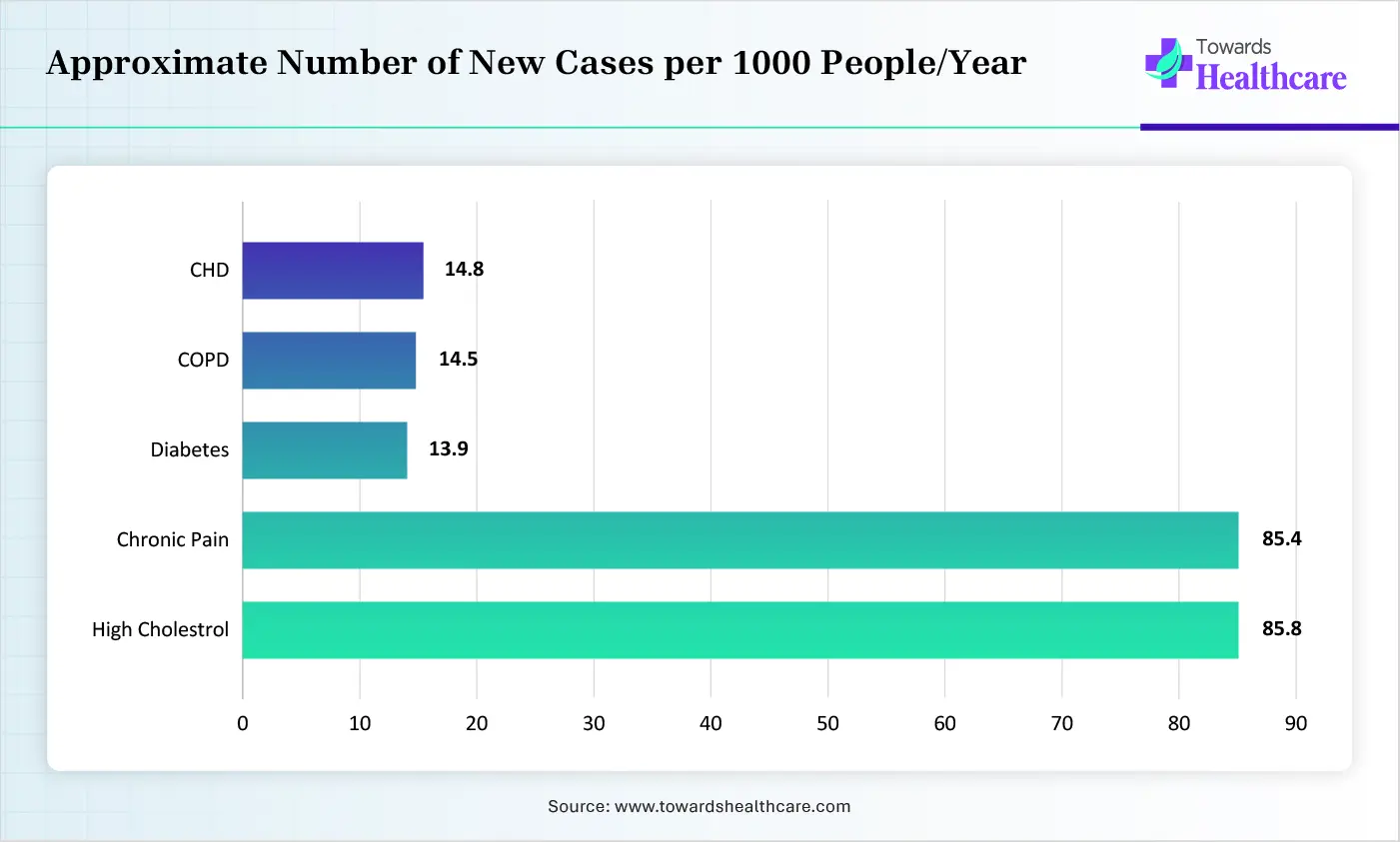
First of all, the notable emergence of advanced remote monitoring approaches refers to reusable, doctor-ordered medical devices for long-term use at home in treating rising new cases of chronic concerns, like high cholesterol, chronic pain, diabetes, COPD, CHD, etc. This primarily comprises wheelchairs, oxygen equipment, & hospital beds, which must be durable & applicable for medical purposes, also essential for living alone.
- Particular involvement of MasVida Health demonstrated CPAP devices that automatically adjust pressure based on breath patterns & connect to the cloud. Subsequently, ResMed implemented an AI-assisted CPAP with real-time sleep monitoring.
- Recently, the FDA approved Rhythm Express Remote Cardiac Monitoring System (RX-1 mini) & also Biobeat's wearable, FDA-cleared for remote monitoring of critical signs, cuffless blood pressure, & oxygen saturation, gained traction.
Inclusion of the Key Aspects of the Durable Medical Equipment in the U.S.
A crucial part of the U.S. DME is the incorporation of Medicare reimbursement, which is specifically controlled through Part B. This mainly covers what is deemed medically essential, supportive for home application, and created for long-term usage. Additionally, this encompasses reimbursement based on fee schedules or competitive bidding, stringent documentation requirements, & mandatory participation of suppliers.
- Prominently, Medicare Part B includes typical payments of 80% of the Medicare-approved amount after the beneficiary completes the annual deductible.
- Furthermore, this leverages competitive bidding to find payment rates for particular items to reduce spending, which requires suppliers to fulfil unique quality standards.
- Over this, suppliers must be enrolled in Medicare, which covers a National Provider Identifier (NPI), & follow specific coverage guidelines, i.e., NCDs or LCDs.
- These Medicare Reimbursement describes the differentiation between rented items and purchased ones, which impacts the payment structure.
- These suppliers should have orders signed by a doctor and related medical records, exploring the continued need to receive payment.
Arising Substantial Obstacles in the Consistent Progression of DME in the U.S.
Certain challenges are limiting the transformation of the U.S. durable medical equipment industry, including often policy upgradation from Medicare and private insurers, which leads to increased claim denial among DME suppliers. This also merges coding errors, like inappropriate HCPCS codes or incorrect documentation of medical necessity, which frequently delay or pause payment.
Another barrier is global interruption, lack of production, and escalation has resulted in major shortages, especially in the care of neonatal & pediatric devices. Somehow, manufacturers need to follow complex, modifying regulatory landscapes (FDA) while ensuring high-quality standards, which can exceed the time to market for innovative products.
Notable Regulatory Updates of the U.S. DME in 2025-2026
| QMSR Execution | Where the FDA substitutes the 21 CFR Part 820 Quality System Regulation with the Quality Management System Regulation (QMSR), using ISO 13485:2016 standards. |
| DMEPOS Competitive Bidding Program | CMS has a goal to reintroduce this program with a potential transition to annual reaccreditation, new payment methodologies for key items, & early authorization exemptions for compliant suppliers. |
| Electronic Submissions | Particularly, electronic submission of 510(k)s via the eSTAR program has been mandatory for De Novo requests beginning after Oct 1, 2025. |
| General Wellness Devices | This has cleared that low-risk, noninvasive physiologic trackers may be treated as general wellness products instead of regulated devices if they omit unique medical claims. |
Immersive Government Incentives in the U.S. Durable Medical Equipment Sector
- Under the FDA Breakthrough Devices Program, the FDA has authorized 1,246 Breakthrough Device designations as of December 31, 2025. This also covers sprint discussions and preferred review for marketing submissions.
- Another program called FDA TEMPO Pilot was unveiled in late 2025, which shows a first-of-its-kind digital health pilot that strengthens access to chronic disease technologies by enabling manufacturers to report real-world performance data while facilitating devices explored by Medicare.
- Moreover, CMS will launch the MAHA ELEVATE Model in October 2026, with a $100 million incentive that tests whole-person care measures for Medicare beneficiaries, probably informing prospective coverage for sophisticated DME.
Outline of Recent Rollouts of DME in the U.S.
- In December 2025, Pride Mobility launched three new power wheelchairs, i.e., Jazzy Max, Jazzy Swift, and Jazzy Injection, to foster an on-the-go lifestyle.
- In August 2025, Airchair, a major manufacturer of onboard wheelchairs, rolled out the Airchair II to comply with U.S. Department of Transportation Regulation 14 CFR Part 382.65 & also to raise passenger comfort.
- In June 2025, Invacare Corp. unveiled an innovative limited-edition product, the Kuschall Element active manual wheelchair, with improved maneuvering, high-quality materials, & an emphasis on performance.
Prominent U.S. Players & Their Lucrative Leap-Forward in DME
| Medtronic | It specializes in diabetes management & cardiac care, to develop advanced insulin pumps, pacemakers, & Deep Brain Stimulation (DBS) therapy for epilepsy. |
| Abbott Laboratories | A firm popular for the FreeStyle Libre, a transformative continuous glucose monitoring system that removes the need for finger sticks. |
| Stryker Corporation | This mainly executes diversity in orthopaedic groundbreaking, such as Mako Robotics for joint replacements and smart bed technology. |
| ResMed | It has been putting efforts into cloud-connected devices for treating sleep apnea and other respiratory issues. |
| Invacare Corporation | This company facilitates robust home medical equipment, especially powered mobility devices & advanced wheelchairs. |
| Sunrise Medical | It unveiled the QUICKIE Q50 R Carbon, an ultra-lightweight, high-performance foldable power wheelchair. |
| Abilitech Medical | This offers wearable, functional assistance devices for patients with upper-limb neuromuscular disorders. |
Conclusion
According to the CDC, the number of people aged 50+ with a chronic condition is estimated to increase by 99.5% by 2050, which will reach nearly 142.66 million. This is showing the prospective opportunities in the diverse chronic concerns, especially diabetes, heart diseases, and others. This will reinforce the comprehensive advances in various durable medical equipment, such as blood pressure & diabetes monitors, mobility aids, with extensive home-based approaches, like wearables. Many leading manufacturers & suppliers are shifting towards RPM solutions by complying with the updated regulatory framework, with a focus on Medicare reimbursement policies. So, the U.S. will bolster novelty in the DME sector & also promote variations in much-needed equipment.
Expert Insights
According to a recent report on the U.S. Durable Medical Equipment Market, published on Towards Healthcare, experts highlight steady growth driven by the rising burden of chronic diseases, an expanding geriatric population, and increasing preference for home-based care. The market is projected to surpass USD 108 billion by 2033, supported by strong demand for monitoring, therapeutic, and mobility devices. Advancements in connected and portable technologies are enhancing patient outcomes and enabling remote care. Favorable reimbursement policies and hospital-to-home transitions further boost adoption, while ongoing innovation and strategic partnerships continue to strengthen long-term market expansion across the United States healthcare ecosystem.
About the Experts

Aditi Shivarkar
Aditi leads as Vice President at Towards Healthcare and brings over 15 years of experience in healthcare research, innovation, and strategy. She works closely with data from across the healthcare sector and turns it into clear direction that companies can actually use. Her work covers pharmaceuticals, medical devices, and digital health. She helps businesses understand where the market is going and how to respond with confidence. Aditi focuses on practical thinking, strong decision-making, and delivering real results that make a difference.

Aman Singh
Aman Singh brings over 13 years of experience in healthcare research and consulting. He studies global healthcare trends and keeps a close eye on areas like biotech, AI in healthcare, and new treatment approaches. At Towards Healthcare, he leads the research team and makes sure the work stays accurate, useful, and easy to understand. Aman breaks down complex changes in the industry and helps businesses make smart, informed decisions.

Piyush Pawar
Piyush Pawar works as Senior Manager for Sales and Business Growth at Towards Healthcare, with more than 10 years of experience in the healthcare space. He works directly with clients and helps them find the right research for their needs. He makes sure clients understand the insights and know how to use them in their business. Piyush builds strong relationships and focuses on helping companies grow by turning research into clear, practical action.




 Request Consultation
Request Consultation


